
Cardioprotective Potential of a Marine-Derived Tetrapeptide (CAAP) from Paralichthys olivaceus with Dual Antioxidant and ACE-Inhibitory Activities
Abstract
Excessive lipid peroxidation and renin–angiotensin system dysregulation are central mechanisms linking oxidative stress to hypertension. In this study, a bioactive tetrapeptide, CAAP (Cys-Ala-Ala-Pro), isolated from the muscle hydrolysate of Paralichthys olivaceus (olive flounder), was evaluated for its dual antioxidant and antihypertensive actions. In Vero cells, CAAP significantly suppressed 2,2′-azobis(2-amidinopropane)-induced lipid peroxidation, reduced malondialdehyde accumulation, and preserved cell viability. Fluorescent imaging and flow cytometric analyses revealed that CAAP mitigated nuclear fragmentation and apoptotic sub-G1 cell populations through modulation of the Bcl-xL/Bax/caspase-3 axis, indicating cell protection. Molecular docking analysis demonstrated that CAAP interacts with the S′2 pocket of angiotensin-converting enzyme (ACE) via hydrogen bonding with Glu281, His513, Lys511, and Tyr520, supporting a strong inhibitory potential (IC50 = 39.3 µg/mL). In spontaneously hypertensive rats, oral administration of CAAP (40 mg/kg) elicited a sustained decrease in systolic blood pressure within 2–6 h post-treatment, comparable to that of captopril. Collectively, these findings highlight CAAP as a multifunctional marine peptide capable of attenuating oxidative and hypertensive injury through cell stabilization and ACE inhibition, suggesting its potential as a natural therapeutic or functional food ingredient for cardiovascular protection.
Keywords:
Flounder fish, Lipid peroxidation, ACE inhibition, Cysteine-Alanine-Alanine-ProlineIntroduction
Cardiovascular diseases (CVDs) remain the ledaing cause of mortality worldwide, accounting for approximately one-third of all global deaths each year.1 Among the multifactorial mechanisms implicated in CVD pathogenesis, oxidative stress and dysregulation of the renin-angiotensin system (RAS) constitute two pivotal biochemical pathways that converge to induce vascular dysfunction, endothelial injury, and hypertension.2,3 Excessive production of reactive oxygen species (ROS) perturbs redox homeostasis, promotes lipid peroxidation, and activates apoptotic signaling cascades, ultimately compromising vascular tone and structural integrity.4
Lipid peroxidation represents a free radical-driven chain reaction that leads to oxidative degradation of polyunsaturated fatty acids (PUFAs), yielding reactive intermediates such as peroxyl radicals and lipid hydroperoxides.5 These unstable intermediates decompose into cytotoxic aldehydes, including malondialdehyde (MDA), a widely recognized biomarker of oxidative lipid injury.6,7 Persistent lipid peroxidation disrupts membrane integrity, alters intracellular calcium flux, and accelerates endothelial apoptosis, thereby contributing to the progression of atherosclerosis and hypertensive vascular remodeling.8,9
The RAS further amplifies oxidative stress through angiotensin-converting enzyme (ACE)-mediated formation of angiotensin II, a potent vasoconstrictor that stimulates NADPH oxidase-dependent ROS production.10 This angiotensin II-induced oxidative cascade establishes a self-perpetuating feedback loop that exacerbates vascular inflammation and hypertension. Although synthetic ACE inhibitors such as captopril and enalapril are widely prescribed, their prolonged use is often associated with adverse reaction including cough, hypotension, and renal impairment.11 Consequently, there has been growing interest in discovering natural ACE inhibitors that exhibit concurrent antioxidant activity and improved safety profiles.12,13
In recent years, bioactive peptides derived from natural sources have attracted attention as multifunctional agents exhibiting antioxidant, antihypertensive, and anti-inflammatory properties.14,15 These peptides are typically liberated from precursor proteins via enzymatic hydrolysis and display superior biocompatibility and gastrointestinal stability compared with larger proteins.16 Notably, short peptides such as Ile–Pro–Pro (IPP) and Val–Pro–Pro (VPP) from casein exert significant blood pressure–lowering effects in both spontaneously hypertensive rats (SHRs) and clinical trials.17,18 Similarly, marine fish species including Pacific thread herring, rainbow trout, and triggerfish have been recognized as rich reservoirs of peptides with ACE-inhibitory and antioxidant potential.19–21
The olive flounder (Paralichthys olivaceus) is described in the Korean traditional medicine book Donguibogam as a nourishing food believed to “invigorates vital energy and restores deficiency”. Traditionally consumed to promote recovery and vitality, it has long been regarded as a restorative dietary resource. In modern biomedical research, bioactive peptides derived from marine fish protein hydrolysates have been widely reported to exhibit diverse health-promoting properties, including nutritional, antioxidative, antihypertensive, antimicrobial, anti-inflammatory, and immuno-modulatory activities.22,23 Moreover, olive flounder has emerged as one of the most commercially important aquaculture species in East Asia, providing a stable and abundant supply of raw material. Based on its long-standing traditional use, the multifunctional potential of marine-derived bioactive peptides, and its industrial relevance, olive flounder was selected as the experimental source in the study.
In our previous study, protein hydrolysates were prepared from olive flounder (Paralichthys olivaceus) muscle using eight different proteolytic enzymes. Among these, the α-chymotrypsin–derived hydrolysate exhibited the highest antioxidant activity. This hydrolysate was subsequently subjected to sequential purification by ultrafiltration, gel filtration chromatography, and reversed-phase high-performance liquid chromatography to isolate low-molecular-weight peptides. Peptide sequences were unambiguously determined by Q-TOF mass spectrometry, leading to the identification of a tetrapeptide composed of cysteine, alanine, alanine, and proline (CAAP) as one of the naturally occurring bioactive peptides present in olive flounder muscle protein hydrolysates.24
Among the isolated peptides, CAAP possesses structural features favorable for redox regulation and enzyme interaction, including a thiol-containing cysteine residue and a short peptide backbone enriched in hydrophobic amino acids. These characteristics are commonly associated with antioxidant and enzyme-modulating activities.24,25 Based on its natural occurrence, defined structure, and reported antioxidant activity, CAAP was selected as the target peptide for further investigation.
In this context, the present study aimed to evaluate the cardiovascular protective potential of CAAP by (i) assessing its ability to attenuate 2,2′-azobis(2-amidinopropane) (AAPH)-induced oxidative injury in Vero cells, (ii) characterizing its ACE-inhibitory activity through biochemical and molecular docking analyses, and (iii) determining its antihypertensive efficacy in SHRs. Through this integrative investigation, we sought to elucidate the mechanistic basis and therapeutic relevance of P. olivaceus-derived peptides in oxidative stress-related vascular disorders.
Experimental
Materials and reagents – The synthetic tetrapeptide CAAP (Cys-Ala-Ala-Pro; purity > 95%) was provided by HLB PEP Co., Ltd. (Gwangju, Republic of Korea). 2,2′-Azobis(2-amidinopropane) dihydrochloride (AAPH), diphenyl-1-pyrenylphosphine (DPPP), thiobarbituric acid (TBA), Hoechst 33342, propidium iodide (PI), and captopril were purchased from Sigma-Aldrich (St. Louis, USA). RPMI-1640 medium, fetal bovine serum (FBS), penicillin–streptomycin, trypsin–EDTA, and Dulbecco’s phosphate-buffered saline (DPBS) were obtained from Gibco (Waltham, USA). Primary antibodies against Bcl-xL, Bax, cleaved caspase-3, and GAPDH were obtained from Cell Signaling Technology (Danvers, MA, USA).
Cell culture and treatment – Vero cells (African green monkey kidney fibroblasts) were obtained from the Korean Cell Line Bank (Seoul, Republic of Korea). Cells were cultured in RPMI-1640 medium supplemented with 10% (v/v) FBS and 1% (v/v) penicillin-streptomycin, and maintained at 37°C in a humidified incubator with 5% CO₂. For experiments, cells were seeded at a density of 1 × 10⁵ cells/mL and allowed to attach overnight prior to treatment. Subsequently, cells were pretreated with CAAP (12.5–100 μg/mL) for 2 h, followed by exposure to AAPH (10 mM) for 24 h.
Lipid peroxidation assay – Lipid peroxidation was determined using the thiobarbituric acid reactive substances (TBARS) assay.6 After treatment, 200 μL of the culture supernatant was mixed with 400 μL of TBARS reagent containing 15% (w/v) trichloroacetic acid, 0.375% (w/v) thiobarbituric acid, and 0.25 M HCl. The mixture was heated at 95°C for 20 min and then cooled to room temperature. Following centrifugation at 3,000 rpm for 10 min, the absorbance of the supernatant was measured at 532 nm using a microplate reader. TBARS levels were calculated from a standard curve constructed with with 1,1,3,3-tetraethoxypropane and expressed as nmol malondialdehyde (MDA) equivalents per mg protein.
Fluorescence microscopy for lipid peroxidation and cell death – For the visualization of lipid peroxidation, cells were stained with DPPP (10 μg/mL) for 10 min at room temperature, washed twice with PBS, and observed under a fluorescence microscope (Olympus, Tokyo, Japan). Cell death and nuclear morphology were assessed by sequential staining with PI (10 μg/mL) for 10 min, followed by Hoechst 33342 (10 μg/mL) for 10 min. Fluorescence images were captured and analyzed using the microscope software provided by the manufacturer.
Cell cycle analysis – Apoptotic sub-G1 populations were determined by flow cytometry following the method of Nicoletti et al.26 After treatment, cells were harvested, washed with ice-cold PBS, and fixed in 70% (v/v) ethanol at 4°C for 30 min. Fixed cells were then washed twice with PBS and stained with PI (100 μg/mL) in the presence of RNase A (100 μg/mL) for 30 min in the dark. DNA content was analysed using a FACS Calibur flow cytometry (BD Biosciences, San Jose, CA, USA).
Western blot analysis – Cells (4 × 10⁵ cells/mL) were lysed in radioimmunoprecipitation assay (RIPA) buffer (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with protease and phosphatase inhibitor cocktails. Equal amounts of total protein (30 μg) were separated by 10% SDS-PAGE and transferred onto PVDF membranes (Millipore, Billerica, MA, USA). Membranes were blocked with 5% (w/v) bovine serum albumin (BSA) for 1 h at room temperature, and incubated overnight at 4°C with primary antibodies against Bcl-xL (1:1000), Bax (1:1000), cleaved caspase-3 (1:1000), and GAPDH (1:10,000). After washing with tris-buffered saline containing 0.1% tween-20 (TBST), the membranes were incubated with HRP-conjugated secondary antibodies (1:10,000) for 1 h at room temperature. Protein bands were visualized using an enhanced chemiluminescence (ECL) detection reagent (Thermo Fisher Scientific, Waltham, MA, USA) and quantified with ImageJ software (National Institutes of Health, Bethesda, MD, USA).
ACE inhibitory assay – ACE inhibitory activity of CAAP was evaluated using the synthetic substrate hippuryl-L-histidyl-L-leucine (HHL) according to the method of Cushman and Cheung.27 The 100 μL reaction mixture comprised 50 mM sodium borate buffer (pH 8.3), 300 mM NaCl, 5 mM HHL, and CAAP at various concentrations. After pre-incubation at 37°C for 10 min, the reaction was initiated by adding ACE (0.1 U/mL) and terminated after 30 min by addition of 1 N HCl. The liberated hippuric acid was extracted with ethyl acetate and quantified spectrophotometrically at 228 nm. The IC50 value was defined as the concentration of CAAP required to inhibit 50% of ACE activity.
Molecular docking simulation – Molecular docking of the CAAP peptide with human ACE was conducted using Discovery Studio 3.5 (Accelrys Inc., San Diego, CA, USA). The crystal structure of ACE (PDB ID: 1O86) was retrieved from the Protein Data Bank and all water molecules were removed. The CAAP peptide structure was constructed using the Builder module, energy-minimized, and prepared as the ligand. Docking was carried out using the CDOCKER algorithm with the CHARMm force field. Binding interactions-including hydrogen bonds, hydrophobic contacts, and electrostatic interactions-were analyzed, and binding energies were calculated as CDOCKER interaction energy (kcal/mol).
In Vivo blood pressure measurement in spontaneously hypertensive rats – Male spontaneously hypertensive rats (SHRs; 9–10 weeks old, 250–300 g) were obtained from SLC Japan (Shizuoka, Japan). Animals were housed two per cage under controlled environmental conditions (24 ± 1°C, 50 ± 10% relative humidity, 12 h light/dark cycle) with free access to standard chow and water. All experimental procedures were approved by the Institutional Animal Care and Use Committee (JEJU-2021-0067) and conducted in accordance with relevant guidelines for animal experimentation. Rats were randomly divided into three groups (n = 4 per group): control (saline), captopril (40 mg/kg), and CAAP peptide (40 mg/kg). Each treatment was administered orally once, and systolic blood pressure (SBP) was measured using the non-invasive tail-cuff method (CODA Monitor, Kent Scientific Co., Torrington, CT, USA). Measurements were performed before administration and at 2, 3, 6, 9, and 12 h post-dosing. For each time point, the mean of three consecutive measurements per rat was used for analysis.
Statistical analysis – All experimental data were obtained from four independent replicates (n = 4) and are expressed as the mean ± standard error of the mean (SEM). Statistical analyses were performed using one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test with SPSS software (version 11.5; IBM Corp., Armonk, NY, USA). For systolic blood pressure data, two-way ANOVA was applied, and differences among group were further evaluated using Tukey’s post hoc test. A p-value of less than 0.05 was considered statistically significant.
Results and Discussion
2,2′-azobis(2-amidinopropane) (AAPH, 10 mM) for 24 h markedly elevated the level of thiobarbituric acid reactive substances (TBARS), reflecting increased malondialdehyde (MDA) production and membrane lipid oxidation in Vero cells. However, pretreatment with the peptide CAAP (12.5–100 μg/mL) markedly and dose-dependently suppressed MDA formation (Fig. 1A), reaching minimal values of approximately 4.47 nmol at 50 μg/mL and 3.86 nmol at 100 μg/mL. Fluorescence imaging using DPPP staining further supported these findings, showing that CAAP markedly reduced the intense lipid peroxidation-associated fluorescence induced by AAPH exposure (Fig. 1B). From a cardiovascular standpoint, lipid peroxidation is a major initiating event in endothelial dysfunction, as oxidized phospholipids impair membrane fluidity, decrease nitric oxide (NO) bioavailability, and promote leukocyte adhesion to the vascular endothelium.28 CAAP can maintain the integrity of the endothelial barrier and reduce the risk of vascular inflammation and thrombus formation by inhibiting the production of peroxyl radicals induced by AAPH. This protective effect likely arises from the specific amino acid composition of CAAP: the cysteine (Cys) residue, with its reactive thiol (-SH) group, can donate electrons to reactive oxygen species (ROS), thereby interrupting lipid peroxidation chain reactions and safeguarding cellular membranes.29,30 Meanwhile, the alanine (Ala) and proline (Pro) residues may contribute by enhancing peptide solubility and facilitating interactions with lipid membranes, thereby further reinforcing the antioxidant defense system. These findings are consistent with previous reports demonstrating that fish-derived bioactive peptides can attenuate oxidative damage in vascular endothelial cells and retard the progression of atherosclerotic lesions.31–34
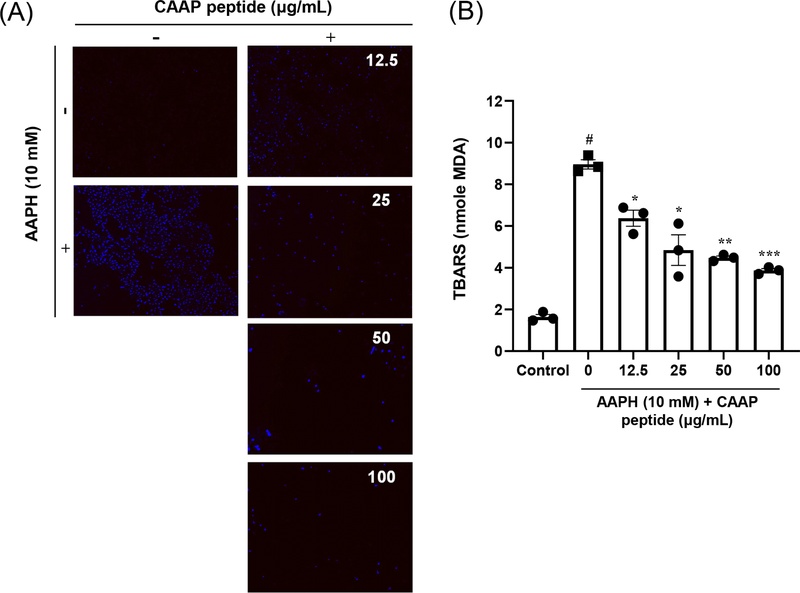
CAAP peptide inhibits AAPH-induced lipid peroxidation in Vero cells. Vero cells were pretreated with CAAP peptide (12.5, 25, 50, and 100 µg/mL) for 2 h, followed by exposure to AAPH (10 mM) for 24 h. (A) Representative fluorescence images of DPPP-stained cells showing attenuation of AAPH-induced lipid peroxidation (blue fluorescence) by CAAP treatment. (B) Quantitative analysis of malondialdehyde (MDA) levels using the thiobarbituric acid reactive substances (TBARS) assay. Data are expressed as mean ± SEM (n = 4); #p < 0.05 vs. Control; *p < 0.05, **p < 0.01, ***p < 0.001 vs. 0 (AAPH-treated group).
To further elucidate whether the cytoprotective action of CAAP extends beyond its direct radical scavenging capacity, apoptosis-related parameters were examined. Hoechst 33342 and propidium iodide (PI) dual staining revealed that AAPH exposure induced pronounced nuclear condensation and membrane rupture-typical features of apoptotic and necrotic cell death (Fig. 2A). In contrast, CAAP treatment markedly mitigated these morphological abnormalities. Flow cytometric analysis demonstrated that the sub-G1 fraction increased to approximately 31.7% following AAPH stimulation but was reduced to 8.4% after treatment with 50 μg/mL CAAP (Fig. 2B). Consistently, western blot analysis showed that CAAP upregulated the anti-apoptotic Bcl-xL while downregulating pro-apoptotic Bax and cleaved caspase-3 (Fig. 3). This modulation of the Bcl-xL/Bax ratio likely contributes to the stabilization of mitochondrial membrane potential, thereby preventing cytochrome c release and subsequent caspase-3—dependent DNA fragmentation.35 Mitochondrial dysfunction induced by oxidative stress is a critical determinant of endothelial apoptosis and vascular rarefaction, processes that promote microcirculatory failure and elevate peripheral resistance.4,36,37 Hence, preservation of mitochondrial integrity by CAAP may attenuate early endothelial injury that predisposes to hypertension and ischemic cardiovascular diseases. Similar mitochondrial-protective and anti-apoptotic effects have been reported for marine fish-derived peptides, such as tilapia oligopeptide and pufferfish peptide, which reduced ROS accumulation, suppressed Bax/caspase-3 activation, and enhanced Bcl-2 or Bcl-xL expression in oxidative stress-challenged endothelial cells.32,38
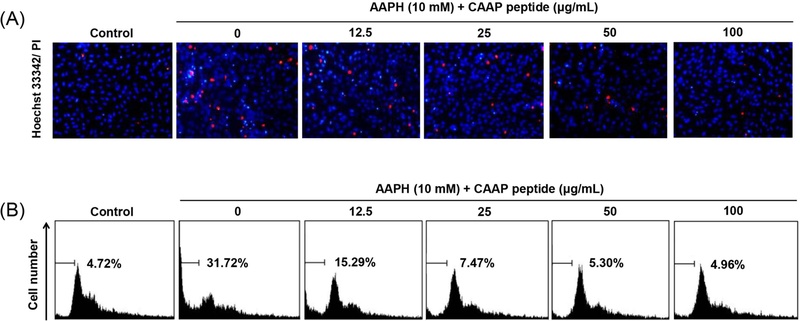
CAAP peptide protects Vero cells from AAPH-induced apoptosis and necrosis. Vero cells were treated with AAPH (10mM) for 24 h in the absence or presence of CAAP (12.5–100 µg/mL). (A) Representative fluorescence images of Hoechst 33342 (blue) and propidium iodide (PI, red) dual staining showing that CAAP markedly reduced nuclear condensation and membrane rupture caused by AAPH exposure. (B) Flow cytometric analysis of sub-G1 hypodiploid cell populations demonstrating the anti-apoptotic effect of CAAP.
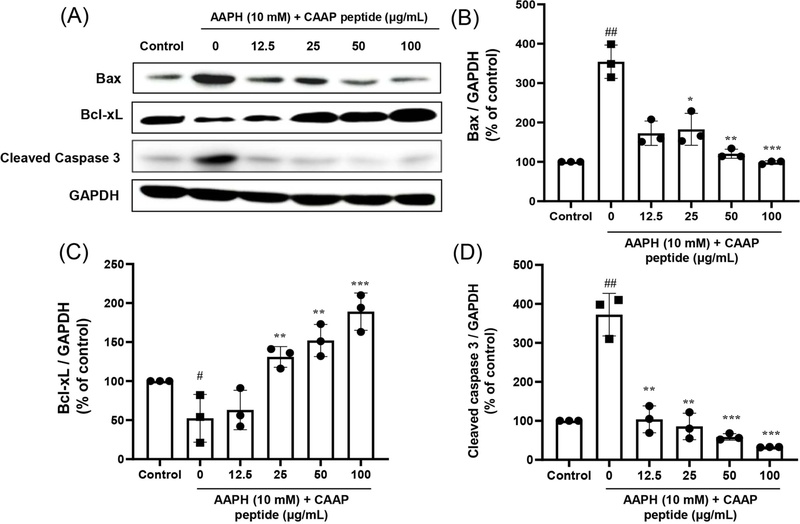
CAAP peptide regulates the expression of apoptosis-related proteins in AAPH-treated Vero cells. Representative immunoblots (A) and quantification (B–D) of Bax, Bcl-xL, and cleaved caspase-3 expression. CAAP increased the anti-apoptotic Bcl-xL level while reducing pro-apoptotic Bax and cleaved caspase-3 expression, restoring the Bcl-xL/Bax ratio and maintaining mitochondrial integrity. Data are expressed as mean ± SEM (n = 4); #p < 0.05, ##p < 0.01 vs. Control; *p < 0.05, **p < 0.01, ***p < 0.001 vs. 0 (AAPH-treated group).
Given the intricate interplay between oxidative stress and the renin–angiotensin system (RAS), we next investigated whether CAAP also modulates angiotensin-converting enzyme (ACE) activity. In vitro ACE inhibition assays demonstrated that CAAP suppressed enzymatic activity in a concentration-dependent manner, with an IC50 value of 39.3 µg/mL. Molecular docking analysis further indicated that CAAP binds favorably within the S′₂ subsite of ACE, establishing multiple hydrogen bonds with key residues such as Glu281, Lys511, His513, and Tyr520 (Fig. 4). These interactions are known to stabilize peptide—ACE complexes and hinder substrate (angiotensin I) access to the catalytic zinc center, thereby reducing angiotensin II generation.39–41 The predicted CDOCKER interaction energy (−52.6 kcal/mol) and binding energy (−183.9 kcal/mol) supports specific affinity, comparable to those reported for food-derived ACE-inhibitory peptides.42–44 From a physiological perspective, excessive ACE activity contributes to vascular dysfunction through angiotensin II-induced vasoconstriction, oxidative stress, and endothelial inflammation—central mechanisms driving hypertension and vascular remodeling.45–47 Hence, the partial inhibition of ACE by CAAP, combined with its antioxidant action, may exert synergistic benefits in preserving vascular homeostasis. Unlike conventional synthetic ACE inhibitors, which primarily target the catalytic site,39 naturally derived peptides such as CAAP possess dual antioxidant and enzyme-modulatory functions that could offer improved vascular protection with reduced adverse effects.12,13,20,21
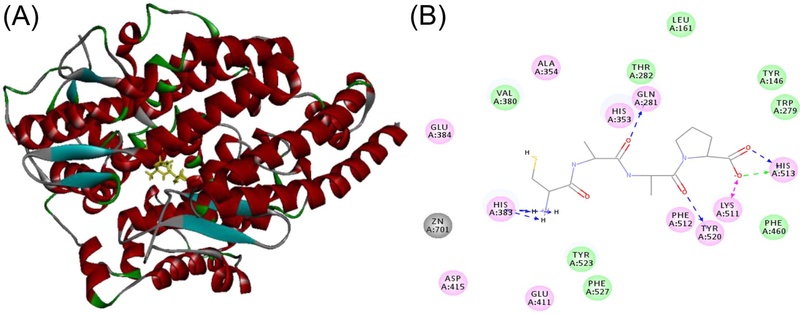
Predicted binding mode of CAAP peptide with angiotensin-converting enzyme (ACE). (A) Three-dimensional structure of the CAAP-ACE complex showing peptide binding within the S′₂ subsite. (B) Two-dimensional diagram illustrating hydrogen bonding interactions between CAAP and key ACE residues (Glu 281, Lys 511, His 513, and Tyr 520). The calculated CDOCKER interaction energy (−52.6 kcal/mol) and binding energy (−183.9 kcal/mol) indicate strong affinity of CAAP for the ACE active site.
In vivo studies using spontaneously hypertensive rats (SHRs) substantiated CAAP’s antihypertensive potential. Oral administration of CAAP (40 mg/kg) significantly decreased systolic blood pressure (SBP) relative to saline-treated controls (Fig. 5). The decline in SBP became evident 3 h after administration, reached its maximal reduction of -167 mmHg at 6 h, and remained significant reduction until 9 h, following a pattern similar to captopril. This blood pressure–lowering effect suggests a complementary mechanism wherein CAAP’s antioxidant and ACE-inhibitory activities act synergistically to restore vascular homeostasis. By scavenging ROS, CAAP may enhance endothelial nitric oxide synthase (eNOS) coupling, thereby improving NO-dependent vasodilation, while ACE inhibition reduces angiotensin II generation, decreasing vasoconstrictor tone. Such integrative modulation of the oxidative and renin–angiotensin axes represent a hallmark of effective vascular protection.48,49 Similar multi-target actions have been documented for short-chain peptides derived from foods, which achieve sustained antihypertensive effects through parallel redox and enzymatic regulation.12,50
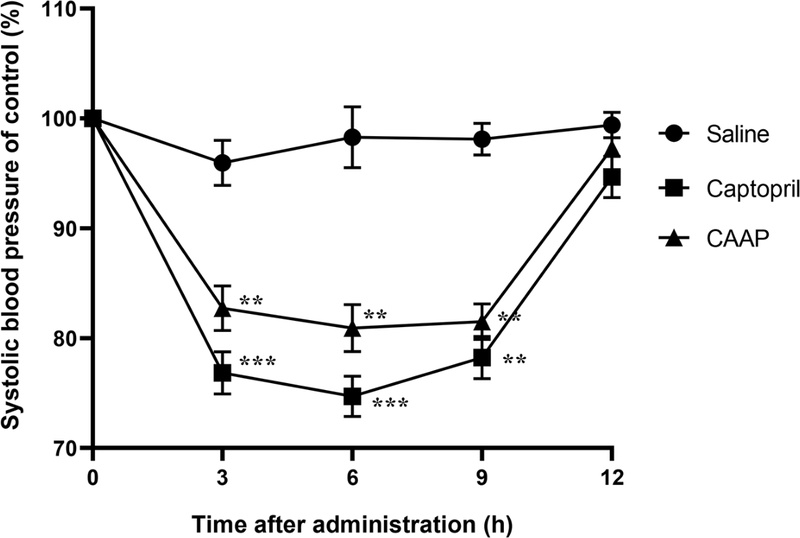
Antihypertensive effect of CAAP peptide in spontaneously hypertensive rats (SHRs). SHRs were orally administered saline (control) captopril (40mg/kg), or CAAP peptide (40mg/kg). Systolic blood pressure (SBP) was measured using a non-invasive tail-cuff system at 0, 3, 6, 9, and 12 h post-administration. CAAP significantly reduced SBP, with the maximal decrease observed at 6 h, comparable to captopril. Data are expressed as mean ± SEM (n = 4). **p < 0.01, ***p < 0.001 vs. control.
The results of this study collectively demonstrate that CAAP is a dual-action marine peptide with potent antioxidant and ACE-inhibitory properties, possessing cardioprotective potential. In the pathogenesis of hypertension and atherosclerosis, oxidative stress provokes endothelial apoptosis and vascular stiffening, whereas excessive activation of the RAS perpetuates inflammation and elevates blood pressure.51,52 By neutralizaing ROS and simultaneously inhibiting ACE activity, CAAP effectively targets these two interrelated pathogenic mechanisms that underlie cardiovascular dysfunction. From a translational perspective, such multifunctional peptides, characterized by their low molecular weight, high solubility, and minimal toxicity, represent attractive candidates for the development of nutraceuticals or as adjuncts to current antihypertensive therapies. Moreover, the structural simplicity of CAAP is likely to favor intestinal absorption and metabolic stability, key determinants of oral bioavailability. Nevertheless, further pharmacokinetic, mechanistic, and In vivo efficacy studies are needed to confirm its systemic distribution, molecular target, and long-term vascular benefits.
In conclusion, the flounder-derived tetrapeptide CAAP (Cys-Ala-Ala-Pro) was identified as a dual-acting marine bioactive peptide exhibiting antioxidant and antihypertensive activities relevant to cardiovascular protection. CAAP inhibited lipid peroxidation, preserved mitochondrial integrity, and suppressed ACE activity through interactions within the S′₂ subsite. Oral administration of CAAP significantly lowered SBP in SHRs, indicating synergistic effects of its antioxidant and ACE-inhibitory action in restoring vascular homeostasis. By targeting oxidative stress and RAS overactivation, CAAP effectively addresses two interrelated mechanisms of vascular dysfunction. This pharmacological multifunctionality highlights the potential of Paralichthys olivaceus peptides as natural scaffolds for developing functional foods or peptide-based antihypertensive agents. Further studies on bioavailability, molecular targets, and long-term efficacy will clarify CAAP’s potential in cardiovascular disease prevention and management.
Acknowledgments
This research was supported by Glocal University Project of Mokpo National University in 2025 and was supported by Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (RS-2022-KS221671).
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Roth, G. A.; Mensah, G. A.; Johnson, C. O.; Addolorato, G.; Ammirati, E.; Baddour, L. M.; Barengo, N. C.; Beaton, A. Z.; Benjamin, E. J.; Benziger, C. P.; Bonny, A.; Brauer, M.; Brodmann, M.; Cahill, T. J.; Carapetis, J.; Catapano, A. L.; Chugh, S. S.; Cooper, L. T.; Coresh, J.; Criqui, M.; DeCleene, N.; Eagle, K. A.; Emmons-Bell, S.; Feigin, V. L.; Fernández-Solà, J.; Fowkes, G.; Gakidou, E.; Grundy, S. M.; He, F. J.; Howard, G.; Hu, F.; Inker, L.; Karthikeyan, G.; Kassebaum, N.; Koroshetz, W.; Lavie, C.; Lloyd-Jones, D.; Lu, H. S.; Mirijello, A.; Misganaw Temesgen, A.; Mokdad, A.; Moran, A. E.; Muntner, P.; Narula, J.; Neal, B.; Ntsekhe, M.; Moraes de Oliveira, G.; Otto, C.; Owolabi, M.; Pratt, M.; Rajagopalan, S.; Reitsma, M.; Ribeiro, A. L. P.; Rigotti, N.; Rodgers, A.; Sable, C.; Shakil, S.; Sliwa-Hahnle, K.; Stark, B.; Sundström, J.; Timpel, P.; Tleyjeh, I. M.; Valgimigli, M.; Vos, T.; Whelton, P. K.; Yacoub, M.; Zuhlke, L.; Murray, C.; Fuster, V. J. Am. Coll. Cardiol. 2020, 76, 2982–3021.
[https://doi.org/10.1016/j.jacc.2020.11.010]

-
Madamanchi, N. R.; Runge, M. S. Free Radic. Biol. Med. 2013, 61, 473–501.
[https://doi.org/10.1016/j.freeradbiomed.2013.04.001]

-
Taniyama, Y.; Griendling, K. K. Hypertension 2003, 42, 1075–1081.
[https://doi.org/10.1161/01.HYP.0000100443.09293.4F]

-
Forrester, S. J.; Kikuchi, D. S.; Hernandes, M. S.; Xu, Q.; Griendling, K. K. Circ. Res. 2018, 122, 877–902.
[https://doi.org/10.1161/CIRCRESAHA.117.311401]

-
Niki, E.; Yoshida, Y.; Saito, Y.; Noguchi, N. Biochem. Biophys. Res. Commun. 2005, 338, 668–676.
[https://doi.org/10.1016/j.bbrc.2005.08.072]

-
Fraga, C. G.; Leibovitz, B. E.; Tappel, A. L. Free Radic. Biol. Med. 1988, 4, 155–161.
[https://doi.org/10.1016/0891-5849(88)90023-8]

-
Zheng, Y.; Sun, J.; Luo, Z.; Li, Y.; Huang, Y. Cell Death Dis. 2024, 15, 859.
[https://doi.org/10.1038/s41419-024-07244-x]

-
Hennig, B.; Chow, C. K. Free Radic. Biol. Med. 1988, 4, 99–106.
[https://doi.org/10.1016/0891-5849(88)90070-6]

-
Ajoolabady, A.; Pratico, D.; Ren, J. Mol. Cell. Endocrinol. 2024, 592, 112309.
[https://doi.org/10.1016/j.mce.2024.112309]

-
Fanelli, C.; Zatz, R. Hypertension 2011, 57, 373–374.
[https://doi.org/10.1161/HYPERTENSIONAHA.110.167775]

-
Reid, I. O. A.; Osman, S. M.; Bakheet, S. M. J. Drug Des. Med. Chem. 2025, 11, 13–30.
[https://doi.org/10.11648/j.jddmc.20251102.11]

-
Suo, S.-K.; Zheng, S.-L.; Chi, C.-F.; Luo, H.-Y.; Wang, B. Front. Nutr. 2022, 9, 957778.
[https://doi.org/10.3389/fnut.2022.957778]

-
Zhuang, X.; Min, D.; Jiang, H.; Zeng, W. Future Foods 2025, 11, 100648.
[https://doi.org/10.1016/j.fufo.2025.100648]

-
Peighambardoust, S. H.; karami, Z.; Pateiro, M.; Lorenzo, J. M. Biomolecules 2021, 11, 631.
[https://doi.org/10.3390/biom11050631]

- Sarker, A. J. Food Process. Preserv. 2022, 46, 16800.
-
Ahmed, T.; Sun, X.; Udenigwe, C. C. Trends Food Sci. Technol. 2022, 120, 265–273.
[https://doi.org/10.1016/j.tifs.2022.01.008]

-
Masuda, O.; Nakamura, Y.; Takano, T. J. Nutr. 1996, 126, 3063–3068.
[https://doi.org/10.1093/jn/126.12.3063]

-
Turpeinen, A. M.; Ehlers, P. I.; Kivimäki, A. S.; Järvenpää, S.; Filler, I.; Wiegert, E.; Jähnchen, E.; Vapaatalo, H.; Korpela, R.; Wagner, F. Clin. Exp. Hypertens. 2011, 33, 388−396.
[https://doi.org/10.3109/10641963.2010.549267]

-
Martínez-Montaño, E.; Sarmiento-Machado, R. M.; Osuna-Ruíz, I.; Benítez-García, I.; Pacheco-Aguilar, R.; Navarro-Peraza, R. S.; Sánchez, M. E. L.; Ortiz, A. V.; Báez, L. J. G.; Bañuelos-Vargas, I.; Rodríguez-Tirado, V. A.; Salazar-Leyva, J. A. Waste Biomass Valorization. 2022, 13, 1015−1027.
[https://doi.org/10.1007/s12649-021-01590-z]

-
Perez-Escalante, E.; Padilla-Zuniga, S. A.; Contreras-Lopez, E.; Sebastian-Nicolas, J. L.; Perez-Flores, J. G.; Olloqui, E. J.; Gonzalez- Olivares, L. G. Biol. Life Sci. Forum. 2022, 18, 55.
[https://doi.org/10.3390/Foods2022-12991]

-
Ribeiro, M. L.; Kefner, A. C. S.; Carvalho, A. L. O.; Magalhães, A. V.; Zingali, R. B.; Cicilini, M. A.; Santos, A. M. C. Food Bioprocess Technol. 2025, 18, 1229−1243.
[https://doi.org/10.1007/s11947-024-03513-x]

-
Shahidi, F.; Zhong, Y. J. AOAC Int. 2008, 91, 914−931.
[https://doi.org/10.1093/jaoac/91.4.914]

-
Fujita, H.; Yoshikawa, M. Immunopharmacology 1999, 44, 123−127.
[https://doi.org/10.1016/S0162-3109(99)00118-6]

-
Ko, J.-Y.; Lee, J.-H.; Samarakoon, K.; Kim, J.-S.; Jeon, Y.-J. Food Chem. Toxicol. 2013, 52, 113−120.
[https://doi.org/10.1016/j.fct.2012.10.058]

-
Ko, J.-Y.; Kim, E.-A.; Lee, J.-H.; Kang, M.-C.; Lee, J.-S.; Kim, J.- S.; Jung, W.-K.; Jeon, Y.-J. Fish Shellfish Immunol. 2014, 36, 320−323.
[https://doi.org/10.1016/j.fsi.2013.11.018]

-
Nicoletti, I.; Migliorati, G.; Pagliacci, M. C.; Grignani, F.; Riccardi, C. J. Immunol. Methods 1991, 139, 271−279.
[https://doi.org/10.1016/0022-1759(91)90198-O]

-
Cushman, D. W.; Cheung, H. S. Biochem. Pharmacol. 1971, 20, 1637−1648.
[https://doi.org/10.1016/0006-2952(71)90292-9]

-
Scioli, M. G.; Storti, G.; D’Amico, F.; Guzmán, R. R.; Centofanti, F.; Doldo, E.; Miranda, E. M. C.; Orlandi, A. J. Clin. Med. 2020, 9, 1995.
[https://doi.org/10.3390/jcm9061995]

-
Ulrich, K.; Jakob, U. Free Radic. Biol. Med. 2019, 140, 14−27.
[https://doi.org/10.1016/j.freeradbiomed.2019.05.035]

-
Pace, M.; Giorgi, C.; Lombardozzi, G.; Cimini, A.; Castelli, V.; d’Angelo, M. Biomolecules 2025, 15, 1115.
[https://doi.org/10.3390/biom15081115]

-
Ngo, D.-H.; Kang, K.-H.; Jung, W.-K.; Byun, H.-G.; Kim, S.-K. J. Funct. Food. 2014, 10, 243−251.
[https://doi.org/10.1016/j.jff.2014.06.021]

-
Chen, J.; Gong, F.; Chen, M.-F.; Li, C.; Hong, P.; Sun, S.; Zhou, C.; Qian, Z.-J. Mar. Drugs 2019, 17, 431.
[https://doi.org/10.3390/md17070431]

-
Wang, Y.-M.; Li, X.-Y.; Wang, J.; He, Y.; Chi, C.-F.; Wang, B. Process Biochem. 2022, 113, 258−269.
[https://doi.org/10.1016/j.procbio.2022.01.008]

-
Liu, Y.; Huang, H.; Chen, Y.; Ryu, B.; Qian, Z.-J.; J. Funct. Food. 2025, 133, 107009.
[https://doi.org/10.1016/j.jff.2025.107009]

-
He, L.; Perkins, G. A.; Poblenz, A. T.; Harris, J. B.; Hung, M.; Ellisman, M. H.; Fox, D. A. Proc. Natl. Acad. Sci. U. S. A. 2003, 100, 1022−1027.
[https://doi.org/10.1073/pnas.0333594100]

-
Zheng, D.; Liu, J.; Piao, H.; Zhu, Z.; Wei, R.; Liu, K. Front. Immunol. 2022, 13, 1039241.
[https://doi.org/10.3389/fimmu.2022.1039241]

-
Kobayashi, N.; DeLano, F. A.; Schmid-Schönbein, G. W. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2114−2121.
[https://doi.org/10.1161/01.ATV.0000178993.13222.f2]

-
Cai, S.; Pan, N.; Xu, M.; Su, Y.; Qiao, K.; Chen, B.; Zheng, B.; Xiao, M.; Liu, Z. Mar. Drugs 2021, 19, 655.
[https://doi.org/10.3390/md19120655]

-
Natesh, R.; Schwager, S. L. U.; Sturrock, E. D.; Acharya, K. R. Nature 2003, 421, 551–554.
[https://doi.org/10.1038/nature01370]

-
Acharya, K. R.; Sturrock, E. D.; Riordan, J. F.; Ehlers, M. R. W. Nat. Rev. Drug Discov. 2003, 2, 891–902.
[https://doi.org/10.1038/nrd1227]

-
Chen, H.; Chen, Y.; Zheng, H.; Xiang, X.; Xu, L. Front. Nutr. 2022, 9, 981163.
[https://doi.org/10.3389/fnut.2022.981163]

-
Yang, D.; Li, L.; Li, C.; Chen, S.; Deng, J.; Yang, S. Front. Nutr. 2022, 9, 920945.
[https://doi.org/10.3389/fnut.2022.920945]

-
Ko, J.-Y.; Lee, J.-H.; Sim, H.-H.; Kim, H. J.; Oak, M.-H. Nat. Prod. Sci. 2023, 29, 337–348.
[https://doi.org/10.20307/nps.2023.29.4.337]

-
Zhao, W.; Zhang, D.; Yu, Z.; Ding, L.; Liu, J. J. Funct. Food. 2020, 64, 103649.
[https://doi.org/10.1016/j.jff.2019.103649]

-
Rajagopalan, S.; Kurz, S.; Munzel, T.; Tarpey, M.; Freeman, B. A.; Griendling, K. K.; Harrison, D. G. J. Clin. Invest. 1996, 97, 1916–1923.
[https://doi.org/10.1172/JCI118623]

-
Montezano, A. C.; Touyz, R. M. Ann. Med. 2012, 44, S2–S16.
[https://doi.org/10.3109/07853890.2011.653393]

-
Forrester, S. J.; Booz, G. W.; Sigmund, C. D.; Coffman, T. M.; Kawai, T.; Rizzo, V.; Scalia, R.; Eguchi, S. Physiol. Rev. 2018, 98, 1627–1738.
[https://doi.org/10.1152/physrev.00038.2017]

-
Pacurari, M.; Kafoury, R.; Tchounwou, P. B.; Ndebele, K. Int. J. Inflamm. 2014, 2014, 689360.
[https://doi.org/10.1155/2014/689360]

-
Dikalov, S.; Nazarewicz, R. R. Antioxid. Redox Signal. 2013, 19, 1085–1094.
[https://doi.org/10.1089/ars.2012.4604]

-
Majumder, K.; Chakrabarti, S.; Morton, J. S.; Panahi, S.; Kaufman, S.; Davidge, S. T.; Wu, J. PLoS One 2013, 8, e82829.
[https://doi.org/10.1371/journal.pone.0082829]

-
Guzik, T. J.; Touyz, R. M. Hypertension 2017, 70, 660–667.
[https://doi.org/10.1161/HYPERTENSIONAHA.117.07802]

-
Jia, G.; Aroor, A. R.; Hill, M. A.; Sowers, J. R. Hypertension 2018, 72, 537–548.
[https://doi.org/10.1161/HYPERTENSIONAHA.118.11065]
