Anti-Inflammatory and Immunoregulatory Activities In Vitro of Coumarins Isolated from the Twigs and Leaves of Wikstroemia indica (L.) C.A. Mey.
Abstract
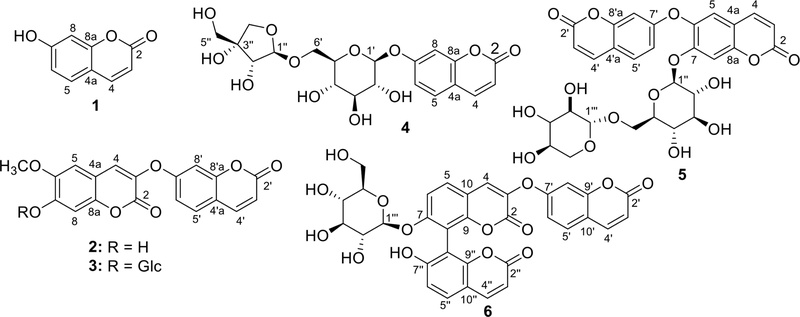
Six coumarins were isolated from two bioactive fractions of the 70% ethanol extract from the twigs and leaves of Wikstroemia indica (L.) C.A. Mey. (Thymelaeaceae), namely the ethyl acetate fraction (WIE) and the water fraction (WIW). These compounds were identified as umbelliferone (1), daphnoretin (2), daphnorin (3), adicardin (4), wikstronutin (5), and triumbelletin-7-O-β-D-glucoside (6). Compound 4 is reported for the first time in the genus Wikstroemia, whereas compound 5 is newly identified in this species. All six compounds were evaluated for their effects on Interleukin-1 beta (IL-1β) and IL-10 secretion in Lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophages, among which compounds 1, 2, 5, and 6 showed stronger activity. These four coumarin derivatives further suppressed the production of Foxp3, Interferon-alpha (IFN-α), IFN-γ, and IL-17 in LPS-stimulated CD4+ T cells. The IC50 values ranged from 18.84 ± 1.16 to 43.45 ± 5.34 μM for Foxp3, 8.96 ± 1.45 to 22.42 ± 4.12 μM for IFN-α, 9.28 ± 1.23 to 16.83 ± 2.15 μM for IFN-γ, and 12.62 ± 2.57 to 22.92 ± 4.41 μM for IL-17. Notably, compound 6 exhibited the strongest immunomodulatory effect. Overall, the findings suggest that coumarins from W. indica possess promising immunomodulatory and anti-inflammatory potential.
Keywords:
Wikstroemia indica, Coumarin, Cytokines, CD4+ T cells, ImmunomodulatoryIntroduction
Chronic inflammation and immune-mediated diseases, including rheumatoid arthritis, inflammatory bowel disease, asthma, and autoimmune disorders, constitute major global health challenges associated with significant morbidity, impaired quality of life, and increased mortality.1,2 Current therapies often suffer from limited efficacy and adverse safety profiles. Therefore, it is necessary to develop novel immunomodulatory agents.1,3
Coumarin derivatives have emerged as promising candidates for the treatment of inflammatory and immunoregulatory disorders.4,5 This group of compounds acts in a “multi-target” manner in immune modulation. They have potential therapeutic applications for autoimmune diseases due to their ability to intervene in a wide range of cytokines and immune signaling pathways, while simultaneously improving the ratio of regulatory T cells (Treg) to pro-inflammatory T cells (Th1, Th17).6
The initiation, progression, and resolution of inflammatory and autoimmune disorders are coordinated by a complex network of cytokines and immune-regulatory signaling pathways. Pro-inflammatory mediators such as IL-1β and IL-17 contribute directly to tissue damage and chronic disease progression, whereas IL-10 and Foxp3-expressing regulatory T cells play essential roles in dampening excessive immune activation and maintaining immune homeostasis.1,3,7 Additionally, IFN-α and IFN-γ are central to antiviral immunity and play critical roles in modulating both innate and adaptive immune responses.8
Wikstroemia indica (L.) C.A. Mey. (W. indica) is a small shrub belonging to the Thymelaeaceae family and found in some regions of Asia well as Australia. In Vietnam, this plant is called “Niet do” and has been used to treat various diseases such as asthma, parotitis, and tonsillitis.9 Phytochemical and pharmacological investigations have shown that W. indica contains numerous secondary metabolites such as flavonoids, coumarins, and lignans, and exhibits various biological activities including anticancer, anti-inflammatory, and anti-HIV effects. Among these, coumarins are the major class of phytochemicals in this plant.10 In some recent studies, extracts and isolated compounds from W. indica have been reported to reduce serum IgE and IL-4 concentrations in mice model, thereby demonstrating the potential of this species in the treatment of allergy-related contact dermatitis.11,12 However, most previous studies have focused on classical inflammatory markers in macrophage models, with limited investigation into immunomodulatory activities in CD4+ T cells or into higher-order pro-inflammatory cytokines such as IFN-α, IFN-γ, IL-17, and the T-cell regulatory factor Foxp3.
In our previous study on coumarins from Paramignya scandens, we reported significant inhibitory effects on LPS-induced IL-1β and IL-10 secretion in RAW 264.7 macrophages.13 Building on these findings, and the traditional uses as well as known pharmacological profiles of W. indica, the present study employs bioactivity-guided fractionation to isolate coumarins from this plant, and evaluates their anti-inflammatory and immunomodulatory activities in LPS-stimulated RAW 264.7 macrophages and CD4+ T cells with a particular focus on Foxp3 modulation and the pro-inflammatory cytokines IFN-α, IFN-γ, and IL-17.
Experimental
General experimental procedures – Bruker 500 and 600 MHz FT-NMR spectrometers (Karlsruhe, Germany) with TMS as an internal standard were used for recording 1D and 2D NMR spectra. Open column chromatography (CC) was conducted using silica gel (Merck, 0.040‒0.063 mm particle size), RP-C18 (Merck, 30‒50 µm particle size), Diaion HP-20 (Mitsubishi Chemical Corp., Fujii, Tokyo, Japan), and MCI gel (Beijing Huideyi Technology Co., Ltd., CHP20P, 75‒150 μm). Thin-layer chromatography (TLC) was carried out by using silica gel 60F254 (Merck, 0.25 mm) and RP-18F254 (Merck, 0.25 mm) plates. Spots were visualized under UV light (254 and 366 nm) or by spraying with 10% sulfuric acid in 96% ethanol (EtOH) followed by heating at 105°C. Semi-preparative HPLC was performed by HPLC 1525 Binary Pumps with a 2489 UV-Vis detector (Waters Corp, USA) and an ODS column (YMC-Pack-ODS-A, 250 × 10 mm i.d., 5 µM, Kyoto, Japan).
Plant materials – The twigs and leaves of Wikstroemia indica (L.) C.A. Mey. were collected in Viet Yen district, Bac Giang province, Vietnam, in September 2022 and identified by MSc. Nguyen Quynh Nga and MSc. Phan Van Truong from the Medicinal Material Resources Center, NIMM, Vietnam. A voucher specimen (DV-080922) was deposited at the NIMM Herbarium.
Extraction and bioassay-guided isolation – The twigs and leaves of W. indica (5.0 kg) were refluxed with 70% EtOH (12 L × 3 hours × 3 times) at 70°C. The extracts were combined, filtered, and the solvent was recovered under reduced pressure to yield a residue (415.0 g, WIT). This residue was dispersed in hot water and subsequently extracted with solvents of increasing polarity, including n-hexane and ethyl acetate (EtOAc), followed by solvent recovery to produce three extracts: the n-hexane extract (175.6 g, WIH), the EtOAc extract (55.1 g, WIE), and a water-soluble extract (150.5 g, WIW).
The evaluation of the effects of WIT and its fractions (WIH, WIE, and WIW) on IL-1β and IL-10 induced secretion in RAW 264.7 macrophages was performed according to the method described by Nguyen et al.13
Isolation and purification of compounds from WIE and WIW fractions – Six compounds (1–6) were obtained from WIE and WIW fractions through chromatographic separation and crystallization (see Supplementary File).
Umbelliferone (1) – 1H-NMR (600 MHz, acetone-d6): δ 6.15 (1H, d, J = 9.6 Hz, H-3), 7.85 (1H, d, J = 9.6 Hz, H-4), 7.48 (1H, d, J = 8.4 Hz, H-5), 6.82 (1H, dd, J = 8.4, 1.8 Hz, H-6), 6.74 (1H, d, J = 1.8 Hz, H-8); 13C-NMR (150 MHz, acetone-d6): δ 162.3 (C-2), 112.6 (C-3), 144.9 (C-4), 112.6 (C-4a), 130.4 (C-5), 113.9 (C-6), 161.3 (C-7), 103.2 (C-8), 156.9 (C-8a).
Daphnoretin (2) – 1H-NMR (600 MHz, acetone-d6): δH 7.82 (1H, s, H-4), 7.21 (1H, s, H-5), 6.88 (1H, s, H-8), 6.31 (1H, d, J = 9.6 Hz, H-3′), 7.97 (1H, d, J = 9.6 Hz, H-4′), 7.68 (1H, d, J = 8.4 Hz, H-5′), 7.08 (1H, dd, J = 8.4, 2.4 Hz, H-6′), 7.06 (1H, d, J = 2.4 Hz, H-8′), 3.89 (3H, s, 6-OCH3); 13C-NMR (150 MHz, aceton-d6): δ 161.0 (C-2), 135.7 (C-3), 130.9 (C-4), 110.1 (C-4a), 109.4 (C-5), 145.7 (C-6), 150.3 (C-7), 102.7 (C-8), 147.4 (C-8a), 56.6 (6-OCH3), 160.5 (C-2′), 114.8 (C-3′), 144.3 (C-4′), 115.3 (C-4′a), 130.5 (C-5′), 114.1 (C-6′), 157.6 (C-7′), 104.7 (C-8′), 156.3 (C-8′a).
Daphnorin (3) – 1H-NMR (600 MHz, DMSO-d6): δ 7.87 (1H, s, H-4), 7.24 (1H, s, H-5), 6.88 (1H, s, H-8), 6.31 (1H, d, J = 9.6 Hz, H-3ʹ), 7.97 (1H, d, J = 9.6 Hz, H-4′), 7.68 (1H, d, J = 8.4 Hz, H-5′), 7.08 (1H, dd, J = 8.4, 2.4 Hz, H-6′), 7.06 (1H, d, J = 2.4 Hz, H-8′), 3.89 (3H, s, 6-OCH3), 5.04 (1H, d, J = 5.4 Hz, H-1′′), 3.32 (2H, m, H-2′′, H-3′′), 3.18 (1H, m, H-4′′), 3.43 (1H, m, H-5′′), 3.69 (1H, m, H-6′′a), 3.43 (1H, m, H-6′′b); 13C-NMR (150 MHz, DMSO-d6): δ 156.9 (C-2), 137.1 (C-3), 129.9 (C-4), 109.5 (C-5), 146.4 (C-6), 149.1 (C-7), 103 (C-8), 146.7 (C-8a), 112.2 (C-4a), 160.0 (C-2′), 114.0 (C-3′), 144.0 (C-4′), 115.3 (C-4′a), 130.5 (C-5′), 114.1 (C-6′), 157.6 (C-7′), 104.7 (C-8′), 156.3 (C-8′a), 99.7 (C-1′′), 76.8 (C-2′′), 73.1 (C-3''), 69.6 (C-4''), 77.1 (C-5''), 60.7 (C-6''), 56.1 (6-OCH3).
Adicardin (4) – 1H-NMR (500 MHz, MeOH-d4): δ 6.32 (1H, d, J = 9.5 Hz, H-3), 7.93 (1H, d, J = 9.4 Hz, H-4), 7.60 (1H, d, J = 8.6 Hz, H-5), 7.11 (1H, d, J = 8.6 Hz, H-6), 7.15 (1H, s, H-8), 5.00 (1H, ovl, H-1'), 3.72 (1H, m, H-2'), 3.51 (1H, m, H-3'), 3.36 (1H, m, H-4'), 3.99 (1H, m, H-5'), 4.07 (1H, m, H-6'a), 3.63 (1H, m, H-6'b), 5.00 (1H, ovl, H-1''), 3.52 (1H, m, H-2''), 4.07 (1H, m, H-4''a), 3.79 (1H, m, H-4''b), 3.63 (2H, m, H-5''); 13C-NMR (125 MHz, MeOH-d4): δ 162.0 (C-2), 112.9 (C-3), 144.3 (C-4), 113.9 (C-4a), 129.0 (C-5), 114.0 (C-6), 160.7 (C-7), 103.7 (C-8), 155.3 (C-8a), 100.6 (C-1'), 75.8 (C-2'), 73.4 (C-3'), 70.3 (C-4'), 76.9 (C-5'), 67.6 (C-6'), 109.8 (C-1''), 76.6 (C-2''), 79.1 (C-3''), 73.6 (C-4''), 64.2 (C-5'').
Wikstronutin (5) – 1H-NMR (500 MHz, MeOH-d4): δ 6.36 (1H, d, J = 9.5 Hz, H-3), 7.89 (1H, ovl, H-4), 7.45 (1H, s, H-5), 7.48 (1H, s, H-8), 6.29 (1H, d, J = 9.5 Hz, H-3ʹ), 7.89 (1H, ovl, H-4ʹ), 7.57 (1H, d, J = 8.6 Hz, H-5ʹ), 6.95 (1H, dd, J = 8.6, 2.2 Hz, H-6ʹ), 6.83 (1H, d, J = 2.2 Hz, H-8ʹ), 5.05 (1H, d, J = 7.8 Hz, H-1ʺ), 3.29 (1H, ovl, H-2ʺ), 3.29 (1H, ovl, H-3ʺ), 3.29 (1H, ovl, H-4ʺ), 3.77 (1H, m, H-5ʺ), 4.07 (1H, d, J = 10.7 Hz, H-6ʺa), 3.64 (1H, m, H-6ʺb), 4.30 (1H, d, J = 7.0 Hz, H-1‴), 3.29 (1H, ovl, H-2‴), 3.41 (1H, t, J = 9.1 Hz, H-3‴), 3.29 (1H, ovl, H-4‴), 3.92 (1H, dd, J = 11.4, 5.4 Hz, H-5‴a), 3.17 (1H, t, J = 10.9 Hz, H-5‴b); 13C-NMR (125 MHz, MeOH-d4): δ 163.0 (C-2), 115.4 (C-3), 145.5 (C-4), 114.5 (C-4a), 122.1 (C-5), 153.9 (C-6), 154.0 (C-7), 106.6 (C-8), 142.1 (C-8a), 162.9 (C-2ʹ), 115.3 (C-3ʹ), 145.0 (C-4ʹ), 115.6 (C-4ʹa), 130.7 (C-5ʹ), 114.9 (C-6ʹ), 163.0 (C-7ʹ), 105.1 (C-8ʹ), 156.7 (C-8ʹa), 101.9 (C-1ʺ), 74.6 (C-2ʺ), 77.9 (C-3ʺ), 71.0 (C-4ʺ), 77.2 (C-5ʺ), 70.7 (C-6ʺ), 105.9 (C-1‴), 75.0 (C-2‴), 78.0 (C-3‴), 71.4 (C-4‴), 66.9 (C-5‴).
Triumbelletin-7-O-β-D-glucoside (6) – 1H-NMR (600 MHz, MeOH-d4): δ 7.73 (1H, s, H-3), 7.32 (1H, d, J = 8.4 Hz, H-5), 7.42 (1H, d, J = 9.0 Hz, H-6), 6.30 (1H, d, J = 9.6 Hz, H-3ʹ), 7.84 (1H, ovl, H-4ʹ), 7.51 (1H, d, J = 8.4 Hz, H-5ʹ), 6.94 (1H, d, J = 8.4 Hz, H-6ʹ), 7.01 (1H, ovl, H-8ʹ), 6.13 (1H, d, J = 9.6 Hz, H-3ʺ), 7.84 (1H, ovl, H-4ʺ), 7.61 (1H, d, J = 9.0 Hz, H-5ʺ), 7.02 (1H, ovl, H-6ʺ), 5.07 (1H, d, J = 7.8 Hz, H-1‴), 3.18 (1H, m, H-2‴), 3.42 (1H, m, H-3‴), 3.33 (1H, m, H-4‴), 3.47 (1H, m, H-5‴), 3.91 (1H, dd, J = 12.0, 2.4 Hz, H-6‴a), 3.68 (1H, dd, J = 12.0, 6.0 Hz, H-6‴b); 13C-NMR (150 MHz, MeOH-d4): δC 159.0 (C-2), 138.8 (C-3), 130.9 (C-4), 130.3 (C-5), 115.4 (C-6), 159.4 (C-7), 111.9 (C-8), 151.8 (C-9), 151.1 (C-10), 160.8 (C-2ʹ), 115.4 (C-3ʹ), 146.1 (C-4ʹ), 130.5 (C-5ʹ), 114.9 (C-6ʹ), 160.6 (C-7ʹ), 106.1 (C-8ʹ), 156.5 (C-9ʹ), 116.4 (C-10ʹ), 163.9 (C-2ʺ), 113.5 (C-3ʺ), 146.1 (C-4ʺ), 130.5 (C-5ʺ), 114.1 (C-6ʺ), 162.7 (C-7ʺ), 108.2 (C-8ʺ), 154.9 (C-9ʺ), 112.5 (C-10ʺ), 102.6 (C-1‴), 74.7 (C-2‴), 78.2 (C-3‴), 71.2 (C-4‴), 78.3 (C-5‴), 62.6 (C-6‴).
Cell culture of RAW 264.7 macrophages and the assays for IL-1β and IL-10 inducible activity were performed according to the protocol described by Nguyen et al..13 Murine monocyte/macrophage RAW 264.7 (ATCC TIB-71) cell line was obtained from American Type Culture Collection (ATCC; Manassas, VA, USA) and was maintained for no more than 25 passages. Lipopolysaccharide (LPS) (Sigma Aldrich, St. Louis, MO, USA). DMSO 1% (Sigma Aldrich, St. Louis, MO, USA). ELISA kits for mouse IL-1 β and IL-10 were purchased from Thermo Fisher.
Cell culture ‒ CD4+ cells – (ATCC, Manassas, VA, USA) were cultured in RPMI-1640 medium containing 1% penicillin-streptomycin (MediaTech, Herndon, VA, USA), 50 μM β-mercaptoethanol (Gibco, Invitrogen, Grand Island, NY), and 1% L-glutamine (Gibco, Carlsbad, CA, USA) in an incubator maintained at 37°C with 5% CO2.
Assay for the inducible activity of immunoregulatory factors (Foxp3, IL-17, IFN-α, and IFN-γ) in CD4+ T Cells ‒ CD4+ T cells were seeded in 96-well tissue culture plates at a density of 1 × 104 cells/well in RPMI 1640-medium supplemented with 10% pooled human serum (H6914, Sigma), and incubated at 37°C in 5% CO2 for 12 hours. The cells were then activated with lipopolysaccharide (LPS, Sigma Aldrich, St. Louis, MO, USA) at a final concentration of 5 ng/mL in the presence or absence of either the crude extracts (20 μg/mL) or isolated compounds at various concentrations. After a further 24 hours of incubation, the culture supernatants were collected, and the levels of Foxp3, IL-17, IFN-α, and IFN-γ were quantified using commercially available ELISA kits (Foxp3 ELISA Kit [ab289644, Abcam], IL-17 ELISA kit [ab119535, Abcam], IFN-α ELISA kit [#BMS216, Thermo Fisher], and IFN-γ ELISA kit [#KHC4021, Thermo Fisher]), following the manufacturers’ instructions. CD4+ T cells incubated with 1% DMSO (Sigma Aldrich) and LPS (5 ng/mL) served as the control group. All assays were performed in quintuplicate and independently analyzed.
Statistical analysis ‒ Using GraphPad Prism 9.5.0, quantitative data are displayed as the mean ± SEM and analyzed using the one-way analysis of variance (ANOVA) and Dunnett's test. P values below 0.05 were deemed statistically significant.
Results and Discussion
The immunomodulatory activities of the total extract and its fractions were evaluated in RAW 264.7 macrophages stimulated with 5 ng/mL LPS. Treatments with the extract and its fractions (20 μg/mL) significantly modulated cytokine production compared with the LPS-stimulated controls (Fig. 1). IL-1β inhibition was observed in the order: WIW (49.74 pgl mL) ≈ WIE (51.23 pg/mL) > WIH (72.38 pg/mL) > WIT (90.58 pg/mL) (Fig. 1A). For IL-10 secretion, the stimulatory effects followed the order: WIW (375.89 pg/mL) ≈ WIE (360.41 pg/mL) > WIH (243.19 pg/mL) > WIT (172.81 pg/mL) (Fig. 1B). Based on these findings, WIW and WIE were selected for further isolation of potential immunoregulatory compounds.
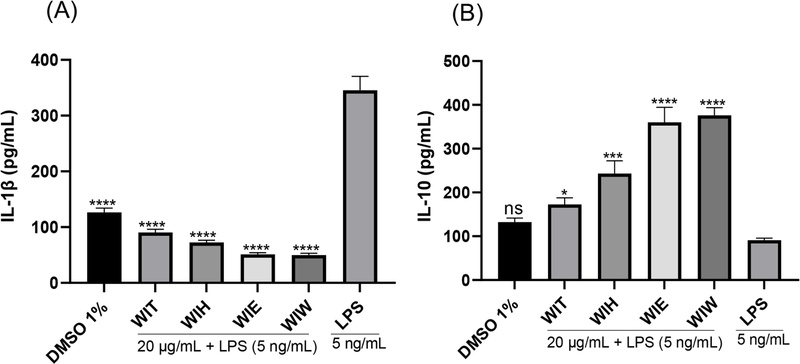
The modulatory effects of the ethanol extract and its fractions from W. indica on LPS-induced IL-1β (A) and IL-10 (B) secretion in RAW 264.7 macrophages. Ethanol extract (WIT), n-hexane fraction (WIH), ethyl acetate fraction (WIE), and water fraction (WIW) were evaluated. Data are presented as mean ± SEM (n = 5). Statistical significance was determined by one-way ANOVA followed by Tukey’s post hoc test. ns: not statistically significant; **p < 0.01, ***p < 0.001, ****p < 0.0001 compared to the LPS-treated group only.
Isolated compounds ‒ Guided by the prominent activities of WIW and WIE fractions on IL-1β and IL-10 cytokines secretion, six coumarin compounds—including umbelliferone (1), daphnoretin (2), daphnorin (3), adicardin (4), wikstronutin (5), and triumbelletin-7-O-β-D-glucoside (6) were isolated by successive silica gel and RP-C18 CC, crystallization in appropriate solvents, and further purification by semi-preparative HPLC (Fig. 2).
Their chemical structures were elucidated by MS and NMR spectroscopic analyses and confirmed comparison with reported data (See the supplementary file).
Screening based on anti-inflammatory responses in LPS-stimulated RAW 264.7 cells showed that all six coumarins (1–6) at 10 μM enhanced IL-10 secretion while suppressing IL-1β release (Fig. 3). Compounds 3 and 4 slightly increased IL-10 levels (132.14 and 122.64 pg/mL, respectively) compared with the LPS control (109.43 pg/mL), but the differences were not statistically significant, and their inhibition of IL-1β was weaker than that of the other coumarins. Therefore, compounds 1, 2, 5, and 6 were further examined at concentrations ranging from 1 to 30 μM. As shown in Fig. 4, these compounds enhanced IL-10 secretion and suppressed IL-1β production in a concentration-dependent manner with IC50 values for IL-1β inhibition were 6.4 ± 1.23 μM (1), 31.1 ± 3.07 μM (2), 19.98 ± 1.92 μM (5), and 14.88 ± 1.04 μM (6). To further characterize their immunomodulatory effects, the inhibitory activities of these compounds on IFN-α, IFN-γ, IL-17, and Foxp3 were also examined (Fig. 5 and Table 1).
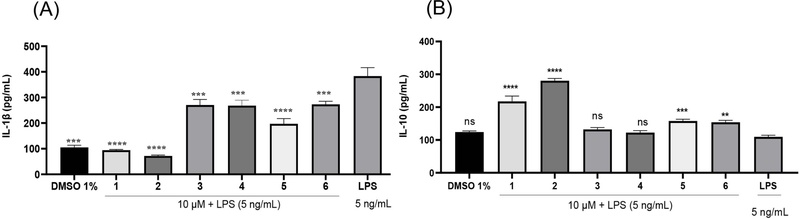
Modulatory effects of compounds 1–6 on LPS-induced secretion in RAW 264.7 macrophages: (A) IL-1β and (B) IL-10. Values are mean ± SEM (n = 5). Statistical significance was determined by one-way ANOVA followed by Tukey’s test; ns, not significant; **p < 0.01, ***p < 0.001; ****p < 0.0001 compared to the cells treated with LPS only.
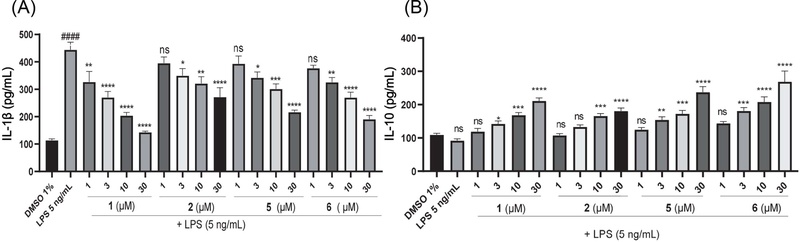
Modulation effects of compounds (1, 2, 5 and 6) on LPS-induced IL-1β and IL-10 secretion in RAW 264.7 macrophages. (A) Inhibition of IL-1β secretion. Cells were treated with DMSO 1%, LPS (5 ng/mL), or the compounds (1–30 μM) for 12 h. (B) Stimulation of IL-10 secretion. Cells were incubated with LPS (5 ng/mL) in the absence (DMSO 1%) or presence of the compounds (1–30 μM) for 12 h. Values are mean ± SEM (n = 5). Statistical significance was determined by one-way ANOVA followed by Tukey’s test; ns, not significant; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 vs. LPS alone.
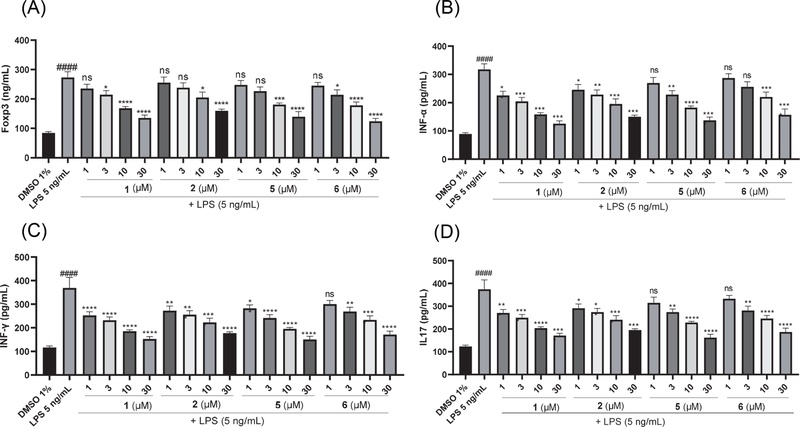
In vitro inhibitory effects of compounds 1, 2, 5, and 6 on (A) Foxp3, (B) IFN-α, (C) IFN-γ, and (D) IL-17 levels in CD4⁺ T cells. Cells were treated with the compounds (1, 3, 10, or 30 μM) in the presence of LPS (5 ng/mL) for 24 h. DMSO served as the physiological control and LPS-only as the pathological control. Data are mean ± SEM (n = 5). Statistical significance was determined by one-way ANOVA followed by Tukey’s test: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 vs. LPS control; ###p < 0.001 vs. DMSO control.
From Fig. 5, it can be observed that the inhibitory effects of compounds 1, 2, 5, and 6 on the production of IFN-α, IFN-γ, and IL-17, as well as on the expression of Foxp3, in LPS-stimulated CD4⁺ T cells increased progressively with the testing concentrations. Compound 1 showed stronger inhibition of the pro-inflammatory cytokines IFN-α, IFN-γ, and IL-17 with IC50 values from 8.96 ± 1.45 to 12.62 ± 2.57 μM compared to other coumarins, but its ability to inhibit Foxp3 (IC50, 23.38 ± 3.47 μM) was weaker than that of compounds 5 and 6. Compound 6, a tricoumarin glycoside—exhibited stronger inhibitory activity on the expression of Foxp3 with IC50 values (18.84 ± 1.16 μM), while showing comparable inhibition of pro-inflammatory cytokines relative to the other compounds. These results suggest that this tricoumarin possesses superior immunomodulatory and anti-inflammatory potential compared with the simple coumarins and dicoumarins investigated in this study.
In comparison to previous studies, which primarily focused on the roots of W. indica and led to the isolation of over 100 compounds, including coumarins 1, 2, 3, and 6.10 In this study, we targeted the twigs and leaves of this species with an emphasis on anti-inflammatory and immunomodulatory activity. This investigation resulted in the isolation of six coumarins (1–6), among which compound 5 was identified for the first time from W. indica, and compound 4 was reported for the first time from the genus Wikstroemia. In addition, previous studies on extracts and compounds from W. indica have showed their in vitro anti-inflammatory effects, including inhibition of NO production, and suppression of inflammatory mediators via pathways.10 In addition the extract of W. indica leaves and stems (1%, v/v) exhibited immunomodulatory anti-inflammatory activity by attenuating DNCB-induced atopic skin lesions in mice, associated with reduced serum IgE and IL-4 levels.12 However, information on higher-order cytokines (IFN-α, IFN-γ, IL-17) and the T-cell transcription factor Foxp3 in LPS-stimulated CD4⁺ T cells remain scarce, particularly for coumarins from W. indica. In this study, all six coumarins (1–6) showed significantly effected IL-1β and IL-10 secretion in LPS-stimulated RAW 264.7 cells. Among them, compounds 1, 2, 5, and 6 exhibited pronounced anti-inflammatory and immunomodulatory activities by markedly inhibiting the secretion IFN-α, IFN-γ, and IL-17, as well as reducing Foxp3 expression in LPS-stimulated CD4+ T cells. The data in this study provide precise IC50 values for both cytokine suppression and Foxp3 modulation, thereby quantitatively defining the immunoregulatory potency of each compound. These results link specific coumarin structural types (simple coumarins, dicoumarins, and tricoumarins) to the modulation of CD4⁺ T-cell subsets and the regulation of cytokines. In addition, previous studies have reported the immunomodulatory mechanisms and signaling pathways such as MAPKs, TGF-β/Smad, NF-κB, and PI3K/AKT for several simple coumarins, including umbelliferone, osthole, daphnetin, and other coumarins in the treatment of autoimmune diseases.6 Although mechanistic assays were not performed in this study, the observed modulation of cytokines (IFN-α, IFN-γ, IL-17, IL-10) and Foxp3 expression is consistent with the involvement of signaling pathways previously reported for coumarins, including STAT3, NF-κB, and MAPKs. This calls for focused, pathway-specific research in further studies. Based on comparative potency, the present work proposes compound 6, a glycosid tricoumarin, as a promising candidate for further mechanistic studies on signaling pathways, given its markedly stronger in vitro immunomodulatory activity than the other coumarins tested. These findings are consistent with, and further substantiate, previous reports on the anti-inflammatory and immunomodulatory potential of extracts and isolated compounds from W. indica, while also elucidating the medicinal value of this species in Vietnamese traditional medicine.
In conclusion, six coumarin compounds (1–6) were isolated from W. indica and screened for their anti-inflammatory and immunomodulatory activities in vitro. Among them, compounds 1, 2, 5, and 6 exhibited significant inhibitory effects on the secretion of the immunoregulatory factor Foxp3 and the pro-inflammatory cytokines IFN-α, IFN-γ, and IL-17 in LPS-stimulated CD4+ T cells. Of note, compound 6 emerges as a promising candidate for immunomodulatory and anti-inflammatory applications. These discoveries added to expanding knowledge on the phytochemistry and biological activities of W. indica supplied useful information for further research on the species.
Acknowledgments
This study was financially supported by the Ministry of Science and Technology Vietnam [NĐT.85.KR/20, contract no. 09/2020/HĐ-NĐT signed 09/9/2020-MOST], and The National Research Foundation of Korea (NRF) grant funded by the Korean government [2019K1A3A1A05088041].
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D. W.; Fasano, A.; Miller, G. W.; Miller, A. H.; Mantovani, A.; Weyand, C. M.; Barzilai, N.; Goronzy, J. J.; Rando, T. A.; Effros, R. B.; Lucia, A.; Kleinstreuer, N.; Slavich, G. M. Nat. Med. 2019, 25, 1822–1832.
[https://doi.org/10.1038/s41591-019-0675-0]

-
Safiri, S.; Kolahi, A.-A.; Cross, M.; Hill, C.; Smith, E.; Carson-Chahhoud, K.; Mansournia, M. A.; Almasi-Hashiani, A.; Ashrafi-Asgarabad, A.; Kaufman, J.; Sepidarkish, M.; Shakouri, S. K.; Hoy, D.; Woolf, A. D.; March, L.; Collins, G.; Buchbinder, R. Arthritis. Rheumatol. 2021, 73, 702–714.
[https://doi.org/10.1002/art.41571]

-
Turner, M. D.; Nedjai, B.; Hurst, T.; Pennington, D. J. Biochim. Biophys. Acta 2014, 1843, 2563–2582.
[https://doi.org/10.1016/j.bbamcr.2014.05.014]

-
Saadati, F.; Modarresi Chahardehi, A.; Jamshidi, N.; Jamshidi, N.; Ghasemi, D. Curr. Res. Pharmacol. Drug Discov. 2024, 7, 100202.
[https://doi.org/10.1016/j.crphar.2024.100202]

-
Mustafa, Y. F. Phytomedicine Plus 2025, 5, 100880.
[https://doi.org/10.1016/j.phyplu.2025.100880]

-
Li, Y.; Wang, G. Q.; Li, Y.-B. Front. Immunol. 2024, 15, 1432846.
[https://doi.org/10.3389/fimmu.2024.1432846]

-
Ruiz de Morales, J. M. G.; Puig, L.; Daudén, E.; Cañete, J. D.; Pablos, J. L.; Martín, A. O.; Juanatey, C. G.; Adán, A.; Montalbán, X.; Borruel, N.; Ortí, G.; Holgado-Martín, E.; García-Vidal, C.; Vizcaya-Morales, C.; Martín-Vázquez, V.; González-Gay, M. Á. Autoimmun. Rev. 2020, 19, 102429.
[https://doi.org/10.1016/j.autrev.2019.102429]

-
Ivashkiv, L. B.; Donlin, L. T. Nat. Rev. Immunol. 2014, 14, 36–49.
[https://doi.org/10.1038/nri3581]

- Vo, V. C. Vietnamese Medicinal Plant Dictionary. Medical Publishing House: Vietnam, 1997; p 875.
-
Huan, D. Q.; Hop, N. Q.; Son, N. T. Curr. Pharm. Biotechnol. 2024, 25, 563–598.
[https://doi.org/10.2174/1389201024666230606122116]

-
Jegal, J.; Park, N.-J.; Lee, S.-Y.; Jo, B.-G.; Bong, S.-K.; Kim, S.-N.; Yang, M. H. Evid. Based Complement. Alternat. Med. 2020, 2020, 4307161.
[https://doi.org/10.1155/2020/4307161]

-
Lee, S.-Y.; Park, N.-J.; Jegal, J.; Jo, B.-G.; Choi, S.; Lee, S. W.; Uddin, M. S.; Kim, S. N.; Yang, M. H. Nutrients 2020, 12, 173.
[https://doi.org/10.3390/nu12010173]

-
Nguyen, T. M.; Do, T. H.; Nguyen, T. T.; Vu, T. D.; Tran, T. H.; Nguyen, H. T.; Doan, T. P.; Oh, W. K.; Park, J. H. Nat. Prod. Sci. 2024, 30, 30–57.
[https://doi.org/10.20307/nps.2024.30.1.30]