Oolonghomobisflavan B Ameliorates Scopolamine-Induced Cognitive Impairment via BDNF-CREB Signaling and Antioxidant Mechanisms in Mice
Abstract
Cognitive impairment, including memory deficits, is a major concern in neurological disorders. Natural polyphenolic compounds have emerged as promising neuroprotective agents due to their antioxidant and anti-inflammatory properties. Previously, we identified a receptor for oolonghomobisflavan B (OHBFB). OHBFB, a polyphenol derived from oolong tea, has shown strong radical-scavenging activity, but its effects on cognitive function and underlying mechanisms remain largely unknown. In this study, mice were orally administered OHBFB (10, 30, 50 mg/kg) for 15 days, followed by scopolamine-induced memory impairment. Behavioral assessments were performed using the novel object recognition (NOR) test and the closed-arm T-maze test. Molecular analyses of brain tissues included Western blotting for brain-derived neurotrophic factor (BDNF) and phosphorylated cAMP response element-binding protein (p-CREB). Safety assessments (body weight and organ weights) and antioxidant capacity (DPPH assay, IC50 = 4.40 μM and ABTS assay, IC50 = 4.08 μM) were also conducted. NOR: novel-object exploration rose from 49.7% (control) to 51.5%, 54.2%, and 59.9% with OHBFB 10, 30, 50 mg/kg. T-maze: performance shifted from 50.5% (control) to 49.6%, 55.1%, and 61.5% at the same doses. Expression of BDNF and p-CREB in brain tissue was restored to near-normal levels in OHBFB-treated groups. OHBFB also exhibited potent antioxidant activity, consistent with its neuroprotective effects, while showing no adverse effects on body weight or organ indices. These findings suggest that, within a scopolamine-induced amnesia mouse model, OHBFB improves cognitive performance while engaging the BDNF-CREB pathway. Given its safety profile, OHBFB may serve as a promising candidate for the treatment of cognitive impairments, including those associated with neurodegenerative disorders.
Keywords:
Oolonghomobisflavan B, Memory, BDNF, Antioxidant, Neuroprotection, ScopolamineIntroduction
Cognitive impairment and memory decline represent key features of various neurological disorders and age-related neurodegeneration. Epidemiological data indicate that cognitive dysfunction is highly prevalent, underscoring the urgent need for novel therapeutic strategies.1,2 Current pharmacological approaches, including cholinesterase inhibitors such as donepezil or galantamine, provide only modest benefits and are often associated with side effects, highlighting the demand for safer and more effective alternatives.3
Natural products, particularly polyphenolic compounds, have received increasing attention as potential neuroprotective agents.4,5 Dietary polyphenols, such as epigallocatechin gallate from green tea and resveratrol from grapes, have been shown to attenuate neuroinflammation, enhance antioxidant defenses, and promote neuronal survival (6–8). Oolong tea, an intermediate-fermented tea, is especially rich in unique flavonoids that may exert beneficial effects on brain health.9,10 However, few studies have investigated the bioactive compounds specific to oolong tea beyond the well-characterized catechins.11,12
Oolonghomobisflavan B (OHBFB) is a dimeric flavan isolated from oolong tea, structurally characterized by multiple hydroxyl substitutions and polyphenolic linkages. Previously, we identified a receptor for OHBFB13. Nevertheless, its neuroprotective potential and underlying mechanisms in the context of cognitive impairment have not been systematically studied.14–17
In the present study, we evaluated whether OHBFB ameliorates scopolamine-induced cognitive impairment and probed associated brain-derived neurotrophic factor (BDNF)-cAMP/calcium response element binding protein (CREB) signaling. Behavioral tests, including the novel object recognition (NOR) test and closed-arm T-maze, were employed to assess memory performance. To explore the underlying mechanisms, we examined the expression of BDNF and p-CREB, two key mediators of synaptic plasticity and learning. We also demonstrated potent radical-scavenging activity of OHBFB in the DPPH assay (IC50 = 4.40 μM), suggesting strong antioxidant capacity. Safety parameters and antioxidant capacity were evaluated to comprehensively assess the therapeutic potential of OHBFB. Oxidative stress is a recognized driver of hippocampal dysfunction, blunting BDNF expression and CREB phosphorylation that are essential for synaptic plasticity and memory consolidation. Accordingly, compounds with radical-scavenging capacity may confer neuroprotection by preserving BDNF-CREB signaling. In this context, we evaluated whether OHBFB’s antioxidant activity aligns with improved cognitive performance and pro-plasticity readouts in a scopolamine model where oxidative stress is elevated.
Experimental
Reagents – Oolonghomobisflavan B (OHBFB) was obtained from Nagara Science Co., Ltd. (Gifu, Japan), The Mouse Phospho-cAMP Response Element Binding Protein ELISA Kit and Mouse Brain-Derived Neurotrophic Factor ELISA Kit were obtained from SunLong Biotech Co., LTD (Zhejiang, China).
Mice and experimental protocols – Female C57BL/6 mice (5 weeks old, 20–21 g) were obtained from ORIENT BIO (Kwangju, Republic of Korea). Mice were housed three per cage with ad libitum access to food and water and maintained under controlled environmental conditions (23°C ± 1°C, 60% ± 5% humidity, 12-hour light/dark cycle). Prior to experimentation, mice were acclimated to the laboratory for one week. Cognitive function was assessed using the novel object recognition and closed-arm T-maze tests to determine the effects of OHBFB. Following acclimation, mice were randomly assigned to one of four groups (n = 12 per group: 1) Normal: distilled water (D.W.) orally and 0.9% NaCl (vehicle) intraperitoneally; 2) Control: scopolamine (SCO) (1 mg/kg, i.p.) and D.W. orally; 3) OHBFB 10: SCO (1 mg/kg, i.p.) and OHBFB (10 mg/kg/day orally); 4) OHBFB 30: SCO (1 mg/kg, i.p.) and OHBFB (30 mg/kg/day orally); and 5) OHBFB 50: SCO (1 mg/kg, i.p.) and OHBFB (50 mg/kg/day orally). SCO (1 mg/kg, i.p.) was administered 30 minutes prior to behavioral testing on day 15 to all groups except the Normal group and Control group. OHBFB was administered orally daily from day 1 to day 21 via gastric gavage (100 μL) at indicated concentrations. After behavior test, animals were anesthetized with isoflurane to provide safe and effective anesthesia throughout the experimental procedures. All animal experiments were conducted in accordance with NIH guidelines and were approved by the Animal Ethics Committee of Daegu Haany University (DHU2025-037).
T-maze test – The closed-arm T-maze test was conducted to assess spatial memory ability. During the maze challenge (experimental day 21 to 22), mice instinctively chose either the left or right path within a T-shaped box. On the training day (experimental day 21), OHBFB administration mouse was placed in the starting area with the right passage blocked and allowed to explore freely for 5 minutes. After 24 hours, on the test day (experimental day 15), the blocking wall was removed, and the mice were allowed to explore both passages (left passage; old route (familiar), right passage; new route (novel)) for 5 minutes. The number of entries into each passage was recorded and calculated as the ratio of new passage entries to total maze entries, multiplied by 100. A higher percentage of new passage entries was interpreted as better spatial memory performance.
Novel object recognition test – The novel object recognition test was conducted to evaluate the mice’s cognitive performance in short-term memory. The experiment took place in a square black box (40 × 40 × 40 cm, with two objects positioned inside). On the training day (experimental day 21), two identical objects were placed in the box, and each mouse was introduced at the center of the box. The female C57BL/6 mice were allowed to freely explore and interact with both objects for 5 minutes, during which the number of touches for each object (familiar object, novel object) was recorded. On the test day, 24 hours later (experimental day 22), the mice were again placed in the center of the box and allowed to explore for the same duration for 5 minutes. However, unlike the training day, one of the two identical objects were replaced with a novel object of a different shape. The number of touches to each object was recorded, and object recognition ability was calculated as the number of touches to the novel object divided by the total number of touches to both objects, multiplied by 100. A higher percentage indicated better cognitive performance and recognition ability.
ELISA assay – BDNF (product NO: QS0107Mo) and p-CREB (product NO: SL0816Mo) protein expression levels were measured using ELISA kits, following the manufacturer’s instructions SunLong Biotech Co., LTD. Absorbance was detected at 450 nm using a microplate reader.
2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical scavenging capacity measurement – Antioxidant activity was evaluated using a DPPH assay. A DPPH radical solution was prepared at a concentration of 120 μM in 95% ethanol. Test compounds (0–7 μM OHBFB) were prepared in 96-well plates, and 10 μL of each compound was added to 195 μL of the ethanolic DPPH solution in the wells. The reaction mixtures were incubated at room temperature for 30 minutes in the dark to prevent light-induced degradation. Absorbance was measured using a microplate reader (517 nm). The free radical scavenging activity (% RSA) was calculated using the following formula:
Where A blank represents the absorbance of the control (DPPH solution without samples), and A sample represents the absorbance of the test sample. Results are expressed as mean values from triplicate analyses.
2,2'-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) decolorization assay – Compounds (0–7 µM OHBFB) were prepared to 96-well plates. Subsequently, ABTS solution was added to each well. The mixtures were shaken continuously for 5 minutes. Absorbance was measured with 734 nm at approximately 27°C using a micro-plate reader. The percentage inhibition of absorbance with 734 nm was calculated relative to the blank (absorbance of the uninhibited radical cation solution) using the following equation:
Where A0 is the blank, and Af is the absorbance measured 5 minutes after the addition of antioxidant samples.
Statistical analysis – Results are presented as averages with standard error of the mean (SEM). Statistical significance was determined using GraphPad Prism 6 software, employing appropriate tests such as unpaired t-tests, and multiple comparison tests (Bonferroni and Tukey’s) as needed.
Results and Discussion
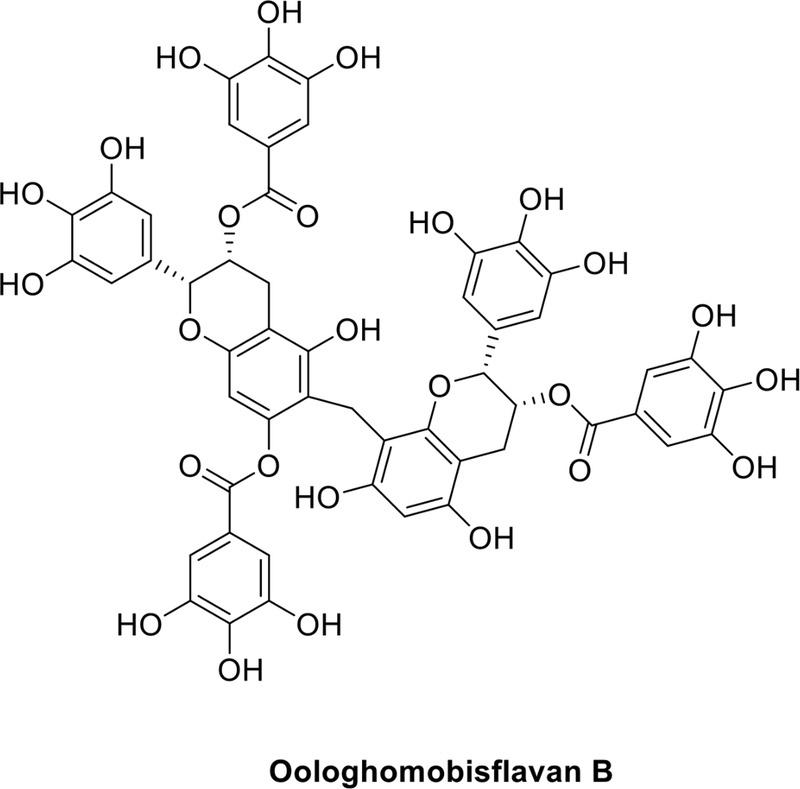
In the present study, we investigated the neuroprotective and cognition-enhancing effects of oolonghomobisflavan B (OHBFB), a polyphenolic compound uniquely identified in oolong tea. The chemical structure of OHBFB was characterized as an epigallocatechin-3-O-gallate dimer, a distinct dimeric flavan derivative (Fig. 1).
To evaluate its cognitive effects, C57BL/6 mice were treated with OHBFB (0, 10, 30, 50 mg/kg, p.o.) for 21 days in a scopolamine (Sco, 1 mg/kg, i.p.)-induced amnesia model. Behavioral performance was assessed using the novel object recognition (NOR) and T-maze tests (Fig. 2). Vehicle-treated normal mice exhibited a strong preference for novel object exploration and intact spatial discrimination, whereas scopolamine treatment significantly impaired both recognition and spatial working memory. Oral administration of OHBFB restored performance in a dose-dependent manner, with 30 and 50 mg/kg showing significant recovery in both NOR and T-maze tasks (Fig. 3A and 3B). In NOR task, in the control group, mice explored the novel object 49.7%; with OHBFB administration this increased to 51.5% at 10 mg/kg, 54.2% at 30 mg/kg, and 59.9% at 50 mg/kg. In T-maze task, in the control group, mice explored the novel object 50.5%; with OHBFB administration this increased to 49.6% at 10 mg/kg, 55.1% at 30 mg/kg, and 61.5% at 50 mg/kg. These results suggest that 30 and 50 mg/kg OHBFB effectively ameliorates Sco-induced cognitive deficits. Collectively, our data support a cognition-improving effect of OHBFB within the scopolamine-induced amnesia model; whether similar benefits occur in healthy mice requires further investigation.
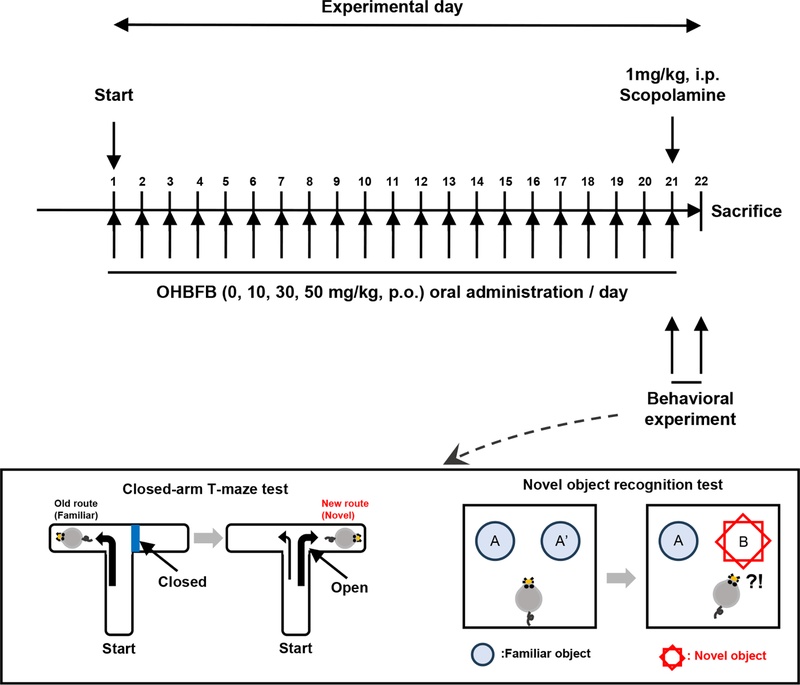
Experimental behavioral testing schedule for the period and diagram. Experimental behavioral testing schedule for the period. The schedule for the period of OHBFB administration and behavioral testing (Novel object recognition test and T-maze test). C57BL/6 mice were treated with oral administration of 0, 10, 30, 50 mg/kg OHBFB for 21 days.
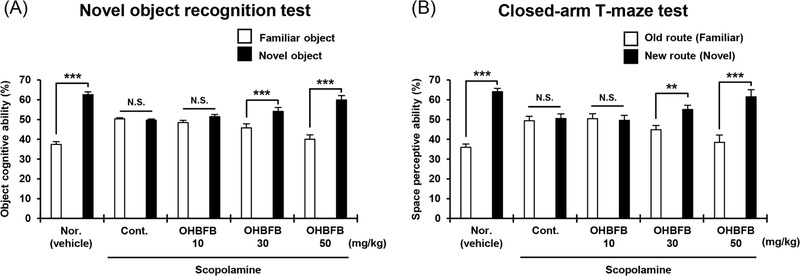
OHBFB improve Scopolamine-induced cognitive impairment. (A) To access Scopolamine-induced cognitive impairment 1 mg/kg, i.p. Scopolamine were administrated before the novel object recognition test. Object cognitive ability was evaluated by novel object recognition test. (B) Space perceptive ability was evaluated by closed arm T-maze test.
To elucidate the underlying molecular mechanisms, hippocampal levels of brain-derived neurotrophic factor (BDNF) and phosphorylated cAMP response element-binding protein (p-CREB) were measured after behavioral testing. Scopolamine markedly reduced both BDNF and p-CREB expression compared with normal controls (Fig. 4A and 4B). Notably, OHBFB administration restored BDNF expression at all tested doses and significantly increased p-CREB levels at 30 and 50 mg/kg. The normalization of BDNF-CREB signaling suggests that OHBFB exerts neuroprotective effects by enhancing pathways critical for synaptic plasticity and memory consolidation.
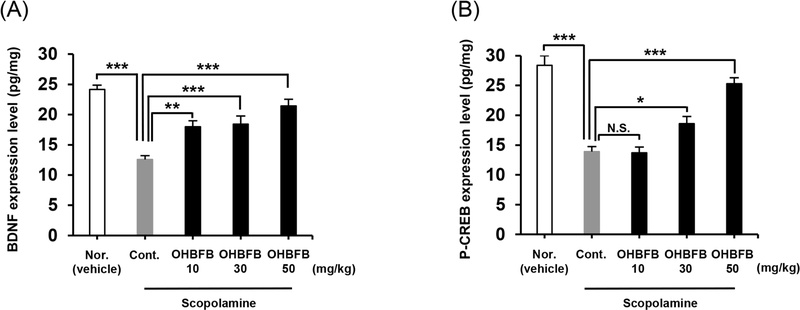
OHBFB exerts its anti-amnesic effect via activation of hippocampal neurotrophins. (A) Hippocampal neurotrophins brain-derived neurotrophic factor (BDNF) and (B) phosphorylated cAMP Response Element Binding protein (p-CREB) protein levels in mouse hippocampus were evaluated using ELISA assay. Data are presented as mean ± SEM (n = 12), Tukey’s test, *p < 0.05, **p < 0.01, ***p < 0.001.
In addition to behavioral and molecular effects, safety assessments were performed. No significant changes were observed in body weight, liver, kidney, spleen weight or brain weight at scopolamine-induced mice after OHBFB treatment (Fig. 5A–E).
Effects of OHBFB on mouse body weight and tissue weights in the SCO-induced cognitive impairment mouse model. Mouse body weight (A) was evaluated during experimental days 21. Liver weight (B), spleen weight (C), kidney weight (D) and brain weight (E) were evaluated at after sacrificed in C57BL/6 mice. Data are presented as mean ± SEM (n = 12), Tukey’s test, *p < 0.05.
OHBFB also demonstrated strong antioxidant capacity, with dose-dependent free radical scavenging activity in both DPPH and ABTS assays (IC50 = 4.40 μM and 4.08 μM, respectively; Fig. 6). Zhang et al. reported an antioxidant effect of OHBFB with a DPPH radical-scavenging IC50 of 4.80 ± 0.30 μM, which is comparable to our result (IC50 ≈ 4.40 μM).9 To the best of our knowledge, the ABTS radical-scavenging potency of OHBFB has not been quantified previously; our study is the first to report an ABTS IC50 of approximately 4.08 μM under the present assay conditions. Notably, the DPPH (4.40 μM) and ABTS (4.08 μM) IC50 values were of similar magnitude, indicating that OHBFB exhibits robust radical-scavenging activity across both alcoholic (lipophilic-leaning) and aqueous (hydrophilic-leaning) conditions. The concordance between assays suggests broad-spectrum antioxidant capacity rather than assay-specific effects. Positive controls behaved as expected, and blank corrections were applied to mitigate chromophore/turbidity interference. Together with the dose-responsive trends, these findings support a consistent antioxidant effect of OHBFB under differing physicochemical environments. These antioxidant properties are likely to contribute, at least in part, to its neuroprotective effects.
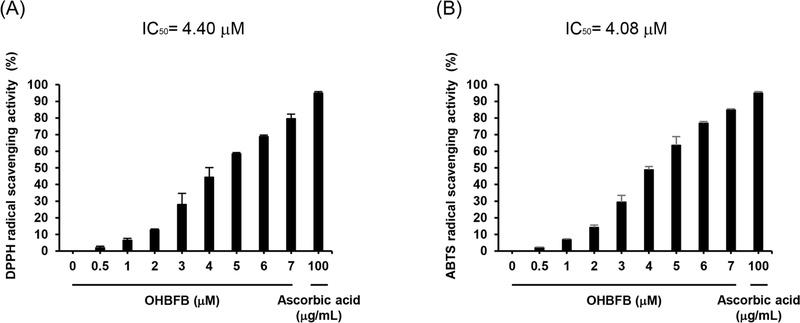
Antioxidant activities of OHBFB. The free radical scavenging activity of OHBFB was evaluated using DPPH and ABTS assays. IC50 values were determined within the concentration range of 0–20 µM. Data are presented as mean ± SEM (n = 6).
Together, our results indicate that OHBFB improves recognition memory and spatial learning, restores hippocampal BDNF-CREB signaling, and exhibits potent antioxidant activity without measurable toxicity. These findings align with previous evidence that natural polyphenols improve cognitive performance through their antioxidant capacity and modulation of neurotrophic signaling.18–20 For example, epigallocatechin gallate (EGCG) from green tea and resveratrol from grapes have been shown to upregulate BDNF levels and enhance synaptic plasticity.21–23 Similarly, our findings demonstrate that OHBFB may share comparable molecular targets, particularly modulation of the BDNF–CREB signaling pathway. OHBFB is a dimeric flavan unique to oolong tea, and its strong scavenging capacity (IC50 = 4.40 μM in DPPH assay) underscores its potential as a distinct antioxidant compound.24,25
The restoration of hippocampal BDNF and p-CREB levels suggests that OHBFB promotes neuronal survival and synaptic plasticity, critical processes disrupted in neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease.26–29 Considering that mild cognitive impairment (MCI) is a prodromal stage of dementia with high risk of progression to Alzheimer’s disease and related neurodegenerative disorders, OHBFB may represent a valuable therapeutic candidate.30,31 Importantly, no adverse effects were observed in terms of body weight, organ indices, or serum liver enzymes, highlighting its favorable safety profile and the potential feasibility of OHBFB as a dietary supplement or therapeutic agent. Moreover, its combined antioxidant and neurotrophic effects suggest broader benefits compared to existing cholinesterase inhibitors, which mainly target cholinergic signaling.32
Despite these encouraging results, several limitations should be noted. First, behavioral assessment was restricted to NOR and T-maze tasks; inclusion of Morris water maze, Y-maze, or passive avoidance tests would strengthen the conclusions.33 Second, molecular analyses were limited to BDNF and p-CREB. Further examination of additional synaptic proteins (e.g., PSD-95, Synapsin I) and antioxidant-related pathways (e.g., Nrf2, HO-1) would provide deeper mechanistic insights.34–36 Third, the bioavailability and pharmacokinetics of OHBFB, particularly its permeability across the blood-brain barrier, remain unknown and merit further investigation.37 Finally, while our work provides substantial preclinical evidence, translational studies and clinical trials are needed to establish its therapeutic potential in humans.38,39
In conclusion, OHBFB alleviates scopolamine-induced cognitive impairment via potent antioxidant activity and activation of the BDNF-CREB pathway, thereby promoting synaptic plasticity and memory consolidation. Importantly, its administration did not induce observable toxicity, underscoring its favorable safety profile.32 Taken together, these findings highlight OHBFB as a promising natural compound for the prevention and treatment of cognitive impairments, including those associated with neurodegenerative disorders.30,31,38 Further studies addressing its pharmacokinetics, blood–brain barrier permeability, and long-term efficacy are warranted to validate the therapeutic application of OHBFB.38–39 Moreover, our behavioral assessment was limited to NOR and T-maze; inclusion of Morris water maze, Y-maze, and passive avoidance, as well as evaluation in additional etiologically relevant models (e.g., Aβ or tau pathology), will be necessary to determine whether OHBFB exerts broader pro-cognitive effects beyond the scopolamine paradigm.
Acknowledgments
This research was supported by the Regional Innovation System & Education (RISE) program through the Gyeongbuk RISE CENTER, funded by the Ministry of Education (MOE) and the Gyeongsangbuk-do, Republic of Korea.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Galvin, J. E. Alzheimer Dis. Assoc. Disord. 2006, 20, 302–310.
[https://doi.org/10.1097/01.wad.0000213858.27731.f8]

-
Hugo, J.; Ganguli, M. Clin. Geriatr. Med. 2014, 30, 421–442.
[https://doi.org/10.1016/j.cger.2014.04.001]

-
Birks, J. S.; Harvey, R. J. Cochrane Database Syst. Rev. 2018, 6, CD001190.
[https://doi.org/10.1002/14651858.CD001190.pub3]

-
Wang, Q.-Q.; Gao, H.; Yuan, R.; Han, S.; Li, X.-X.; Tang, M.; Dong, B.; Li, J.-X.; Zhao, L.-C.; Feng, J.; Yang, S. PLoS One 2020, 15, e0237017.
[https://doi.org/10.1371/journal.pone.0237017]

-
Bae, J.; Kumazoe, M.; Murata, K.; Fujimura, Y.; Tachibana, H. Mol. Nutr. Food Res. 2020, 64, e1900986.
[https://doi.org/10.1002/mnfr.201900986]

-
Shi, J.; Yu, J.; Pohorly, J. E.; Kakuda, Y. J. Med. Food 2003, 6, 291–299.
[https://doi.org/10.1089/109662003772519831]

-
Miranda, M.; Morici, J. F.; Zanoni, M. B.; Bekinschtein, P. Front. Cell. Neurosci. 2019, 13, 363.
[https://doi.org/10.3389/fncel.2019.00363]

-
Huang, E. J.; Reichardt, L. F. Annu. Rev. Neurosci. 2001, 24, 677–736.
[https://doi.org/10.1146/annurev.neuro.24.1.677]

-
Zhang, S.; Duangjan, C.; Tencomnao, T.; Wu, L.; Wink, M.; Lin, J. Front. Aging Neurosci. 2022, 14, 967316.
[https://doi.org/10.3389/fnagi.2022.967316]

-
Ojea Ramos, S.; Feld, M.; Fustiñana, M. S. Front. Mol. Neurosci. 2022, 15, 988790.
[https://doi.org/10.3389/fnmol.2022.988790]

-
Bellavite, P. Antioxidants 2023, 12, 280.
[https://doi.org/10.3390/antiox12020280]

-
Minocha, T.; Birla, H.; Obaid, A. A.; Rai, V.; Sushma, P.; Shivamallu, C.; Moustafa, M.; Al-Emam, A.; Tikhonova, M. A.; Yadav, S. K.; Poeggeler, B.; S ingh, D.; Singh, S . K. Oxid. Med. Cell. Longev. 2022, 2022, 6038996.
[https://doi.org/10.1155/2022/6038996]

-
Bae, J.; Kumazoe, M.; Lee, K.-W.; Fujimura, Y.; Tachibana, H. Phytomedicine 2023, 118, 154970.
[https://doi.org/10.1016/j.phymed.2023.154970]

-
Franzoni, F.; Scarfò, G.; Guidotti, S.; Fusi, J.; Asomov, M.; Pruneti, C. Front. Neurosci. 2021, 15, 729757.
[https://doi.org/10.3389/fnins.2021.729757]

-
Jalouli, M.; Rahman, M. A.; Biswas, P.; Rahman, H.; Harrath, A. H.; Lee, I.-S.; Kang, S.; Choi, J.; Park, M. N.; Kim, L. Front. Pharmacol. 2025, 16, 1492517.
[https://doi.org/10.3389/fphar.2025.1492517]

-
Wu, Q.; Yuan, X.; Bai, J.; Han, R.; Li, Z.; Zhang, H.; Xiu, R. Aging 2019, 11, 6120–6133.
[https://doi.org/10.18632/aging.102171]

-
Valotto Neto, L. J.; Reverete de Araujo, M.; Moretti Junior, R. C.; Mendes Machado, N.; Joshi, R. K.; Dos Santos Buglio, D.; Barbalho Lamas, C.; Direito, R.; Fornari Laurindo, L.; Tanaka, M.; Barbalho, S. M. Antioxidants 2024, 13, 393.
[https://doi.org/10.3390/antiox13040393]

-
Hunt, T.; Pontifex, M. G.; Vauzour, D. FEBS Lett. 2024, 598, 2949–2962.
[https://doi.org/10.1002/1873-3468.14988]

-
Domínguez-López, I.; López-Yerena, A.; Vallverdú-Queralt, A.; Pallàs, M.; Lamuela-Raventós, R. M.; Pérez, M. Nutr. Rev. 2025, 83, e533–e546.
[https://doi.org/10.1093/nutrit/nuae034]

-
Chu, Z.; Han, S.; Luo, Y.; Zhou, Y.; Zhu, L.; Luo, F. Crit. Rev. Food Sci. Nutr. 2024, 64, 10281–10302.
[https://doi.org/10.1080/10408398.2023.2222404]

-
Islam, M. R.; Rauf, A.; Akter, S.; Akter, H.; Al-Imran, M. I. K.; Islam, S.; Nessa, M.; Shompa, C. J.; Shuvo, M. N. R.; Abdulmonem, I. K. W. A.; Aljohani, A. S. M.; Imran, M.; Iriti, M. Mol. Cell. Biochem. 2025, 480, 3363–3383.
[https://doi.org/10.1007/s11010-025-05211-4]

-
Wang, R.; Zhang, Y.; Li, J.; Zhang, C. Neuroscience 2017, 344, 39–47.
[https://doi.org/10.1016/j.neuroscience.2016.08.051]

-
Shen, J.; Xu, L.; Qu, C.; Sun, H.; Zhang, J. Behav. Brain Res. 2018, 349, 1–7.
[https://doi.org/10.1016/j.bbr.2018.04.050]

-
Ferrer, R. A.; Chen, B.-Y.; Garcia, J. P. T.; Rejano, C. J. F.; Tsai, P.-W.; Hsueh, C.-C.; Tayo, L. L. Biology 2025, 14, 487.
[https://doi.org/10.3390/biology14050487]

-
Alberini, C. M. Physiol. Rev. 2009, 89, 121–145.
[https://doi.org/10.1152/physrev.00017.2008]

-
Bathina, S.; Das, U. N. Arch. Med. Sci. 2015, 11, 1164–1178.
[https://doi.org/10.5114/aoms.2015.56342]

-
Pisani, A.; Paciello, F.; Del Vecchio, V.; Malesci, R.; De Corso, E.; Cantone, E.; Fetoni, A. R.; J. Pers. Med. 2023, 13, 652.
[https://doi.org/10.3390/jpm13040652]

-
Alqahtani, S. M.; Al-Kuraishy, H. M.; Al Gareeb, A. I.; et al. Neuromolecular Med. 2025, 27, 36.
[https://doi.org/10.1007/s12017-025-08857-x]

-
Thiels, E.; Klann, E. Rev. Neurosci. 2001, 12, 327–345.
[https://doi.org/10.1002/env.476]

-
Petersen, R. C.; Smith, G. E.; Waring, S. C.; Ivnik, R. J.; Tangalos, E. G.; Kokmen, E. Arch. Neurol. 1999, 56, 303–308.
[https://doi.org/10.1001/archneur.56.3.303]

-
Petersen, R. C.; Morris, J. C. Arch. Neurol. 2005, 62, 1160–1167.
[https://doi.org/10.1001/archneur.62.7.1160]

-
Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J. P. E.; Tognolini, M.; Borges, G.; Crozier, A. Antioxid. Redox Signal. 2013, 18, 1818–1892.
[https://doi.org/10.1089/ars.2012.4581]

-
Ghafarimoghadam, M.; Mashayekh, R.; Gholami, M.; Fereydani, P.; Shelley-Tremblay, J.; Kandezi, N.; Sabouri, E.; Motaghinejad, M. Physiol. Behav. 2022, 244, 113652.
[https://doi.org/10.1016/j.physbeh.2021.113652]

-
Vorhees, C. V.; Williams, M. T. Nat. Protoc. 2006, 1, 848–858.
[https://doi.org/10.1038/nprot.2006.116]

-
She, L.; Sun, J.; Xiong, L.; Li, A.; Li, L.; Wu, H.; Ren, J.; Wang, W.; Liang, G.; Zhao, X. Phytomedicine 2024, 122, 155168.
[https://doi.org/10.1016/j.phymed.2023.155168]

-
Hediyal, T. A.; Vichitra, C.; Anand, N.; Bhaskaran, M.; Essa, S. M.; Kumar, P.; Qoronfleh, M. W.; Akbar, M.; Kaul-Ghanekar, R.; Mahalakshmi, A. M.; Yang, J.; Song, B.-J.; Monaghan, T. M.; Sakharkar, M. K.; Chidambaram, S. B. Front. Immunol. 2024, 15, 1324018.
[https://doi.org/10.3389/fimmu.2024.1324018]

-
Tang, G.-Y.; Meng, X.; Gan, R.-Y.; Zhao, C.-N.; Liu, Q.; Feng, Y.-B.; Li, S.; Wei, X.-L.; Atanasov, A. G.; Corke, H.; Li, H.-B. Int. J. Mol. Sci. 2019, 20, 6196.
[https://doi.org/10.3390/ijms20246196]

-
Cummings, J.; Lee, G.; Zhong, K.; Fonseca, J.; Taghva, K. Alzheimers Dement. 2021, 7, e12179.
[https://doi.org/10.1002/trc2.12179]

-
Egan, M. F.; Kost, J.; Tariot, P. N.; Aisen, P. S.; Cummings, J. L.; Vellas, B.; Sur, C.; Michelson, D. N. Engl. J. Med. 2018, 378, 1691–1703.
[https://doi.org/10.1056/NEJMoa1706441]