Areca catechu L.: A Comprehensive and Updated Review of its Phytochemical and Pharmacological Properties
Abstract
Areca catechu L., the primary source of areca alkaloids, has long been considered the only species in the genus containing these compounds. However, a recent study detected the same pyridine alkaloids in A. triandra, suggesting that alkaloid biosynthesis may also occur in other species of Areca. The immature fruit or pericarp of A. catechu is the principal component of betel quid, a masticatory deeply rooted in ancient oriental culture and consumed by over 600 million people globally. Its longstanding medicinal applications have attracted extensive modern research into its chemical composition, biological activities, and toxicity. This review provides a comprehensive update on the major chemical constituents of A. catechu, including alkaloids, flavonoids, tannins, other phenolic compounds, triterpenoids and phytosterols, fatty acids, glycerides, and polysaccharides, and their reported pharmacological activities, such as antioxidant, anti-inflammatory, analgesic and antinociceptive, cytotoxic, antibacterial and antifungal, hypoglycemic, lipid-regulating, antiparasitic, immunomodulatory, neuroprotective, digestive, antiviral, cardiovascular, anti-allergic, and other effects. Information was systematically collected from PubMed, Google Scholar, ACS, Web of Science, and ScienceDirect to provide an up-to-date summary of its phytochemistry and bioactivity.
Keywords:
Areca catechu, Arecaceae, Areca nut, Phytochemicals, Alkaloid, ArecolineIntroduction
The genus Areca comprises approximately 48–54 species, belongs to the subtribe Arecinae (tribe Areceae, subfamily Arecoideae, family Arecaceae), and represents one of the most diverse palm groups in the Asia–Pacific region.1−3 Of these, Areca catechu L., commonly known as the areca nut palm or betel nut palm, is the most well-known species in the genus due to its cultural, economic, and medicinal significance.4 Although many older sources claimed that A. catechu is the only species in the genus containing the main areca alkaloids, a more recent study (Wu et al., 2019) detected these alkaloids in A. triandra, suggesting that alkaloid biosynthesis in the genus Areca may not be confined to A. catechu.2
A. catechu is locally known as “Binglang” in China, “Supari” in India, “Cau” in Vietnam, and “Pinang” in Indonesia, reflecting its widespread cultural and economic significance across tropical Asia, where it is widely cultivated in South and Southeast Asia, including India, Bangladesh, Sri Lanka, Myanmar, Thailand, Vietnam, Indonesia, Philippines, Pakistan, and New Guinea.3−5. A. catechu has a long history of use in traditional medicine across Asia, with its seeds and pericarp widely incorporated into classical herbal systems. According to the Chinese Pharmacopoeia, more than 100 traditional drug formulations contain areca nut (Arecae Semen) for the treatment of parasitic infections, liver disorders, dyspepsia, abdominal distension, pain, diarrhea, edema, and jaundice.2,4,6,7 In traditional Chinese medical literature, such as the Compendium of Materia Medica, areca nut is further described as a remedy for dysentery, abdominal bloating, and constipation.6,7 In the Ayurvedic system of India, A. catechu seeds are used as a deworming agent, digestive stimulant, astringent, and emmenagogue, and are widely incorporated into folk medicinal preparations.3,6,8 In Southeast Asia, traditional uses vary by region: in Cambodia, areca nut is used to treat diarrhea, dysentery, and liver disorders, whereas in Malaysia, it is used for lumbago and abdominal distension.6 The immature fruit or pericarp of A. catechu is the principal component of betel quid, a masticatory deeply rooted in ancient Oriental culture, typically chewed together with betel leaf (Piper betel) and slaked lime.5,6,9 According to the World Health Organization (WHO), more than 600 million people worldwide consume areca nut, a practice deeply embedded in the ceremonies, rituals, and cultural customs of betel-quid-chewing communities.2,5,10
The traditional medicinal applications of A. catechu have stimulated extensive research into its chemical composition and biological activities. The purpose of this review is to provide a comprehensive and up-to-date synthesis of the major phytochemical constituents and pharmacological properties of A. catechu. Available research demonstrates that the species contains a broad array of compounds, including alkaloids, flavonoids, tannins, phenolic derivatives, triterpenoids, phytosterols, fatty acids, glycerides, and polysaccharides, that collectively contribute to its wide spectrum of biological activities. These include antioxidants, anti-inflammatory, analgesic, antinociceptive, cytotoxic, antimicrobial, hypoglycemic, lipid-modulating, antiparasitic, immunomodulatory, neuroprotective, digestive, antiviral, cardiovascular, and anti-allergic effects. Consolidating these findings provides a crucial foundation for understanding the pharmacological potential of A. catechu and informing future research directions.
Phytochemistry of A. catechu
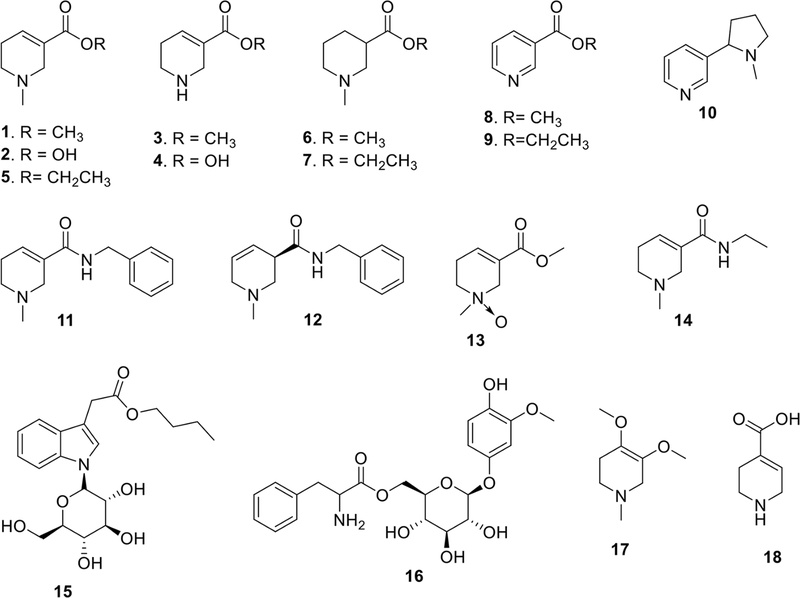
Alkaloids – A. catechu contains a characteristic group of alkaloids that are structurally related to pyridine and tetrahydropyridine derivatives. The total alkaloid content of areca nut has been reported as 0.3–0.7% of dry weight.5,11,12 To date, a total of 18 alkaloids have been reported from A. catechu including arecoline (1),13,14 arecaidine (2),13,14 guavacoline (3),13 guavacine (4),13,14 ethyl N-methyl-l,2,5,6-tetrahydro-pyridine-3-carboxylate (homoarecoline, 5),13,14 methyl N-methylpiperidine-3-carboxylate (6),15 ethyl N-methylpiperidine-3-carboxylate (7),15 methyl nicotinate (8),4,15 ethyl nicotinate (9),4,15 nicotine (10),15 arecatemines A–C (11–13),14 N-ethyl-1-,2,5,6-tetrahydro-1-methyl-3-pyridinecarboxamide (14),14 acatechus A and B (15 and 16),4 arecolidine (17),16 and isoguvacine (18)16,17 (Fig. 1). Among them, four compounds—arecoline (1), arecaidine (2), guvacoline (3), and guvacine (4)—are consistently recognized as the principal alkaloids and have been repeatedly confirmed through both historical isolation studies and modern analytical approaches such as LC-MS and GC-MS.5,18 Notably, arecoline (1) is generally regarded as the predominant alkaloid in A. catechu, both in abundance and toxicological significance.5 Quantitative analyses of fresh seeds indicate concentration ranges of 0.30–0.63% for arecoline (1), 0.31–0.66% for arecaidine (2), 0.03–0.06% for guvacoline (3), and 0.19–0.72% for guvacine (4).5,12
In addition to the major alkaloids, several studies have mentioned putative minor constituents such as arecolidine (17) and isoguvacine (18). Yet, evidence for their genuine occurrence in A. catechu is weak, as primary reports with full spectroscopic characterization are lacking. One secondary source suggested that arecolidine (17) was first isolated in 1915, but no re-isolation or structural confirmation was achieved for decades.19 In 1993, Dehmlow et al. attempted both re-isolation from areca nuts and synthesis, but failed in both cases. They concluded that the originally proposed structure of arecolidine (17) was likely incorrect and that the compound was too unstable to be isolated.19 Recently, Wang et al. reported the detection of isoguvacine (18) in areca nut seed extracts using HPLC-ESI-QTOF/MS.17 Most recently, five new alkaloids have been isolated from A. catechu: arecatemines A–C from the nuts14 and acatechus A and B from the dried fruits.4
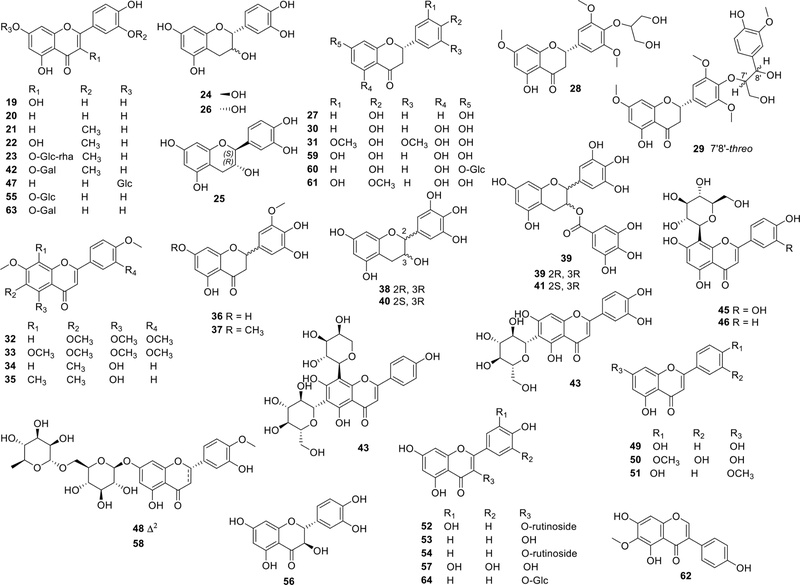
Flavonoids – Flavonoids are one of the major classes of phenolic compounds in A. catechu, occurring predominantly in the seeds (nuts), with lower concentrations in other plant parts such as the pericarp and leaves.5 The flavonoid profile of A. catechu includes several monomeric and glycosidic forms. Notable flavonoids isolated from in A. catechu include: quercetin (19),4,17,20,21 luteolin (20),4,20 chrysoeriol (21),20,22 isorhamnetin (22),4,20,21 isorhamnetin 3-O-(6"-O-α-L-rhamnopyranosyl)-β-D-glucopyranoside (23),4 (+)-catechin (24),4,21 (−)-catechin (25),4 epicatechin (26),23 liquiritigenin (27),4,21 calquiquelignans M and N (28–29),7 naringenin (30),7, 24 dihydrotricin (31),7 sinesetin (32),7 nobiletin (33),7 8-demethyleucalyptin (34),7 eucalyptin (35),7 5,7,4'-trihydroxy-3',5'-dimethoxyflavanone (36),21 (±)-4',5-dihydroxy-3',5',7-trimethoxyflavanone (37)20 epigallocatechin (38),17,24 epigallocatechin gallate (39) 17,25
In addition, recent UHPLC-MS/MS and HPLC-ESI-QTOF/MS profiling have revealed the presence of numerous additional flavonoids in different parts of areca nut (flower, husk, and seed), including (−)-gallocatechin (40),17,24 (−)-gallocatechin gallate (41),17 isorhamnetin-3-O-galactoside (42),17,24 schaftoside (43),24 isoorientin (44),24 orientin (45),24 vitexin (46),24 cymaroside (47),24 diosmin (48),24 apigenin (49),24 diosmetin (50),24 genkwanin (51),24 rutin (52),17,24 kaempferol (53),24 kaempferol-3-O-rutinoside (54),24 kaempferol-3-O-glucoside (64),24 isoquercitrin (55),24 taxifolin (56),24 myricetin (57),24 hesperidin (58),24 eriodictyol (59),24 naringenin-7-O-glucoside (60),17 hesperetin (61),24 tectorigenin (62),24 quercetin-3-O-galactoside (63),17 and kaempferol-3-O-glucoside(64).17
Tannins – The tannin fraction in A. catechu is predominantly composed of condensed tannins, which are not easily hydrolyzed under normal conditions. Although hydrolysable tannins are also present in smaller amounts,26 the condensed forms dominate, especially in unripe nuts.5,12 These condensed tannins comprise both proanthocyanidins and A- or B-type procyanidins (oligomers of flavan-3-ols, mainly catechin and epicatechin).5,12,27 The arecatannins isolated from betel nuts consist of (−)-epicatechin units in the upper units and (+)-catechin units in the terminal unit, with C4-C8 linkages in most cases, or C4-C6 in some terminal catechin units.27 Such structural arrangements render them resistant to hydrolysis under normal conditions, although they can undergo oxidative dehydration under acidic or alkaline conditions to form insoluble reddish-brown polymers. This reaction explains the red juice observed during betel quid (BQ) chewing, resulting from interactions between tannins and slaked lime,28 and also contributes to the astringent taste of areca nut.
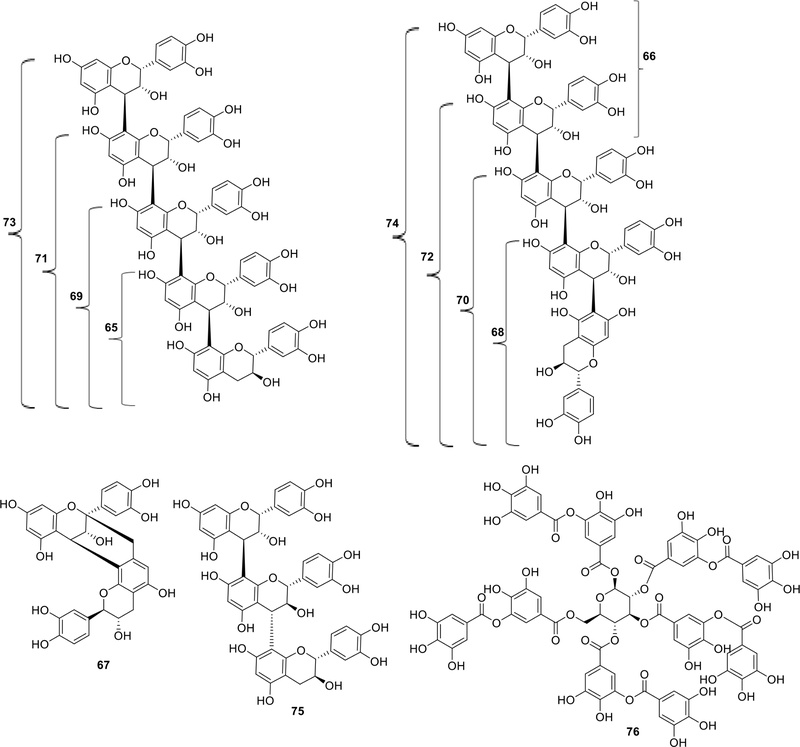
To date, 11 oligomeric tannins have been isolated and structurally characterized from A. catechu,27 including procyanidin B1 (65),4 procyanidin B2 (66), procyanidin A1 (67), procyanidin B7 (68), arecatannins A1–A3 (69, 71, and 73), arecatannins B1–B3 (70, 72, and 74), arecatannin C1 (75). In the original study by Ma et al. (1996),27 the pentameric A-type procyanidin isolated from A. catechu nuts was designated as arecatannin A2 (C75H62O30.5H2O), with the structure determined as epicatechin (4β→8)-epicatechin-(4β→8)-epicatechin-(4β→8)-epicatechin-(4β→8)-catechin. Subsequent literature and chemical databases have standardized the nomenclature according to degree of polymerization, under which this compound is now commonly referred to as arecatannin A3 (73).5 To avoid ambiguity, we follow this modern convention in the present review. In this system, arecatannins A1–A3 (69, 71, and 73) correspond to trimeric, tetrameric, and pentameric A-type procyanidins, respectively. Similarly, the corresponding B-type pentamer, herein designated as arecatannin B3, was described in the same study as the B-type analog of A3.27 We confirm that arecatannin B3 corresponds to the pentameric B-type procyanidin (C75H62O30), establishing for the first time a clear correspondence between its structure, polymer length, and nomenclature. In addition to clarifying the correspondence between structure, polymer length, and nomenclature, this review also presents, for the first time, an updated overview of procyanidin B7 (68) and arecatannin B3 (74) from A. catechu as reported in the original isolation study by Ma et al..27 Recently, the presence of tannic acid (76) (a hydrolysable tannin) in A. catechu nuts has been confirmed through chemical analysis, using near-infrared spectroscopy (NIRS) combined with high-performance liquid chromatography (HPLC).29
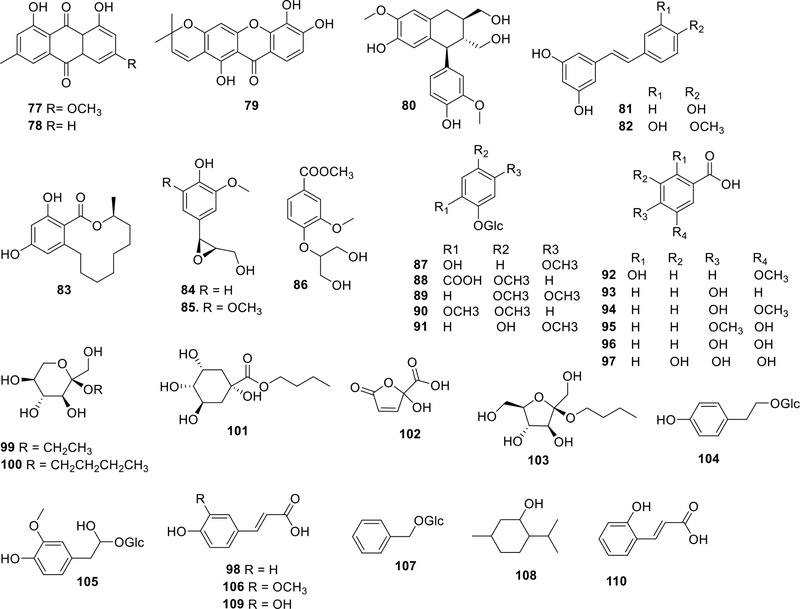
Other phenolic constituents – In addition to the flavonoids and tannins described above, a variety of other phenolic compounds have been reported from A. catechu, including emodin-type anthraquinones [physcion (77) and chrysophanol (78)],4,20 the xanthone jacareubin (79),20 the lignan (+)-isolariciresinol (80),7 stilbenes [resveratrol (81)4 and rhapontigenin (82)7]. Several other phenolic and related minor metabolites have also been identified either by isolation or LC-MS analysis, including de-O-methyllasiodiplodin (83),21 epoxyconiferyl alcohol (84),22 4-[3'-(hydroxymethyl)oxiran-2'-yl]-2,6-dimethoxyphenol (85),22 glyceryl-2-vanillic acid methyl ester (86),7 2-hydroxy-5-methoxyphenyl-β-D-glucopyranoside (87),4 2-(β-D-glucopyranosyloxy)-5-methoxy-benzoic acid (88),4 3,4-dimethoxyphenyl-β-D-glucopyranoside (89),4 2,4-dimethoxyphenyl-β-D-glucopyranoside (90),4 4-hydroxy-3-methoxyphenyl-β-D-glucopyranoside (91),4 2-hydroxy-5-methoxy-benzoic acid (92),4 4-hydroxybenzoic acid (93),4,22 4-hydroxy-3-methoxy-benzoic acid (vanillic acid, 94),4,21 3-hydroxy-4-methoxy-benzoic acid (isovanilic acid, 95),4 3,4-dihydroxy-benzoic acid (protocatechuic acid, 96),4 gallic acid (97),17,29 p-coumaric acid (98),17,30 β-D-fructopyranoside, ethyl (99),4 β-D-fructopyranoside, butyl (100),4 butyl quinate (101),4 2,5-dihydro-2-hydroxy-5-oxo-2-furancarboxylic acid (102),4 β-D-fructofuranoside, butyl (103),4 2-(4-hydroxy-phenyl) ethl-β-D-glucopyranoside (104),4 stroside B (105),4 ferulic acid (106),4,17 β-D-glucopyranoside, benzyl (107),4 5-methyl-2-(1-methylethyl)-cyclohexanol (108), caffeic acid (109),17 and o-coumaric acid (110).17
Triterpenoids and phytosterols – Several triterpenoids and steroidal compounds have been identified in A. catechu seeds through phytochemical studies. Reported constituents include β-sitosterol (80),4,31 stigmastane-3β,5,6α-triol (81),4 arborinol (82),4 sitosterol-3-O-β-glucopyranoside (83),4 and 6,20-di-O-β-D-glucopyranosyl-20(S)-protopanaxatriol (ginsenoside Rg1) (84),4 arborinol methyl ether (85),32 5,8-epidioxiergosta-6,22-dien-3β-ol (89),21 stigmasta-4-en-3-one (90),21 cycloartenol (88),21 and stigmasterol (91).31
Fatty acids and glycerides – Several studies have investigated the fatty acid composition of A. catechu (betel nut) using gas chromatographic analysis. Based on these studies, the major fatty acids consistently identified in the seed oil include lauric (92), myristic (93), pentadecanoic (94), palmitic (95), stearic (96), oleic (97), and linoleic (98) acids.30,31
Detailed fractionation of A. catechu seed fat demonstrated that the lipid fraction is mainly composed of mixed triglycerides.33 The principal glycerides were identified as fully saturated glycerides (56%; including trimyristin, dimyristolaurin, dimyristopalmitin, and lauromyristopalmitin), mono-unsaturated-di-saturated glycerides (30%; mainly hexadecenolauromyristin, hexadecenodimyristin, oleodimyristin, oleomyristopalmitin, and linoleomyristopalmitin), and di-unsaturated–mono-saturated glycerides (13.7%; mainly oleolinoleolaurin, oleolinoleomyristin, and oleolinoleopalmitin).33
Polysaccharides – In addition, A. catechu contains appreciable amounts of polysaccharides with complex sugar compositions. Hu et al. optimized the extraction of polysaccharides from A. catechu and found that the polysaccharide fraction (AUPs) consisted of mannose, glucose, galactose, and arabinose in different molar ratios.34 Ji et al. isolated a neutral polysaccharide, PAP1b, using hot-water extraction followed by ethanol precipitation and column chromatography; PAP1b was mainly composed of mannose, galactose, xylose, and arabinose in proportions of approximately 4.1:3.3:0.9:1.7, with a molecular weight of ~37.3 kDa.35 In the most recent study, Zhang et al. characterized A. catechu seed polysaccharides (AKP), which were primarily composed of mannose (≈ 58.7%) and galactose (≈ 33.6%).36
Pharmacological Properties of A. catechu
Antioxidant activity – Extracts of A. catechu have demonstrated notable antioxidant potential in various in vitro assays, with efficacy varying substantially depending on the plant organ, extraction solvent, and developmental stage of the nut, consistent with comparative studies.37-39 A wide range of assays has been employed to evaluate the antioxidant properties of A. catechu extracts, including DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging, ABTS [2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)] radical cation decolorization, ferric-reducing antioxidant power (FRAP), hydroxyl radical scavenging, nitric oxide radical scavenging, superoxide anion scavenging, lipid-peroxidation inhibition (FTC, ferric thiocyanate method), and ferrous ion (Fe²⁺) chelation assays.10,23,24,37,38,40-44 Collectively, these methods consistently demonstrate that A. catechu contains strong radical-quenching components.
In addition, the total phenolic content (TPC) and total flavonoid content (TFC) of A. catechu extracts are commonly quantified to evaluate correlations with their antioxidant potential.10,23,37,41 Most studies report a strong positive correlation between TPC/TFC and antioxidant performance, indicating that areca seeds are an excellent food material with potential antioxidant properties. For example, Hamsar et al. reported that methanolic extracts from different plant parts—particularly unripe seeds—exhibited markedly higher antioxidant activity in the DPPH radical scavenging assay compared with root and adventitious root extracts. The methanol extract of unripe seeds contained the highest total phenolic (186.2 ± 0.04 mg GAE/g) and flavonoid (18.13 ± 0.007 mg QE/g) contents, which correlated with its strong free radical-quenching ability, comparable to that of BHT and vitamin C.37 Rangani et al. examined the relationship between polyphenolic and alkaloid contents and the antioxidant capacity of ripe and unripe A. catechu nuts collected from major cultivation areas in Sri Lanka.10 The study showed that antioxidant activities (DPPH, ABTS, FRAP, NO, and hydroxyl radical scavenging) correlated strongly with total phenolic, flavonoid, and tannin contents, particularly in ripe nuts, but weakly or negatively with total alkaloid content. These results indicate that the antioxidant potential of A. catechu is mainly attributable to its polyphenolic constituents rather than arecoline-type alkaloids, and that both phytochemical composition and antioxidant capacity vary with fruit maturity and geographic origin.10 Similarly, Chavan and Singhal demonstrated that polyphenol-rich fractions obtained after separating phenolics from arecoline (1) exhibited substantially higher antioxidant activity than the alkaloid-rich fraction. The study also identified major phenolic constituents contributing to this activity, further reinforcing the dominant role of polyphenols in the antioxidant profile of A. catechu.45 Fan et al. compared multiple extraction methods for areca husks and seeds and found that ultrasonic-microwave synergistic extraction yielded the highest TPC (270.92 mg GAE/g DW) and purity (42.84%) in the seed extract.41 These extracts exhibited strong antioxidant activity, with IC50 values for DPPH, ABTS, and superoxide anion of 55.40, 81.87, and 138.65 µg/mL, respectively, mainly due to proanthocyanidin polymers. Zhang et al. optimized ethanol extraction of areca seed polyphenols and identified epicatechin and syringic acid as the major compounds. The extracts exhibited significant antioxidant activity in DPPH, hydroxyl radical scavenging, and reducing power assays, showing activity comparable to or exceeding ascorbic acid in certain tests.23
In addition, one study successfully isolated 11 phenolic compounds from A. catechu and assessed their antioxidant activities using the DPPH radical-scavenging assay.44 Most of the isolated compounds exhibited considerable radical-scavenging activity. Notably, (2S,3R)-ent-catechin (25) (SC50 = 19.2 µM), jacareubin (79) (SC50 = 19.7 µM), and luteolin (20) (SC50 = 32.8 µM) showed strong antioxidant activity compared with the positive control, ascorbic acid (SC50 = 28.9 µM). Other compounds, including the newly identified 4-[3'-(hydroxymethyl)oxiran-2'-yl]-2,6-dimethoxyphenol (85), epoxyconiferyl alcohol (84), and protocatechuic acid (96), exhibited moderate activity, whereas certain flavanones exhibited weaker effects. These results highlight that specific phenolic constituents of A. catechu contribute markedly to its overall antioxidant capacity. Recent studies have shown that polysaccharides extracted from A. catechu seeds also exhibit remarkable antioxidant activity, including DPPH, ABTS, and FRAP scavenging, indicating that compounds beyond polyphenols contribute to the plant’s total radical-scavenging potential.34,35
Furthermore, recent cellular studies have confirmed that polyphenol extracts of areca nuts also provide intracellular antioxidant protection. Yi et al. investigated LPS-stimulated RAW 264.7 macrophages, showing that the extracts significantly reduced reactive oxygen species (ROS) levels, upregulated Nrf2 (nuclear factor erythroid 2–related factor 2) and HO-1 (heme oxygenase-1) expression, and modulated MAPK (mitogen-activated protein kinase) signaling pathways.3 The polyphenol fraction was prepared via ethanol extraction followed by XAD-7 resin purification, with catechin (24) and proanthocyanidin B1 (65) as major constituents, highlighting their importance for intracellular antioxidant effects.3 Collectively, these findings demonstrate that the antioxidant potential of A. catechu depends mainly on its polyphenolic and polysaccharide composition, extraction solvent, plant part, and developmental stage, whereas alkaloids play a minimal role in the overall activity.
Anti-inflammatory activity – Several studies have demonstrated the anti-inflammatory potential of A. catechu extracts in both in vitro and in vivo models. The anti-inflammatory effects include inhibition of pro-inflammatory mediators such as nitric oxide (NO) and interleukin-6 (IL-6), accompanied by reduced expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), as well as modulation of NF-κB signaling in LPS-stimulated RAW 264.7 macrophages.42,46–49 In addition, A. catechu and its respective fractions, particularly the aqueous fraction, demonstrated an anti-inflammatory effect on carrageenan-induced edema in mice and rats. These extracts also inhibited edema induced by prostaglandin E2 (PGE2) and arachidonic acid, but had no effect on histamine- or serotonin-induced inflammation.48 More recently, a 70% ethanolic extract of A. catechu nuts exhibited strong anti-inflammatory and cartilage-protective effects in a rat model of monosodium iodoacetate-induced osteoarthritis, with comparable efficacy to meloxicam.50 Furthermore, a study focusing on the procyanidin-rich fraction of A. catechu provided more direct mechanistic evidence. The extract significantly suppressed COX-2 expression and prostaglandin E2 production both in LPS-stimulated cells and in animal models of inflammation, confirming that condensed tannins such as procyanidins contribute substantially to the anti-inflammatory properties of A. catechu.51 Collectively, these findings indicate that polar fractions of A. catechu, rich in alkaloids and polyphenolic compounds, may contribute to its anti-inflammatory activity through the modulation of key inflammatory pathways.
Analgesic and antinociceptive effects – Hydroalcoholic and crude extracts of A. catechu nuts have demonstrated potent analgesic effects in rodent models.42,48 In the hot plate test, oral administration of 250–1000 mg/kg hydroalcoholic extract increased pain latency, with maximal analgesia (54.5%) observed at 1000 mg/kg for 1 h.42 In the formalin-induced pain model, extracts dose-dependently reduced paw licking time; notably, 500 mg/kg inhibited both the early (0–5 min, 39.5%, p < 0.05) and late (20–25 min, 92.7%, p < 0.01) phases, indicating efficacy against neurogenic and inflammatory pain, while 250 and 1000 mg/kg primarily affected the late phase.42 Similarly, crude and aqueous fractions (10–100 mg/kg) of nuts reduced formalin-induced nociception in a dose-dependent manner, with efficacy comparable to aspirin.48
Supporting evidence from non-nut plant parts shows that methanol extracts of stems and leaves also exhibit dose-dependent antinociceptive activity in acetic acid-induced writhing in mice, with leaf extracts demonstrating higher efficacy than stem extracts and comparable to aspirin at low doses.52 These findings collectively indicate that A. catechu nut extracts exert analgesic effects through both central and peripheral mechanisms, while extracts from other plant parts provide supporting evidence of antinociceptive potential, warranting further investigation.
Cytotoxicity – Numerous studies have demonstrated that the nut of A. catechu and its principal alkaloid, arecoline (1), exert cytotoxic effects on diverse normal human cell types, including hepatocytes, splenocytes, myoblasts, lymphocytes, endothelial cells, and epithelial cells.53–62 Early studies linked cytotoxicity of A. catechu nut extracts or fractions to increased reactive oxygen species (ROS), leading to protein, lipid, and DNA damage.57,63,64 In the work by Wang et al., the crude areca nut extract suppressed splenic T-cell activation and reduced interleukin-2 (IL-2) and interferon-γ (IFN-γ) production in murine T-cells, an effect that was attenuated by the antioxidant N-acetyl-l-cysteine (NAC), implicating oxidative stress as a mediator.63 Subsequently, highly oligomeric procyanidins from the nut induced lymphocyte apoptosis via depletion of intracellular thiols, also linked to oxidative stress.57,64 showed that the 30–100 kDa fraction of areca nut extract triggered autophagy in both normal and malignant cells, which was partially reversed by ROS scavengers. The ethanol extract of A. catechu seeds showed higher cytotoxicity against human squamous carcinoma cells (HSC-3, IC50 = 164.06 µg/mL) than HSC-2 cells (IC50 = 629.50 µg/mL), while no cytotoxic effect was observed on human keratinocyte (HaCat) cells.65 Epidemiological studies in Taiwan have linked betel-quid chewing, which includes A. catechu, with increased risk of oral squamous cell carcinoma (OSCC).66
Numerous studies have detailed the cytotoxic effects of arecoline (1) across multiple epithelial cell types. Tsai et al. demonstrated that arecoline (1) induces DNA double-strand breaks, evidenced by γ-H2AX phosphorylation (phosphorylated histone H2AX), and activates the ATM/Chk1/Chk2 DNA damage response pathway (ATM: ataxia-telangiectasia mutated; Chk1/Chk2: checkpoint kinases 1 and 2), leading to G2/M cell cycle arrest and suppression of p53-dependent DNA repair.59 These effects were observed in several epithelial cell lines, including KB (human oral epidermoid carcinoma), HEp-2 (human laryngeal epithelial carcinoma), and 293 (HEK-293, human embryonic kidney epithelial) cells. Chang et al. similarly observed that arecoline (1) activates Chk1/Chk2, induces cell cycle arrest, and modulates matrix metalloproteinase-9 (MMP-9) and tissue inhibitors of metalloproteinases (TIMPs) expression in SAS (human tongue squamous cell carcinoma) oral epithelial cells, suggesting additional involvement in extracellular matrix remodeling and potential relevance to tissue fibrosis and malignant transformation.60
In endothelial cells, Tseng et al. reported that arecoline (1) at concentrations ≥ 0.4 mM induces cell retraction, detachment, apoptosis in the sub-G0/G1 phase, and G2/M cell cycle arrest.58 These findings were confirmed by Ullah et al.,55 who observed vacuolation, mitochondrial damage, and necrosis in primary human endothelial cells, providing additional evidence that arecoline (1) exerts cytotoxic effects through organelle injury and necrosis. Chou et al. reported that arecoline (1) causes dose-dependent cytotoxicity in normal rat hepatocytes (Clone-9), inducing G0/G1 cell-cycle arrest, apoptosis, and necrosis via upregulation of TGF-β and p21WAF1, with phosphorylation of p53 (Ser15).67 Yang et al. reported that arecoline (1) had dose-dependent effects on oral fibroblasts and oral cancer cell lines. Low concentrations (< 0.8 μg/mL) increased cell proliferation, whereas higher concentrations (25–400 μg/mL) reduced viability as shown by the MTT (methyl thiazolyl tetrazolium) assay, accompanied by cell-cycle alterations.66 Shih et al. showed that arecoline (1, 50–200 μM) induces dose-dependent neurotoxicity by increasing reactive oxygen species (ROS) through NADPH oxidase 2 (NOX2) activation and suppressing glutathione (GSH) and superoxide dismutase (SOD). These oxidative changes trigger apoptosis, evidenced by increased cytochrome c, Bax, caspase-9/-3, and reduced Bcl-2, which can be attenuated by catalase or NOX inhibitors.68
In epithelial cells, arecoline (1) demonstrated dose- and time-dependent apoptosis via both mitochondrial (intrinsic) and death-receptor (extrinsic) pathways, including cleaved-caspase-3, poly (ADP-ribose) polymerase (PARP) cleavage, and truncated Bid (tBid).61,62 further showed that arecoline (1) induces cell cycle arrest and apoptosis through Akt/mTOR (protein kinase B/mammalian target of rapamycin) inhibition mediated by PHLPP2 (PH domain and leucine-rich repeat protein phosphatase 2) upregulation.54 reported that arecoline (1) induces cytotoxicity in K-562 cells associated with epigenetic dysregulation, while Yan et al. described arecoline-induced neurotoxicity via endoplasmic reticulum (ER) stress and downregulation of the Nrf2/HO-1 (nuclear factor erythroid 2–related factor 2/heme oxygenase-1) pathway.69
In addition to arecoline (1), flavonoids and polyphenolic fractions from A. catechu contribute to cytotoxicity.64 observed ROS-mediated autophagy in the 30–100 kDa fraction of areca nut in both normal and malignant cells, whereas apoptosis in lymphocytes was induced by highly oligomeric procyanidins via thiol depletion Wang et al. reported that highly oligomeric procyanidins induce apoptosis in lymphocytes via thiol depletion.57 From A. catechu seeds, two isolated flavonoids (calquiquelignans M and N, 28–29) showed moderate cytotoxicity against HepG2 liver cancer cells, with IC50 values of 49.8 μM and 53.6 μM, respectively.7 Collectively, these findings indicate that A. catechu extracts and its bioactive compounds induce oxidative stress, apoptosis, and organelle injury, which underline the cytotoxic effects observed across diverse human cell types.
Antibacterial and antifungal activities – The aqueous extract of A. catechu seeds showed broad-spectrum anti-microbial activity in vitro, inhibiting Gram-negative bacteria at 3.3–7 µg/mL, Gram-positive bacteria at 16 µg/mL, and Candida albicans at 16 µg/mL, while also suppressing aß toxin production by Aspergillus flavus.70 Fan et al. demonstrated that phenolic-rich seed extracts obtained via ultrasonic-microwave extraction showed potent inhibition against Streptococcus mutans, Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa, with MIC values ranging from 2.5 to 25 mg/mL, which was attributed to the presence of proanthocyanidin polymers.41 Similarly, Jam et al. evaluated aqueous, ethanolic, and methanolic extracts of areca nut fruit against several pathogenic bacteria (S. aureus, E. coli, S. enterica, and E. aerogenes).71 The methanolic and ethanolic extracts displayed strong inhibitory effects in both agar diffusion and broth microdilution assays, while the aqueous extract was less active. The methanolic extract exhibited the highest activity (MIC of 1.56 mg/mL against E. coli) and reduced microbial contamination in cake samples (100 mg/L) during storage, supporting its potential as a natural food preservative.71
In an in vivo experiment, Sari et al. reported that areca nut extract significantly reduced Staphylococcus aureus infection and enhanced immune function in rats, suggesting both direct antibacterial and immunomodulatory roles of its phenolic constituents.72 Earlier, Jiang et al. demonstrated the antimalarial potential of A. catechu extract in an in vivo murine model, providing early evidence of its anti-infective capacity.73 Subsequently, methanolic and chloroform extracts were shown to possess notable antibacterial and antimalarial activities.74 Moreover, a polyphenolic fraction obtained through ethanol extraction exhibited pronounced antitubercular activity against Mycobacterium tuberculosis, while the crude methanolic extract prepared via microwave-assisted extraction also showed moderate antibacterial activity against both S. aureus (Gram-positive) and Escherichia coli (Gram-negative).25
Several isolated constituents from A. catechu have demonstrated remarkable antimicrobial activities. Among the phenolic compounds isolated from the fruit,20 jacareubin (79) showed potent antibacterial activity against Staphylococcus aureus and methicillin-resistant S. aureus (MRSA), suggesting that phenolic derivatives contribute to the plant’s antibacterial potential. In addition, compounds derived from the pericarp—namely fernenol (117), arundoin (118), and a mixture of stigmasterol (122) and β-sitosterol (111)—exhibited pronounced antifungal effects.31 These triterpenoids markedly inhibited both mycelial growth and spore germination of Colletotrichum gloeosporioides and were more effective than benomyl in postharvest mango tests. Collectively, these findings indicate that structurally diverse metabolites of A. catechu—including polyphenols, triterpenoids, and sterols—possess broad-spectrum antimicrobial activity, supporting their potential as natural antibacterial, antifungal, and antimalarial agents.
Hypoglycemic effects – Extracts and isolated constituents of A. catechu have been reported to possess notable hypoglycemic potential through multiple mechanisms.75–78 Among different plant parts, the nut, leaf, and flower extracts all exhibit glucose-lowering or enzyme-inhibitory activity in experimental models. The ethanolic extract of the seeds of A. catechu exhibited in vitro inhibition of intestinal α-glucosidases (IC50 = 12 µg/mL for maltase, 30 µg/mL for sucrase) and significant antihyperglycemic activity in maltose-loaded rats at 250 and 500 mg/kg.76 Hypoglycemic effects have also been reported for leaf and flower extracts of A. catechu. The methanolic leaf extract reduced fasting blood glucose in streptozotocin-induced diabetic rats,77 while both ethanol and aqueous flower extracts at 500 mg/kg/d orally for 21 days decreased glucose, triglyceride, total cholesterol, and urea levels in alloxan-induced diabetic rats.78 Furthermore, areca nut procyanidins have been shown to ameliorate hyperglycemia in streptozotocin (STZ)-induced diabetic mice, in part by suppressing hepatic gluconeogenesis through the down-regulation of phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6-phosphatase (G6Pase) expression.79
Recent studies have highlighted the potent hypoglycemic potential of A. catechu seed extracts. The hydroalcoholic seed extract showed significant antihyperglycemic effects in alloxan-induced diabetic rats, both as a monotherapy (1000 mg/kg p.o.) and in combination with gliclazide (25 mg/kg p.o.), with improvements observed in oral glucose tolerance tests and histological evidence of pancreatic β-cell proliferation and organ protection (Al-Omran et al., 2024).80 In addition, deep eutectic solvent (DES) extracts from waste seeds exhibited strong in vitro α-glucosidase inhibitory activity (IC50 = 0.0066 mg/mL), surpassing acarbose (IC50 = 0.012 mg/mL), further highlighting the seeds as a promising source of α-glucosidase inhibitors.81
Regulating effects on blood lipids – Various extracts of A. catechu have been reported to modulate blood lipid profiles in experimental models. The ethanol extract of areca nut (ETAN, 0.5% w/w in diet) significantly reduced plasma cholesterol by approximately 25% in rats fed a high-fat diet, accompanied by decreased activities of small intestinal pancreatic cholesterol esterase (pCEase) and acyl coenzyme A-cholesterol acyltransferase (ACAT).82 Similarly, the water extract of areca nut (WEAN, 0.5% w/w, p.o.) reduced triglyceride absorption and plasma lipid levels in rats.83,84
Antiparasitic effects – Extracts and bioactive constituents of A. catechu exhibit broad antiparasitic activity in both in vitro and in vivo studies. The methanol extract of A. catechu nuts and its butanol subfraction showed antimalarial effects against Plasmodium falciparum using the SYBR-green assay, with the butanol fraction being most potent (IC₅₀ = 18 µg/mL).74 The aqueous seed extracts demonstrated significant anthelmintic activity against Ascaridia galli in poultry. Treatment with 11–22 mg/mL extract induced paralysis and death of adult worms in vitro, while oral administration (26 mg/kg) in infected chickens reduced worm burden and improved intestinal histopathology.85
Additionally, the aqueous extracts of A. catechu nuts, alone or combined with Chinese honeysuckle (Quisqualis indica L.) seeds, exhibited notable anthelmintic activity against Ascaridia spp. and liver flukes, as well as antibacterial effects against Escherichia coli in vitro, with A. catechu showing faster worm paralysis and broader bacterial inhibition.86 More recently, ethanolic seed extracts displayed potent in vitro anthelmintic activity against Ascaridia galli in poultry, causing worm paralysis and death comparable to albendazole.87 These studies confirm the traditional use of areca nut as an anthelmintic and provide experimental support for its antiparasitic potential.
Immunomodulatory effects – Crude extracts of A. catechu have shown potential immunomodulatory activity in both in vitro and in vivo studies. In vivo, oral administration of areca nut extract to Sprague-Dawley rats enhanced immune responses against Staphylococcus aureus infection, reflected by increased white blood cell (WBC) counts and enhanced macrophage activity, without detectable hepatotoxicity or nephrotoxicity.72 Complementarily, in vitro studies on human polymorphonuclear leukocytes (PMNs) demonstrated that the A. catechu extracts reduce intracellular reactive oxygen species and myeloperoxidase release, suggesting a modulatory effect on inflammatory responses.88 Conversely, isolated arecoline (1), the primary alkaloid of A. catechu, has been reported to induce immunosuppression, hepatotoxicity, and depletion of antioxidant defenses in mice, highlighting the potential for adverse effects at high doses or with chronic exposure.53 These findings underscore the importance of distinguishing between the effects of whole plant extracts and purified alkaloids, with crude extracts exhibiting modulatory and protective properties, while isolated arecoline (1) may exert toxic effects on immune function.
Effects on the nervous system – Extracts and bioactive constituents of A. catechu exhibit multifaceted neuropharmacological actions in preclinical models, including antidepressant-like activity, cholinergic and GABAergic neuromodulation, putative cognitive effects related to cholinergic modulation, and neurotoxicity under certain exposure conditions.
Antidepressant-like activity – Extracts and bioactive constituents of A. catechu have demonstrated antidepressant-like effects in preclinical models. Early studies showed that ethanol, methanol, and hexane extracts inhibit monoamine oxidase (MAO) activity in rat brain homogenates, providing a biochemical basis for mood-regulating effects.89 The ethanolic extract of areca nut reduced immobility time in the Forced Swim Test (FST) and Tail Suspension Test (TST) in rats, with effects comparable to fluoxetine.90 Ethanol and aqueous fractions of areca nut also increased hippocampal serotonin and noradrenaline levels by ~30–35%, reducing depressive-like behaviors.91 More recently, chronic stress models demonstrated that A. catechu extracts ameliorate depression-like behaviors, improving immobility and sucrose preference in rats.92 Additionally, the alkaloid arecoline (1) alleviated depression-like behaviors in adult mice, accompanied by modulation of cortical synaptic transmission in the anterior cingulate cortex (ACC).93 In silico analyses further suggest that areca compounds, including arecoline (1), guvacoline (3), and homoarecoline (5), may interact with antidepressant targets, supporting their potential as adjuncts to conventional therapies.94 Dichloromethane extracts (DHEAN) also show antidepressant-like activity and mitigate morphine withdrawal symptoms.95,96
Cholinergic and GABAergic neuromodulatory effects – The major alkaloid arecoline (1) contributes to neuromodulatory effects primarily via cholinergic mechanisms, including muscarinic receptor stimulation and catecholamine release.97–100 Acetylcholinesterase inhibitory activity has been demonstrated for A. catechu extracts and fractions.99 In addition, arecaidine (2) and guvacine (3), also present in the extracts, act as GABA receptor antagonists, with IC50 values of 122 ± 12 µM and 8 ± 1 µM, respectively,97,98 thereby modulating inhibitory (GABAergic) neurotransmission.
Putative cognitive effects related to cholinergic modulation – Although related to cholinergic modulation rather than direct excitatory neurotransmission, arecoline’s cholinergic stimulation may indirectly influence cognitive processes, supporting putative cognitive effects; however, direct in vivo evidence for neuroprotection or memory enhancement remains limited.99,100
Neurotoxicity – At higher doses or during chronic exposure, A. catechu constituents, particularly arecoline (1), have been reported to induce neurotoxicity through oxidative stress, suppression of antioxidant defenses, and modulation of signaling pathways.62,68
Effects on the digestive system – The extract of A. catechu (ANE) dose-dependently increased contractions of colonic muscle strips in rats, and the effect was partially inhibited by atropine, indicating involvement of muscarinic (M) receptors.101 Raw and charred Semen Arecae enhanced gastrointestinal motility in chronically stressed rats, accompanied by increased motilin and substance P levels, while cholecystokinin (CCK) mRNA expression was down-regulated.102 More recently, Wang et al. showed that areca nut (AN) and charred areca nut (CAN) improve intestinal motility in loperamide-induced constipation models through modulation of gut microbiota and metabolic profiles.103 In the latest study,104 Ji et al. reported that areca nut polysaccharides, after in vitro digestion and fecal fermentation, modulate gut microbiota and increase the production of short-chain fatty acids (SCFAs), which may contribute to enhanced digestive function. These findings indicate that A. catechu exerts pro-motility effects through both direct stimulation of smooth muscle and microbiota-mediated mechanisms.
Antiviral activity – The aqueous extract of A. catechu seeds exhibited potent inhibition of HIV-1 protease (human immunodeficiency virus type 1), reducing enzymatic activity by more than 70% at a concentration of 0.2 mg/mL.105 Further isolation identified arecatannins, oligomers of flavan-3-ols, as active constituents: arecatannin B1 (70) displayed strong inhibition (78% at 0.5 mM), while arecatannin A1 (69) showed moderate activity (56.8% at 0.5 mM). In contrast, monomeric flavan-3-ols, including (+)-catechin (24) and (−)-epicatechin (25), exhibited little to no inhibition, except for (−)-epicatechin gallate (41, 44.4% at 0.5 mM), indicating that oligomeric procyanidins are likely the principal compounds responsible for the observed anti-HIV-1 protease activity.
The aqueous extract of A. catechu inhibited the replication of Newcastle Disease Virus (NDV) and Egg Drop Syndrome Virus (EDS) in embryo cultures (Nelson Anthikat & Michael, n.d.), indicating potential antiviral properties.70 Lee et al. reported that a methanolic extract of Areca catechu inhibited syncytium formation in baby hamster kidney (BHK) cells infected with Newcastle disease virus (NDV), likely by blocking trafficking of hemagglutinin-neuraminidase (HN) glycoprotein, a viral surface protein important for cell binding and viral release, with an IC50 for α-glucosidase of around 10 µg/mL.106
More recently, Zamanian et al. reported that the extract significantly inhibited herpes simplex virus type 1 (HSV-1) infection in Vero cells, with an IC50 of 110.52 ± 1.36 µg/mL and no cytotoxicity up to 400 µg/mL. The extract also reduced expression of HSV-1 genes UL46 (2 h pre-infection) and US6 (gD) (simultaneous infection and 4 h post-infection), suggesting interference with viral attachment and/or replication.107 These findings support A. catechu as a potential source of natural antiviral agents.
Cardiovascular effects – Several studies have investigated the cardiovascular effects of A. catechu and its constituents. A condensed-tannin–rich fraction from A. catechu seeds exhibited antihypertensive activity, producing a dose-dependent reduction in arterial blood pressure in hypertensive rats.108 In a subsequent study, a seed extract induced vasodilation in isolated rat aortic rings, an effect that required an intact endothelium, indicating involvement of endothelial mechanisms in vascular relaxation.109 Arecoline (1), the major alkaloid of A. catechu, has been shown to exert detrimental cardiac effects. In Sprague–Dawley rats, arecoline (1) induced myocardial injury via apoptosis mediated by the Fas/Fas-ligand pathway, with severity increasing in a dose-dependent manner.110 In vitro and in vivo studies further identified a novel heptapeptide derived from defatted kernel protein hydrolysates that exhibited angiotensin-converting enzyme (ACE)-inhibitory activity and significantly lowered blood pressure in spontaneously hypertensive rats.111 More recently, arecoline (1) was reported to induce cardiotoxicity and pathological myocardial hypertrophy through activation of multiple hypertrophy-related signaling pathways—including interleukin-6 (IL-6)/MEK5/ERK5, Janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3), mitogen-activated protein kinase (MAPK), and calcineurin - in Sprague–Dawley rats.112
Anti-allergic effects – In vitro, water extract of Semen Arecae (WEAN) inhibited degranulation of RBL-2H3 mast cells induced by DNP-BSA and compound 48/80, with IC50 values of 53 and 52 µg/mL, respectively, and in vivo, oral administration of WEAN at 300 mg/kg reduced compound 48/80-induced systemic anaphylaxis in mice by 46%.113 Polyphenol-enriched extracts from areca nuts, administered in drinking water (0.05% and 0.1%, w/w), decreased ovalbumin-induced allergic responses in sensitized mice, including diarrhea, mast cell infiltration and degranulation in the duodenum, and serum OVA-specific IgE and IL-4 levels.114 These findings provide direct evidence that A. catechu extracts can suppress allergic responses mediated by mast cells and IgE.
Other pharmacological effects – In addition, A. catechu extracts exhibit several additional bioactivities. For hepatoprotection, aqueous seed extracts reduced oxidative liver damage in CCl4-treated rodents, acting through antioxidant and anti-inflammatory pathways.38 The ethanolic nut extract (ETAN) showed aphrodisiac effects, enhancing sexual behavior in male rats by increasing mounting frequency and intromission latency, while also boosting reproductive organ weights and sperm counts.115 The potential skin-lightening activity of the ethanolic extract of A. catechu nut (ETAN) was revealed via its tyrosinase inhibition in B16 melanoma cells (IC50 = 0.48 mg/mL).116
In addition, the ethanolic extract of A. catechu nut promoted burn wound healing in normal and dexamethasonesuppressed rats by accelerating epithelialization and enhancing wound contraction.117
In conclusion, by systematically compiling and critically analyzing information from diverse sources (PubMed, Google Scholar, ACS, Web of Science, and ScienceDirect), this review highlights over 130 phytochemical constituents of A. catechu, along with their biological activities and those of its extracts. This comprehensive overview integrates the most accurate and recent findings, serving as a reliable reference for future research and potential therapeutic applications.
Acknowledgments
This research was supported by the National Institute of Food and Drug Safety Evaluation (23212NIFDS204) and Gyeongsangbuk-do RISE (Regional Innovation System & Education) project (2025-RISE-15-107).
Conflict of Interest
The authors declare no conflict of interest.
References
-
Loo, A. H.; Dransfield, J.; Chase, M. W.; Baker, W. J. Mol. Phylogenet. Evol. 2006, 39, 598–618.
[https://doi.org/10.1016/j.ympev.2005.12.006]

-
Wu, J.; Zhang, H.; Wang, S.; Yuan L.; Grünhofer P.; Schreiber L.; Wan, Y. J. Plant Res. 2019, 132, 531–540.
[https://doi.org/10.1007/s10265-019-01115-9]

-
Yi, S.; Zou, L.; Li, Z.; Sakao, K.; Wang, Y.; Hou, D.-X. Foods 2022, 11, 3607.
[https://doi.org/10.3390/foods11223607]

-
Cao, M.; Yuan, H.; Daniyal, M.; Yu, H.; Xie, Q.; Liu, Y.; Li, B.; Jian, Y.; Peng, C.; Tan D.; Peng, Y.; Choudhary, M. I.; Rahman, A.-U.; Wang, W. Fitoterapia 2019, 138, 104276.
[https://doi.org/10.1016/j.fitote.2019.104276]

-
Sun, Y.; Feng, J.; Hou, W.; Qi, H.; Liu Y. Front. Pharmacol. 2024, 15, 1407212.
[https://doi.org/10.3389/fphar.2024.1407212]

-
Aswany, T.; Helen P. A. M.; Ijinu T. P.; Pongillyathundiyil, S.; Ramachandran, R.; George, V.; Pushpangadan, P. Ann. Phytomed. 2023, 12, 490–502.
[https://doi.org/10.54085/ap.2023.12.2.86]

-
Yuan, M.; Ao, Y.; Yao, N.; Xie, J.; Zhang, D.; Zhang, J.; Zhang X.; Ye, W. Molecules 2019, 24, 2862.
[https://doi.org/10.3390/molecules24162862]

-
Kirtikar, K. R.; Basu B. D. Indian medicinal plants: Sudhindra Nath Basu, Panini Office; Bahadurganj, India, 1918; pp 2547–2548.
[https://doi.org/10.5962/bhl.title.137025]

-
Gupta, P.; Warnakulasuriya, S. Addict. Biol. 2002, 7, 77–83.
[https://doi.org/10.1080/13556210020091437]

-
Rangani, S. C.; Marapana, R. A. U. J.; Senanayake, G. S. A.; Perera, P. R. D.; Pathmalal, M. M.; Amarasinghe, H. K. Appl. Food Res. 2023, 3, 100361.
[https://doi.org/10.1016/j.afres.2023.100361]

-
Liu, Y.-J.; Peng W.; Hu M.-B.; Xu, M.; Wu, C.-J. Pharm. Biol. 2016, 54, 2753–2760.
[https://doi.org/10.3109/13880209.2016.1160251]

-
Guo, Z.; Wang, Z.; Luo, Y.; Ma, L.; Hu, X.; Chen, F.; Li, D.; Jia, M. Food Frontiers 2024, 5, 1909–1932.
[https://doi.org/10.1002/fft2.443]

-
Huang, J. L.; McLeish, M. J. J. Chromatogr. A 1989, 475, 447–450.
[https://doi.org/10.1016/S0021-9673(01)89702-8]

-
Tang, S.-N.; Zhang, J.; Liu, D.; Liu, Z.-W.; Zhang X.-Q.; Ye W.-C. J. Asian Nat. Prod. Res. 2017, 19, 1155–1159.
[https://doi.org/10.1080/10286020.2017.1307187]

-
Holdsworth, D. K.; Jones, R.; Self, R. Phytochemistry 1998, 48, 581–582.
[https://doi.org/10.1016/S0031-9422(98)00016-8]

-
Tang, W.; Eisenbrand, G. In Chinese Drugs of Plant Origin: Chemistry, Pharmacology, and Use in Traditional and Modern Medicine; Tang, W.; Eisenbrand, G. Ed; Springer; Germany, 1992, pp 139–143.
[https://doi.org/10.1007/978-3-642-73739-8_20]

-
Wang, R.; Pan, F.; He, R.; Kuang, F.; Wang, L.; Lin, X. J. Appl. Res. Med. Aromat. Plants. 2021, 25, 100336.
[https://doi.org/10.1016/j.jarmap.2021.100336]

-
Jain, V.; Garg, A.; Parascandola, M.; Chaturvedi, P.; Khariwala, S. S.; Stepanov I. J. Agric. Food Chem. 2017, 65, 1977–1983.
[https://doi.org/10.1021/acs.jafc.6b05140]

-
Dehmlow, E. V.; Westerheide R. Heterocycles 1994, 37, 355–366.
[https://doi.org/10.3987/com-93-s14]

- Zhang Xing, Z. X.; Mei WenLi M. W.; Zeng YanBo Z. Y.; Liu Jian L. J.; Dai WenJun D. W., Dai HaoFu D. H. J. Trop. Subtrop. Bot. 2009, 17, 74–76.
- Yang, W.-Q.; Wang, H.-C.; Wang, W.-J.; Wang, Y.; Zhang, X.-Q.; Ye, W.-C. Zhong Yao Cai 2012, 35, 400–403.
- Xing, Z.; Jiao, W.; Zhuang, H.; Wen-li, M., Hao-Fu, D. Chem. Res. Chin. Univ. 2010, 26, 161–164.
-
Zhang, W.-M.; Huang, W.-Y.; Chen, W.-X.; Han, L.; Zhang, H.-D. Molecules 2014, 19, 16416–16427.
[https://doi.org/10.3390/molecules191016416]

-
Song, F.; Tang, M.; Wang, H.; Zhang, Y.; Zhu, K.; Chen, X.; Chen, H.; Zhao, X. Ind. Crops Prod. 2022, 189, 115787.
[https://doi.org/10.1016/j.indcrop.2022.115787]

-
Raju, A.; De Subrata, S.; Ray, M. K.; Degani M. S. Int. J. Mycobacteriol. 2021, 10, 13–18.
[https://doi.org/10.4103/ijmy.ijmy_199_20]

-
Wang, C.-K.; Lee W.-H. J. Agric. Food Chem. 1996, 44, 2014–2019.
[https://doi.org/10.1021/jf950611o]

-
Ma, Y. T.; Hsu, F. L.; Lan, S. J. J.; Chen, C. F. J. Chin. Chem. Soc. 1996, 43, 77–81.
[https://doi.org/10.1002/jccs.199600013]

-
Joo, Y. J.; Newcombe, D.; Nosa, V.; Bullen C. Subst. Use Misuse. 2020, 55, 1422–1442.
[https://doi.org/10.1080/10826084.2019.1666144]

-
Dai, J.; Tang, W.; Zhang, J.; Kang, X.; Dai, W.; Ji, J.; Wang S. Microchem. J. 2024, 196, 109586.
[https://doi.org/10.1016/j.microc.2023.109586]

-
Xiao, Y.; Yang, Y.; Yong, J.; Lu, C. Biomed. Res. Rev. 2019, 3, 1–4.
[https://doi.org/10.15761/BRR.1000131]

-
Yenjit, P.; Issarakraisila, M.; Intana, W.; Chantrapromma, K. Postharvest Biol. Technol. 2010, 55, 129–132.
[https://doi.org/10.1016/j.postharvbio.2009.09.003]

-
He, X.; Li, Y.; Zhang, C.; Hu, X. Acta. Crystallogr. Sect. E Struct. Rep. Online 2010, 66, o2273–o2274.
[https://doi.org/10.1107/S1600536810030758]

-
Pathak, S.; Mathur S. J. Sci. Food Agric. 1954, 5, 461–465.
[https://doi.org/10.1002/jsfa.2740051003]

-
Hu, M.; Peng, W.; Liu, Y.; Wu, N.; Zhao, C.; Xie, D.; Yan, D.; Zhang, X.; Tao, X.; Wu, C.-J. J. Food Process. Preserv. 2017, 41, e12798.
[https://doi.org/10.1111/jfpp.12798]

-
Ji, X.; Guo, J.; Pan, F.; Kuang, F.; Chen, H.; Guo, X.; Liu, Y. Front. Nutr. 2022, 9, 853115.
[https://doi.org/10.3389/fnut.2022.853115]

-
Zhang, J.; Feng, X.; Dai, J.; Wang, S.; Kang, X.; Dai, W.; Ji, J. Tropical Plants 2024, 4, e020.
[https://doi.org/10.48130/tp-0024-0049]

-
Hamsar, M. N.; Ismail S.; Mordi M. N.; Ramanathan S.; Mansor S. M. Free Radicals Antioxidants 2011, 1, 28–33.
[https://doi.org/10.5530/ax.2011.1.6]

-
Pithayanukul, P.; Nithitanakool S.; Bavovada R. Molecules 2009, 14, 4987–5000.
[https://doi.org/10.3390/molecules14124987]

- Wetwitayaklung, P.; Phaechamud T.; Limmatvapirat C., Keokitichai S. Naresuan Uni. J. 2006, 14, 1–14.
-
Zhang, W.; Li, B.; Han, L.; Zhang H. Afr. J. Biotechnol. 2009, 8, 3887–3892.
[https://doi.org/10.1016/j.transproceed.2009.06.205]

-
Fan, X.; Jiang, C.; Dai, W.; Jing, H.; Du, X.; Peng, M.; Zhang, Y.; Mo, L.; Wang, L.; Chen, X. J. Food Meas. Charact. 2022, 16, 1502–1515.
[https://doi.org/10.1007/s11694-021-01244-7]

-
Bhandare, A. M.; Kshirsagar, A. D.; Vyawahare, N. S.; Hadambar, A. A.; Thorve, V. S. Food Chem. Toxicol. 2010, 48, 3412–3417.
[https://doi.org/10.1016/j.fct.2010.09.013]

-
Ahn, B.-Y. J. Korean Soc. Appl. Biol. Chem. 2009, 52, 92-95.
[https://doi.org/10.3839/jksabc.2009.016]

- Xing, Z.; Jiao, W.; Zhuang, H.; Wen-li, M.; Hao-fu, D. Chem. Res. Chinese Universities 2010, 26, 161–164.
-
Chavan, Y. V.; Singhal R. S. J. Sci. Food Agric. 2013, 93, 2580–2589.
[https://doi.org/10.1002/jsfa.6081]

-
Lee, K. P.; Sudjarwo, G. W.; Kim, J.-S.; Dirgantara, S.; Maeng, W. J.; Hong, H. Nutr. Res. Pract. 2014, 8, 267–271.
[https://doi.org/10.4162/nrp.2014.8.3.267]

- Sharaf, S.; Chinchu, J. U.; Rahitha Devi, S. J.; Prakash Kumar, B. Indian J. Biochem. Biophys. 2021, 58, 464–471.
-
Khan, S.; Mehmood, M. H.; Ali, A. N. A.; Ahmed, F. S.; Dar, A.; Gilani, A.-H. J. Ethnopharmacol. 2011, 135, 654–661.
[https://doi.org/10.1016/j.jep.2011.03.064]

-
Bhandare, A.; Kshirsagar, A.; Vyawahare, N.; Sharma, P.; Mohite R. J. Ethnopharmacol. 2011, 136, 267–270.
[https://doi.org/10.1016/j.jep.2011.04.039]

-
Humaryanto, H.; Rahman A. O.; Quzwain F. Biomol. Health Sci. J. 2023, 6, 48–53.
[https://doi.org/10.4103/bhsj.bhsj_34_22]

-
Huang, P.-L.; Chi, C.-W.; Liu T.-Y. Food Chem. Toxicol. 2010, 48, 306–313.
[https://doi.org/10.1016/j.fct.2009.10.014]

- Barman, M. R.; Uddin, S.; Akhter, S.; Ahmed, N.; Haque, Z.; Rahman, S.; Mostafa, F.; Zaman, M.; Noor, F. A., Rahmatullah, M. Adv. Nat. Appl. Sci. 2011, 5, 223–226.
-
Dasgupta, R.; Saha, I.; Pal, S.; Bhattacharyya, A.; Sa, G.; Nag, T. C.; Das, T.; Maiti, B. R. Toxicology 2006, 227, 94–104.
[https://doi.org/10.1016/j.tox.2006.07.016]

-
Lin, P.-C.; Chang, W.-H.; Chen, Y.-H.; Lee, C.-C.; Lin Y.-H., Chang, J.-G. J. Toxicol. Environ. Health A 2011, 74, 737–745.
[https://doi.org/10.1080/15287394.2011.539123]

-
Ullah, M.; Cox, S.; Kelly, E.; Boadle, R.; Zoellner, H. J. Oral Pathol. Med. 2014, 43, 761–769.
[https://doi.org/10.1111/jop.12186]

-
Chiang, S.-L.; Jiang, S.-S.; Wang, Y.-J.; Chiang, H.-C.; Chen, P.-H.; Tu, H.-P.; Ho, K.-Y.; Tsai, Y.-S.; Chang, I.-S.; Ko Y.-C. Toxicol. Sci. 2007, 100, 66–74.
[https://doi.org/10.1093/toxsci/kfm201]

-
Wang, C.-C.; Huang P.-L.; Liu, T.-Y.; Jan, T.-R. Toxicol. In Vitro 2009, 23, 1234–1241.
[https://doi.org/10.1016/j.tiv.2009.07.033]

-
Tseng, S.-K.; Chang, M.-C.; Su, C.-Y.; Chi, L.-Y.; Chang, J. Z.-C.; Tseng, W.-Y.; Yeung, S.-Y.; Hsu, M.-L.; Jeng, J.-H. Clin. Oral Investig. 2012, 16, 1267–1273.
[https://doi.org/10.1007/s00784-011-0604-1]

-
Tsai, Y.-S.; Lee, K.-W.; Huang, J.-L.; Liu, Y.-S.; Juo, S.-H. H.; Kuo, W.-R.; Chang, J.-G.; Lin, C.-S., Jong, Y.-J. Toxicology 2008, 249, 230–237.
[https://doi.org/10.1016/j.tox.2008.05.007]

-
Chang, M.-C.; Chan, C.-P.; Wang, W.-T.; Chang, B.-E.; Lee, J.-J.; Tseng, S.-K.; Yeung S.-Y.; Hahn, L.-J.; Jeng, J.-H. Head Neck 2013, 35, 1295–1302.
[https://doi.org/10.1002/hed.23119]

-
Li, M.; Gao, F.; Zhou, Z.-S.; Zhang, H.-M.; Zhang, R.; Wu, Y.-F.; Bai, M.-H.; Li, J.-J.; Lin, S.-R.; Peng, J.-Y. Oncol. Rep. 2014, 31, 2422–2428.
[https://doi.org/10.3892/or.2014.3091]

-
Gu, L.; Xie, C.; Peng, Q.; Zhang, J.; Li, J.; Tang, Z. Toxicology 2019, 419, 32–39.
[https://doi.org/10.1016/j.tox.2019.03.006]

-
Wang, C.; Liu, T.; Wey, S.; Wang, F.; Jan, T. Food Chem. Toxicol. 2007, 45, 1410–1418.
[https://doi.org/10.1016/j.fct.2007.02.005]

-
Lin, M.-H.; Hsieh, W.-F.; Chiang, W.-F.; Hong, W.-Z.; Hsu, Y.-R.; Cheng, Y.-C.; Chen, T.-C.; Hsu K.-C.; Lina, P.-Y.; Liu S.-Y. Oral Oncol. 2010, 46, 822–828.
[https://doi.org/10.1016/j.oraloncology.2010.08.002]

-
Sari, L. M.; Subita, G. P.; Auerkari. E. I. Asian J. Pharm. Clin. Res. 2017, 10, 286–291.
[https://doi.org/10.22159/ajpcr.2017.v10i10.20287]

- Yang, Y.-Y.; Koh, L.-W.; Tsai, J.-H.; Tsai, C.-H.; Wong, E. F.-C.; Lin, S.-J.; Yang, C.-C. Jpn. J. Clin. Oncol. 2004, 34, 176–183.
-
Chou, W.-W.; Guh, J.-Y.; Tsai, J.-F.; Hwang, C.-C.; Chen, H.-C.; Huang, J.-S.; Yang, Y.-L.; Hung, W.-C., Chuang, L.-Y. Toxicology 2008, 243, 1–10.
[https://doi.org/10.1016/j.tox.2007.09.003]

-
Shih, Y.-T.; Chen, P. S.; Wu, C.-H.; Tseng, Y.-T.; Wu, Y.-C.; Lo, Y.-C. Free Radic. Biol. Med. 2010, 49, 1471–1479.
[https://doi.org/10.1016/j.freeradbiomed.2010.07.017]

-
Yan, H.; Ding, J.; Li, X.; Li, S.; Zhang, D. Environ. Toxicol. 2024, 39, 3410–3424.
[https://doi.org/10.1002/tox.24194]

-
Anthikat, R. R. N., Michael A. J. Young Pharm. 2009, 1, 42.
[https://doi.org/10.4103/0975-1483.51874]

-
Jam, N.; Hajimohammadi, R.; Gharbani, P.; Mehrizad, A. Biomed. Res. Int. 2021, 2021, 6663399.
[https://doi.org/10.1155/2021/6663399]

-
Sari, L. M.; Hakim, R. F.; Mubarak, Z.; Andriyanto, A. Vet. World 2020, 13, 134–140.
[https://doi.org/10.14202/vetworld.2020.134-140]

- Jiang, J.-H.; Jung S.-Y.; Kim Y. C.; Shin S. R.; Yu S. T., Park H. Korean J. Oriental Physiol. Pathol. 2009, 23, 494–498.
-
Boniface, P.; Verma S. K.; Cheema H. S.; Darokar M. P.; Pal A. Int. J. Infect. Dis. 2014, 21, 228–229.
[https://doi.org/10.1016/j.ijid.2014.03.897]

- Chempakam, B. Indian J. Exp. Biol. 1993, 31, 474–475.
- Amudhan, M., Begum V. Pharmacogn. Mag. 2008, 4, 223.
- Mondal, S.; Bhattacharya, S.; Biswas, M. J. Adv. Pharm. Educ. Res. 2012, 2, 10–17.
-
Ghate, R.; Patil, V. P.; Hugar, S.; Matha, N. H.; Kalyane, N. V. Asian Pac. J. Trop. Dis. 2014, 4, S148–S152.
[https://doi.org/10.1016/S2222-1808(14)60430-8]

-
Huang, P.-L.; Chi, C.-W., Liu, T.-Y. Food Chem. Toxicol. 2013, 55, 137–143.
[https://doi.org/10.1016/j.fct.2012.12.057]

-
Al-Omran, S. A.; S D.; KV A. K., Nagaraja S. J. Pharmacol. Pharmacother. 2024, 15, 69–77.
[https://doi.org/10.1177/0976500X241231365]

-
Liu, J.; Ma, L.; Deng, S.; Chen, X.; Li, Q.; Xu, A.; Tong, T.; Tan, S.; Wang, M.; Cai, J. Wang, H. Foods 2024, 13, 1070.
[https://doi.org/10.3390/foods13071070]

-
Park, Y. B.; Jeon, S.-M.; Byun, S.-J.; Kim, H.-S.; Choi, M.-S. Life Sci. 2002, 70, 1849–1859.
[https://doi.org/10.1016/S0024-3205(02)01485-6]

-
Byun, S. J.; Kim H. S.; Jeon S. M.; Park Y. B.; Choi M. S. Ann. Nutr. Metab. 2001, 45, 279–284.
[https://doi.org/10.1159/000046739]

-
Jeon, S. M.; Kim H. S.; Lee T. G.; Ryu S. H.; Suh P. G.; Byun S. J.; Park Y. B.; Choi M. S. Ann. Nutr. Metab. 2000, 44, 170–176.
[https://doi.org/10.1159/000012841]

-
Mubarokah, W. W.; Nurcahyo, W.; Prastowo, J.; Kurniasih K. Vet. World 2019, 12, 877–882.
[https://doi.org/10.14202/vetworld.2019.877-882]

- Hai, N.; Atsushi, M.; Nguyen, H. T. T.; Nguyen, T. V. Asian J. Pharm. Clin. Res. 2019, 12, 88–92.
-
Sungpradit, S.; Leesombun, A.; Chanakarn, C.; Nakthong, C.; Boonmasawai, S. BMC Vet. Res. 2025, 21, 491.
[https://doi.org/10.1186/s12917-025-04951-1]

-
Lai, Y.-L.; Lin J.-C.; Yang S.-F.; Liu T.-Y.; Hung, S.-L. J. Periodontal Res. 2007, 42, 69-76.
[https://doi.org/10.1111/j.1600-0765.2006.00917.x]

-
Dar, A.; Khatoon S., Rahman G. Phytomedicine 1997, 4, 41–45.
[https://doi.org/10.1016/S0944-7113(97)80026-8]

-
Bende, M. M.; Dudhgaonkar, S.; Jagdhani, R. S.; Bachewar, N. P. Int. J. Basic Clin. Pharmacol. 2016, 5, 2098–2103.
[https://doi.org/10.18203/2319-2003.ijbcp20163243]

-
Abbas, G.; Naqvi, S.; Erum, S.; Ahmed, S.; Rahman, A.-U.; Dar, A. Phytother. Res. 2013, 27, 39–45.
[https://doi.org/10.1002/ptr.4674]

-
Yao, C.; Zhang, Y.; Sun, X.; Pei, H.; Wei, S.; Wang, M.; Chang, Q.; Liu, X.; Jiang, N. Biomed. Pharmacother. 2023, 164, 114459.
[https://doi.org/10.1016/j.biopha.2023.114459]

-
Chen, Q.-Y.; Zhang, Y.; Ma, Y.; Zhuo, M. Mol. Brain. 2024, 17, 39.
[https://doi.org/10.1186/s13041-024-01106-5]

-
Novianty, R.; Wardana R. D.; Ningsih U. P.; Rabb, R. S. J. Photon 2023, 13, 70–77.
[https://doi.org/10.37859/jp.v13i2.5041]

-
Dar, A.; Khatoon, S. Pharmacol. Biochem. Behav. 2000, 65, 1–6.
[https://doi.org/10.1016/S0091-3057(99)00179-3]

-
Kumarnsit, E.; Keawpradub, N.; Vongvatcharanon, U.; Sawangjaroen, K.; Govitrapong, P. Fitoterapia 2005, 76, 534–539.
[https://doi.org/10.1016/j.fitote.2005.04.015]

-
Lodge, D.; Johnston, G.; Curtis, D.; Brand S. Brain Res. 1977, 136, 513–522.
[https://doi.org/10.1016/0006-8993(77)90075-0]

-
Johnston, G.; Krogsgaard-Larsen, P.; Stephanson, A. Nature 1975, 258, 627–628.
[https://doi.org/10.1038/258627a0]

-
Lim, D.-Y., Kim I.-S. Acta Pharmacol. Sin. 2006, 27, 71–79.
[https://doi.org/10.1111/j.1745-7254.2006.00233.x]

-
Gilani, A. H.; Ghayur, M. N.; Saify, Z. S.; Ahmed, S. P.; Choudhary, M. I.; Khalid A. Life Sci. 2004, 75, 2377–2389.
[https://doi.org/10.1016/j.lfs.2004.03.035]

-
Xie, D.-P.; Li, W.; Qu, S.-Y.; Zheng, T.-Z.; Yang, Y.-L.; Ding, Y.-H.; Wei, Y.-L., Chen, L.-B. World J. Gastroenterol. 2002, 8, 350.
[https://doi.org/10.3748/wjg.v8.i2.350]

- Zhang, S.; Yang, P.; Li, X.; Wang, X.; Song, J.; Peng, W.; Wu, C. Evid. Based Complement. Alternat. Med. 2017, 2017, 1273561.
-
Wang, L.-S.; Wu, J.-S.; Zhang, F.; Huang, Y.; Jiang, Y.-X.; Li Y.-H. Front. Nutr. 2024, 11, 1455824.
[https://doi.org/10.3389/fnut.2024.1455824]

-
Ji, X.; Jiang, K.; Liu, Y.; Zhao, C.; Du, J.; Chen, L.; Zhu, Z.; Li, X. Foods 2025, 14, 2954.
[https://doi.org/10.3390/foods14172954]

-
Kusumoto, I. T.; Nakabayashi, T.; Kida, H.; Miyashiro, H.; Hattori, M.; Namba, T.; Shimotohno, K. Phytother. Res. 1995, 9, 180–184.
[https://doi.org/10.1002/ptr.2650090305]

-
Lee, D.; Boo K. H.; Kim Y. C.; Lee J.-M.; Kang S.; Lee W. S.; Riu K. Z. Korean J. Food. Sci. Technol. 2014, 46, 245–248.
[https://doi.org/10.9721/KJFST.2014.46.2.245]

- Zamanian, M.; Asgari, E.; Afaridoon, S.; Akbarzadeh, T.; Sharifi, Z.; Saeedi, M. Iran J. Microbiol. 2024, 16, 251.
-
Inokuchi, J.; Okabe, H.; Yamauchi, T.; Nagamatsu, A.; Nonaka G.; Nishioka I. Life Sci. 1986, 38, 1375–1382.
[https://doi.org/10.1016/0024-3205(86)90470-4]

-
Goto, H.; Tanaka, N.; Tanigawa, K.; Shimada, Y.; Itoh, T.; Terasawa K. Phytother. Res. 1997, 11, 457–459.
[https://doi.org/10.1002/(SICI)1099-1573(199709)11:6<457::AID-PTR123>3.0.CO;2-J]

-
Lin, W.-Y.; Tsai, B. C.-K.; Day, C. H.; Chiu, P.-L.; Chen, R.-J.; Chen, M. Y.-C.; Padma, V. V.; Luk, H.-N.; Lee H.-C.; Huang C.-Y. Environ. Toxicol. 2021, 36, 1567–1575.
[https://doi.org/10.1002/tox.23153]

-
Liu, X.; Li, G.; Wang, H.; Qin, N.; Guo, L.; Wang, X.; Shen, S. Molecules 2021, 26, 3308.
[https://doi.org/10.3390/molecules26113308]

-
Ho, T.-J.; Tsai, B. C.-K.; Kuo, C.-H.; Luk, H.-N.; Day, C. H.; Hsieh, D. J.-Y.; Chen, R.-J.; Kuo, W.-W.; Kumar, V. B.; Yao, C.-H. Chem. Biol. Interact. 2022, 354, 109810.
[https://doi.org/10.1016/j.cbi.2022.109810]

-
Lee, J. H.; Chang, S. H.; Park, Y. S.; Her, E.; Lee, H. Y.; Park, J. W.; Han, J. W.; Kim, Y. M.; Choi, W. S. J. Pharm. Pharmacol. 2004, 56, 927–933.
[https://doi.org/10.1211/0022357023808]

-
Wang, C.-C.; Lin, Y.-R.; Liao, M.-H.; Jan, T.-R. BMC Complement. Altern. Med. 2013, 13, 154.
[https://doi.org/10.1186/1472-6882-13-154]

- Anthikat, R. R. N.; Micheal A.; Ignacimuthu S. J. Mens Health. 2013, 10, 65–70.
-
Lee, K.-K.; Choi, J.-D. Int. J. Cosmet. Sci. 1999, 21, 275–284.
[https://doi.org/10.1046/j.1467-2494.1999.196590.x]

- Verma, D. K.; Bharat, M.; Nayak, D.; Shanbhag, T.; Shanbhag, V.; Rajput, R. S. Int. J. Pharmacol. Clin. Sci. 2012, 1, 74–78.