Effect of Cultivation Stages of Hericium erinaceus on the Contents of Major Components and α-Glucosidase Inhibitory Activity
Abstract
Hericium erinaceus, also known as lion’s mane mushroom, is an edible and medicinal mushroom that belongs to the family Hericiaceae. We previously reported hericene A as an anti-diabetic constituent of H. erinaceus and the effect of cultivation substrates on its content was investigated. As the continuation, the contents of five major compounds such as hericenes A-D, which exerted α-glucosidase inhibitory activity, together with ergosterol were investigated depending on cultivation stages. H. erinaceus was cultured for 25 days (5 stages) to induce fruiting bodies, and the contents of the compounds at each stage were quantified. All the five compounds were detected in fruiting body by HPLC analysis. Among the hericene derivatives in the mushroom, the content of hericene A was the highest, followed by hericene C and the content of hericenes B and D was relative low. All four hericene derivatives present in the highest content at stage 4 whereas the content of ergosterol was highest at stage 5. The highest α-glucosidase inhibitory activity of H. erinaceus was measured at stage 4, which correlates with the contents of hericene derivatives. Conclusively, H. erinaceus with better efficacy and high content of active constituents can be secured by the optimization of cultivation conditions.
Keywords:
Hericium erinaceus, cultivation perioid, hericenes A-D, ergosterol, quantitation, anti-diabetesIntroduction
Mushrooms, filamentous fungi with fruiting bodies, are rich in nutrients and various active substances.1 Mushrooms originally grow in wild, and also have been obtained through cultivation for sustainable supply.2-4 Hericium erinaceus, commonly called lion’s mane mushroom, belongs to the family Hericiaceae and has been used in folklore for the treatment of metabolic diseases and gastrointestinal disorders and to improve memory and enervation.5, 6 As constituents, H. erinaceus was reported to contain various bioactive constituents such as hericenones and erinacines as well as polysaccharides and sterols.7-10 Recent study also suggested the beneficial effects of this mushrooms such as neurotrophic, antibacterial, anti-cancer and anti-inflammatory activity.11-15
Due to its characteristic components and beneficial effects, we have been interested in this mushrooms. We previously characterized the neurotrophic and anti-diabetic components including newly reported ones.16,17 In addition, the changes of bioactive component according to the cultivation conditions including medium composition were also suggested.18
Mushrooms contain diverse compounds which contributed to its biological activity, sometimes by synergistic effects of various compounds.19 The content of compounds varies during cultivation period, which showed differential patterns depending on each compound.20-22 The content of each compound affects the efficacy of mushroom, which need to be optimized for its harvest time considering targeted efficacy.
As a continuation of our research for securing mushrooms with better activity, the effects of cultivation periods on the activity together with its composition was investigated in this study. α-Glucosidase are essential for the degradation of carbohydrate to monosaccharide which can be easily absorbed. Therefore, inhibition of α-glucosidase is an important therapeutic due to its hypoglycemic effect by the decrease of the carbohydrates absorption.23 We previously reported the α-glucosidase inhibitory activity of H. erinaceus and characterized the active component.17 To find the best harvest time for α-glucosidase inhibition, H. erinaceus was harvested at 5 different cultivation stages and the α-glucosidase inhibitory activity was compared. In addition, the changes of major constituents were investigated by quantitation using HPLC analysis. The correlations between contents of compounds and α-glucosidase inhibitory activity were also suggested. Therefore, these results could be applied for securing H. erinaceus with high α-glucosidase inhibitory activity and for quality control by the quantitation of active constituents.
Experimental
Strain of H. erinaceus – The strain of H. erinaceus was obtained from Jeollanamdo Agricultural Research Institute (Naju, Korea). They were grown on the potato dextrose agar (PDA 200 g/l diced potatoes; 20 g/l glucose; 15 g/l agar) medium at 25oC for regular subculture or on different culture conditions as indicated.
Mushroom cultivation with different cultivation days – Growing media composed of sawdust (SD, 49%) as a base substrate supplemented with cottonseed hulls (CH, 19%), rice bran (RB, 7%), cottonseed meal (CM, 12%) and beet (BT, 13%) was sufficiently mixed with water by a mixer, and the excess water was drained to reach a substrate moisture level of about 70%. Then, each growing medium (1 kg of wet weight) was packed into a propylene bottle and autoclaved at 121oC for 90 min.
After sterilization, the growing media were inoculated with spawn and incubated at 25oC in the dark with 80% relative humidity for mycelial colonization for 37 days. Soon after the hyphae of H. erinaceus reached the bottom of the bottles, the bottles were moved to a cold room with light at 18oC with 90% relative humidity to induce fructification. Ventilation, and relative humidity were controlled in the experimental mushroom house during the mushroom production period. The bottles were then opened to allow the development of fruiting bodies.
For the measurement the effect of incubation days for fruiting body growth on the content of major components, mycelium of H. erinaceus was grown for 37 days and then fruiting bodies were induced for further 9, 13, 17, 21 or 25 days, namely stages 1-5, respectively.
Preparation of sample solutions – Dried fruiting bodies were pulverized and 1 g of samples was extracted with 10 mL ethyl acetate for 24 h at room temperature. The supernatant was evaporated in vacuo to yield ethyl acetate extract. Ethyl acetate extract of H. erinaceus was dissolved in methanol at a concentration of 10 mg/mL and was filtered through a 0.45 mm PTFE filter. The solutions were stored at -20oC until HPLC analysis.
Purification of major components – Major components of H. erinaceus were purified from the ethyl acetate extract for the quantitation and evaluation of biological activities. Hericenes A and D were isolated from the fruiting bodies of H. erinaceus as previously reported.17 Ergosterol, hericene B and hericene C were purified by semi-preparative HPLC eluting with methanol-water (97:3) with retention times of 17.9, 36.2 and 48.5 min, respectively.
Measurement of α-glucosidase inhibitory activity – α-Glucosidase inhibitory assays were performed using α-glucosidase (from Saccharomyces cerevisiae, 1 U/mL) and p-nitrophenyl α-D-glucopyranoside, as reported previously.16 The amount of p-nitrophenol cleaved by the enzyme was determined by measuring the absorbance at 405 nm in a 96-well microplate reader.
Quantitation of major components using HPLC analysis – A stock solution was prepared at the concentration of 1.0 mg/mL of each component and diluted appropriately for HPLC analysis. Waters HPLC A system equipped with Waters 600 Q-pumps, a 996 photodiode array detector, and Waters Empower software was used for the quantitation of major components. The separation was carried out on RP-C18 column (5 µm, 10 × 150 mm) using isocratic elution of methanol-water (97:3). The solvent flow rate of 2 ml/min was maintained throughout the analysis and the injection volume was 10.0 µL. All the analysis was carried out at room temperature at a wavelength of 292 nm with a run time of 60 min.
Result and Discussion
To analyze the efficacy and compositional changes according to the growth period, fruiting bodies of H. erinaceus were harvested in 5 stages depending on cultivation days. As shown in Fig. 1A, the size of the fruiting bodies grew up to stage 4, and then maintained thereafter.
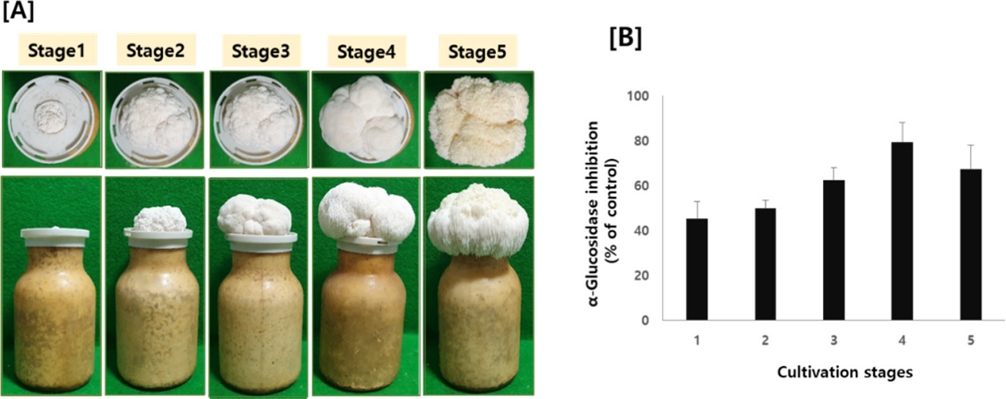
[A] Morphology and [B] α-glucosidase inhibitory activity of H. erinaceus depending on cultivation stages.
Next, α-glucosidase inhibitory activity of H. erinaceus from different cultivation days was measured. Although all the samples from different cultivation days showed α-glucosidase inhibitory activity, the efficacy was quite different, as shown in Fig. 1B. Especially, the inhibitory effect gradually increased and showed the strongest inhibition at stage 4, and then decreased at stage 5.
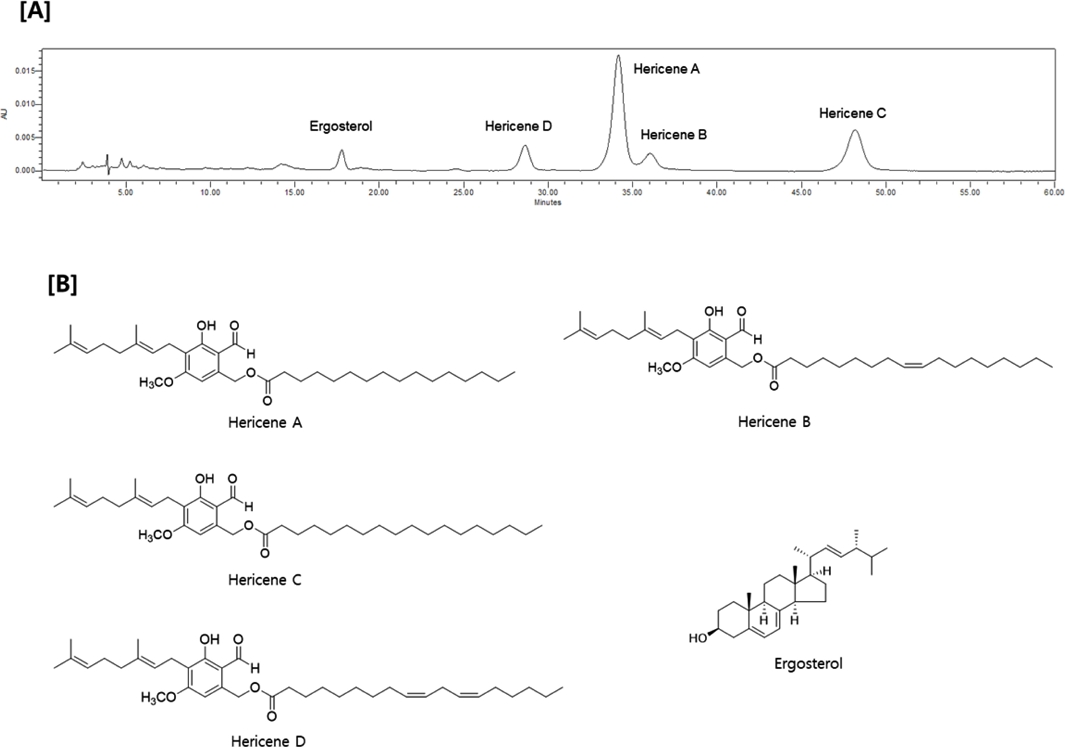
Since the α-glucosidase inhibitory activity of this mushroom was changed depend on the cultivation period, we tried to measure the change in the components. For the quantitation of the constituents of H. erinaceus, analytical method was established using HPLC analysis. HPLC chromatogram of the extract of H. erinaceus showed five major peaks, which identified as ergosterol and four hericene derivatives, hericenes A – D (Fig. 2) by further separation and characterization by spectroscopic analysis together with the comparison with literature values.16,24
For the quantitation of the constituents of H. erinaceus, analytical method was established using HPLC analysis. Calibration curves were obtained using the purified compounds and used for quantitation of compounds in the extract of H. erinaceus. Next the content of each major component in different H. erinaceus samples were quantitated using the established HPLC analysis. As shown in Fig. 3A, the changes of each compound was well detected in the HPLC chromatograms of different extracts. The content of the hericene derivatives gradually increased as the cultivation days, showing the highest content in stage 4, and then decreased thereafter. When looking at the content of each compound, the content of HA was the highest, followed by HC, HD and HB (Fig. 3B). On the other hand, the content of ergosterol increased slowly and showed the highest content in the later stage. As such, the total content of hericene derivatives showed the highest content in stage 4, whereas the content of ergosterol was the highest at stage 5 (Fig. 3C). Thus, it was confirmed that the content of hericene derivatives and ergosterol showed different patterns depending on cultivation days.
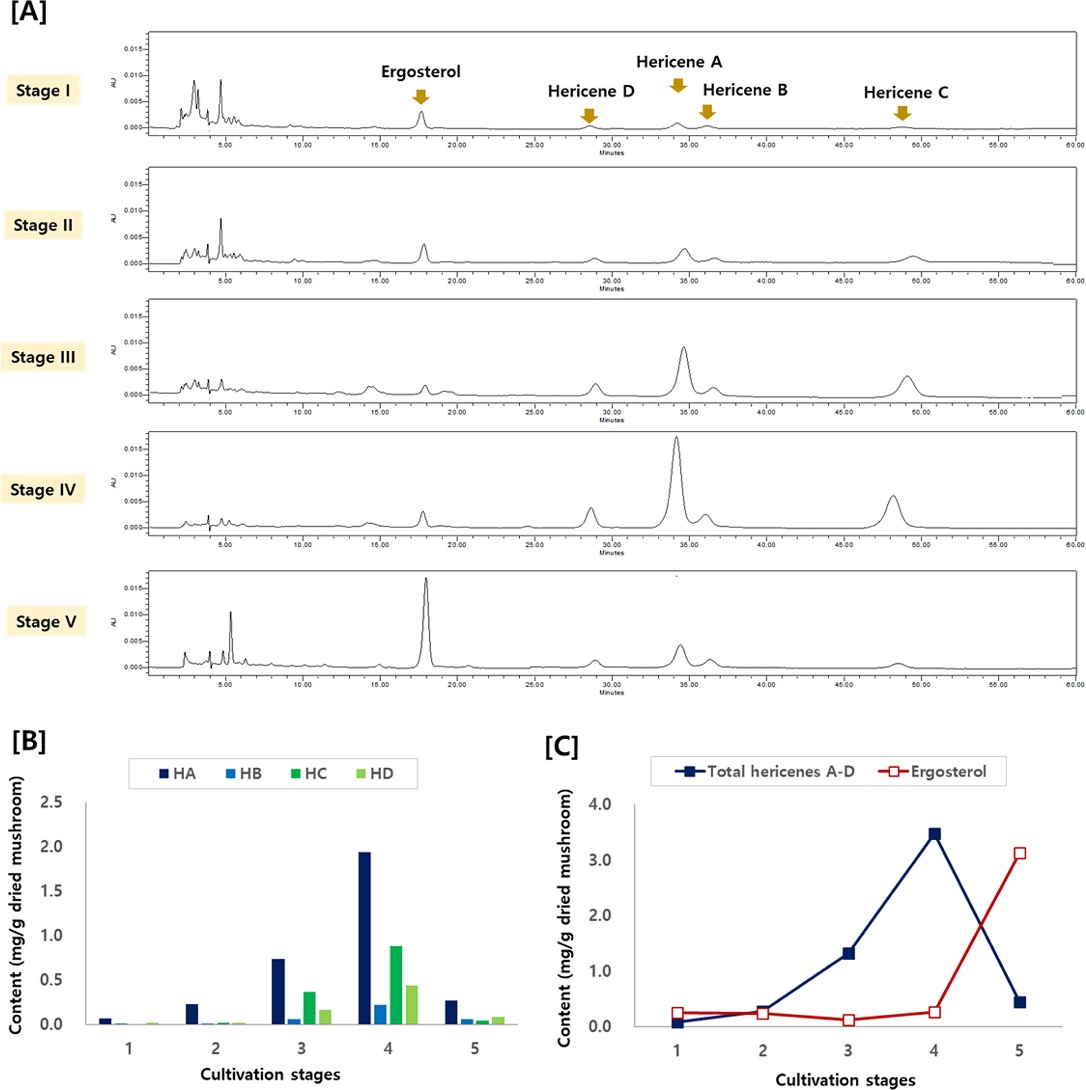
[A] HPLC chromatograms, [B] content of each hericenes A-D (HA, HB, HC and HD, respectively) and [C] total hericenes derivatives and ergosterol depending on cultivation stages of H. erinaceus.
Since the inhibitory effect and the content of the major compounds showed differences in each cultivation stages, the correlation between the content of each compound and the efficacy was analyzed. First, the inhibitory efficacy of the isolated compounds was evaluated. All the hericene derivatives, HA, HB, HC and HD showed excellent α-glucosidase inhibitory efficacy with IC50 values of 6.7, 1.0, 0.9 and 3.9 μM, respectively. However, ergosterol did not exert inhibitory efficacy against α-glucosidase (IC50>100 μM). As expected from the α-glucosidase inhibitory efficacy of major compounds, correlation between the α-glucosidase inhibitory activity of H. erinaceus and the total content of hericene derivatives were observed with R2 = 0.7197, whereas low correlation with the content of ergosterol (Fig. 4).
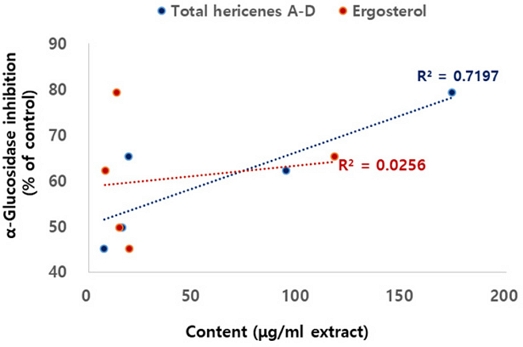
Correlation between α-glucosidase inhibitory activity and the content of total hericenes A-D and ergosterol.
Although many mushrooms have been collected traditionally in wild, it is obtained by cultivation these days for its sustainable supply. The quality of the mushroom is greatly affected by not only the species and strains but also the cultivation condition, thus, many researchers have attempted to optimize the culture condition for better products.17,25-27 The cultivation period is one of the important factors during cultivation. It plays an important role for its size, shape and freshness. In addition, it also affects the composition of the mushroom which resulted in the efficacy. Our present study showed that difference of the cultivation period at which the content of hericene derivatives and ergosterol were maximal amounts. After the content of hericene derivatives was maximal, ergosterol showed the highest content thereafter. Our data also suggested that the content of hericene derivatives plays an important role in anti-diabetic efficacy. Although ergosterol did not show anti-diabetic effect in our assay system, it is known as a precursor of vitamin D. When irradiated with UV, ergosterol in the mushroom is converted to vitamin D, so it is reported that a high content of ergosterol has a protective effect on osteoporosis.28,29 Conclusively, H. erinaceus with better efficacy and increased content of active constituents can be secured by the optimization of cultivation conditions.
Acknowledgments
This work was supported by the support of R&D Program for Forest Science Technology (Project No. 2023506A00-2323-AB01) provided by Korea Forest Service (Korea Forestry Promotion Institute) and by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT, 2022R1A2C1008081).
Conflict of interest
The authors declare that there is no conflict of interest.
References
-
Ha, J. W.; Kim, J.; Kim, H. W.; Jang, W.; Kim, K. H. Nat. Prod. Sci. 2020, 26, 118-131.
[https://doi.org/10.20307/nps.2020.26.2.118]

-
Das, S. K.; Masuda, M.; Sakurai, A.; Sakakibara, M. Fitoterapia 2010, 81, 961-968.
[https://doi.org/10.1016/j.fitote.2010.07.010]

- Berovic, M. Adv. Biochem. Eng. Biotechnol. 2019, 169, 3-25.
-
Berovic, M.; Podgornik, B. B.; Gregori, A. Int. J. Med. Mushrooms 2022, 24, 1-22.
[https://doi.org/10.1615/IntJMedMushrooms.2021042445]

-
Sabaratnam, V.; Kah-Hui, W.; Naidu, M.; Rosie David, P. J. Tradit. Complement. Med. 2013, 3, 62-68.
[https://doi.org/10.4103/2225-4110.106549]

-
Khan, M. A.; Tania, M.; Liu, R.; Rahman, M. M. J. Complement. Integr. Med. 2013, 10, 253-258.
[https://doi.org/10.1515/jcim-2013-0001]

-
Kawagishi, H.; Ando, M.; Sakamoto, H.; Yoshida, S.; Ojima, F.; Ishiguro, Y.; Ukai, N.; Furukawa, S. Tetrahedron Lett. 1991, 32, 4561-4564.
[https://doi.org/10.1016/0040-4039(91)80039-9]

-
Kawagishi, H.; Shimada, A.; Shirai, R.; Okamoto, K.; Ojima, F.; Sakamoto, H.; Ishiguro, Y.; Furukawa, S. Tetrahedron Lett. 1994, 35, 1569-1572.
[https://doi.org/10.1016/S0040-4039(00)76760-8]

-
Krzyczkowski, W.; Malinowska, E.; Suchocki, P.; Kleps, J.; Olejnik, M.; Herold, F. Food Chem. 2009, 113, 351-355.
[https://doi.org/10.1016/j.foodchem.2008.06.075]

-
Han, Z.-H.; Ye, J.-M.; Wang, G.-F. Int. J. Biol. Macromol. 2013, 52, 66-71.
[https://doi.org/10.1016/j.ijbiomac.2012.09.009]

-
Mizuno, T.; Wasa, T.; Ito, H.; Suzuki, C.; Ukai, N. Biosci. Biotechnol. Biochem. 1992, 6, 347-348.
[https://doi.org/10.1271/bbb.56.347]

-
Liu, X. T.; Winkler, A. L.; Schwan, W. R.; Volk, T. J.; Rott, M.; Monte, A. Planta Med. 2010, 76, 464-466.
[https://doi.org/10.1055/s-0029-1186227]

-
Liang, B.; Guo, Z.; Xie, F.; Zhao, A. BMC Complement. Altern. Med. 2013, 13, 253.
[https://doi.org/10.1186/1472-6882-13-253]

-
Kim, M. S.; Sharma, B. R.; Rhyu, D. Y. Nat. Prod. Sci. 2016, 22, 175-179.
[https://doi.org/10.20307/nps.2016.22.3.175]

- Hong, S. M.; Yoon, D. H.; Lee, M. K.; Lee, J. K.; Kim, S. Y. Oxid. Med. Cell. Longev. 2022, 2022, 9973678.
-
Lee, S. K.; Ryu, S. H.; Turk, A.; Yeon, S. W.; Jo, Y. H.; Han, Y. K.; Hwang, B. Y.; Lee, K. Y.; Lee, M. K. J. Ethnopharmacol. 2020, 262, 113197.
[https://doi.org/10.1016/j.jep.2020.113197]

-
Ryu, S. H.; Hong, S. M.; Khan, Z.; Lee, S. K.; Vishwanath, M.; Turk, A.; Yeon, S. W.; Jo, Y. H.; Lee, D. H.; Lee, J. K.; Hwang, B. Y.; Jung, J. K.; Kim, S. Y.; Lee, M. K. Bioorg. Med. Chem. Lett. 2021, 31, 127714.
[https://doi.org/10.1016/j.bmcl.2020.127714]

-
Turk, A.; Kim, B. S.; Ko, S. M.; Yeon, S. W.; Ryu, S. H.; Kim, Y. G.; Hwang, B. Y.; Lee, M. K. Nat. Prod. Sci. 2021, 27, 187-192.
[https://doi.org/10.20307/nps.2021.27.3.187]

-
Jen, C. I.; Su, C. H.; Lu, M. K.; Lai, M. N.; Ng, L. T. J. Food Biochem. 2021, 45, e13694.
[https://doi.org/10.1111/jfbc.13694]

-
Hu, D.; Yang, X.; Hu, C.; Feng, Z.; Chen, W.; Shi, H. ACS Omega 2021, 6, 29506-29515.
[https://doi.org/10.1021/acsomega.1c03561]

-
Deng, K.; Lan, X.; Fang, Q.; Li, M.; Xie, G.; Xie, L. Front. Mol. Biosci. 2021, 8, 632341.
[https://doi.org/10.3389/fmolb.2021.632341]

-
Turk, A.; Yeon, S. W.; Ryu, S. H.; Ko, S. M.; Kim, B. S.; Hwang, B. Y.; Lee, M. K. Sci. Hortic. 2021, 288, 110407.
[https://doi.org/10.1016/j.scienta.2021.110407]

-
Baron, A. D. Diabetes Res. Clin. Pract. 1998, 40, S51-S55.
[https://doi.org/10.1016/S0168-8227(98)00043-6]

-
Chen, B.; Han, J.; Bao, L.; Wang, W.; Ma, K.; Liu, H. Planta Med. 2020, 86, 571-578.
[https://doi.org/10.1055/a-1146-8369]

-
Cui, J. D. Crit. Rev. Biotechnol. 2015, 35, 475-484.
[https://doi.org/10.3109/07388551.2014.900604]

-
Li, X.; Pang, Y.; Zhang, R. Bioresour. Technol. 2001, 80, 157-161.
[https://doi.org/10.1016/S0960-8524(00)00170-X]

-
Oh, J.; Yoon, D. H.; Shrestha, B.; Choi, H. K.; Sung, G. H. J. Microbiol. 2019, 57, 54-63.
[https://doi.org/10.1007/s12275-019-8486-z]

-
Tang, J.; Qian, Z.; Wu, H. Bioresour. Technol. 2018, 268, 60-67.
[https://doi.org/10.1016/j.biortech.2018.07.128]

-
Jiang, Q.; Zhang, M.; Mujumdar, A. S. Trends Food Sci. Technol. 2020, 105, 200-210.
[https://doi.org/10.1016/j.tifs.2020.09.011]