
Comparative Extraction Using Steam and Simultaneous Extraction Techniques, Chemical Profiling, and Antibacterial Activity of Essential Oil from Tagetes erecta and Tagetes patula
Abstract
The extraction of essential oil can be done in several ways, with some emphasizing quality and others emphasizing quantity. Essential oil components of Tagetes erecta and Tagetes patula were analyzed and compared using two common extraction techniques, i.e., steam distillation (SD) and simultaneous distillation extraction (SDE), coupled with GC-MS for their chemical composition and antimicrobial activity. In both species, the highest % yield was obtained by the SD with yields of 0.05% and 0.12%, respectively. Thus, the comparative study of extraction methods unveiled SD as the best method for the extraction of essential oil, providing a high yield with a greater percentage of major compounds. The GC-MS analysis unveiled 24 different compounds in T. erecta and 27 compounds in T. patula. SD of T. erecta and T. patula revealed a higher concentration of β-Caryophyllene with compositions of 37.25% and 65.47%. A comparative analysis of the antibacterial activity of essential oil extracted using SD revealed that T. erecta is effective against all four tested bacterial strains, i.e., Enterococcus faecalis, Staphylococcus aureus, Klebsiella pneumoniae and Salmonella Typhi. Notably, the largest zone of inhibition (ZOI) was observed against K. pneumoniae (12 mm) for T. erecta. In contrast, T. patula demonstrated activity against two bacterial strains, i.e., K. pneumoniae and S. typhi, with a maximum ZOI of 11 mm, observed against K. pneumoniae. The stronger antibacterial activity of T. erecta oil might be due to the synergistic interaction of the most abundant bioactive compounds obtained through SD.
Keywords:
Tagetes, Marigold, Steam distillation, Simultaneous distillation extraction, GC-MS, Antimicrobial activityIntroduction
Tagetes is an important genus of the Asteraceae family, also referred to as Marigold and is originally found in Mexico and Guatemala, although it is currently grown all over the globe due to its decorative use.1 Out of 55 species of Tagetes, T. patula L. (French marigold) and T. erecta L. (African marigold) are commonly available species in Nepal.2 In Nepal, Tagetes flowers are mostly cultivated for decorative and religious duties. However, the use of Tagetes extends beyond ornamentation; it holds significant importance in folk medicine for curing various conditions, including wounds, fever, scabies, digestive and nervous disorders, dental problems, skin infections, hepatic abnormalities, and ulcers.3,4Tagetes is an important source of lutein ester, a nutrient vital for eye health as well as guarding against age related muscular degeneration.5,6 The essential oil of T. erecta contains different bioactive chemicals, like β-caryophyllene, piperitone, and piperitenone.7 At the same time, the major compounds found in T. patula are piperitone, terpinolene, and β-caryophyllene, which exhibit anti-inflammatory, anticancer, insecticidal, antioxidant, and antibacterial properties.4,8,9 Essential oil of T. erecta and T. patula benefit is not only limited to the therapeutic agent but also have great significance in the cosmetic and perfume industry as a flavoring agent.10 Major chemical constituents in Tagetes include tagetin, polyphenols, quercetin, quercetagetin, tannins, steroids, flavonoids, and saponins.10,11 β-caryophyllene is widely known to reduce acute and chronic pain through endo-cannabinoid and opioid systems and shows neuroprotective, cardioprotective, cytotoxic, and hypolipidemic activities as well.12
Despite their wide application, scope, and use, the quality and quantity of essential oils extracted from these species are low enough to compete in a global commercialized market. To make its essential oil competitive in the global market, it is crucial to increase the chemical content by choosing the right extraction process. Hence elaborate study is necessary as extraction techniques, geographic area, cultivation process, storage, plant species, and other factors can have an impact on the yield and composition of the essential oil. The choice of an effective extraction technique can significantly improve the quality and composition of the essential oil.13 Various extraction techniques are utilized to extract essential oil. However, the most widely used techniques are steam distillation (SD) and simultaneous distillation extraction (SDE). Both SD and SDE are the common and popular methods for isolating volatile compounds from plant materials.14,15
In the past, SD was widely used for extraction due to its environmentally friendly and reduced operational cost, as it used water as a solvent. However, water substantially diluted the volatile component, which was conquered by the SDE, where an organic solvent is used.13,16–21 Each method has its advantages as well as disadvantages; therefore, the obtained oil was analyzed using the GC-MS technique to enhance the quality and quantity of essential oil. A systematic comparison of steam distillation (SD) and simultaneous distillation extraction (SDE) for Tagetes species remains unexplored, particularly regarding the resulting chemical profiles and their corresponding biological activities. Such an investigation is critical for establishing an optimized, standardized extraction protocol that maximizes both yield and the concentration of key bioactive constituents, thereby enhancing the commercial viability and therapeutic relevance of Tagetes essential oils.
This study provides a systematic evaluation of steam distillation and simultaneous distillation extraction to determine their efficiency in optimizing both the yield and the bioactive quality of Tagetes essential oils. While SD proved more effective for overall yield, SDE with specific solvents enabled targeted recovery of individual compounds. The novelty of this work lies in its integrated approach directly linking extraction outcomes to antibacterial efficacy, thereby establishing an evidence-based framework for selecting methods based on application-specific goals, such as maximizing yield, enriching particular constituents, or enhancing bioactivity.
Experimental
Chemical – Hexane, dichloromethane (DCM), diethyl ether, ultra-pure water from the Milli-Q water purification system, and acetone. The solvents were selected based on three criteria: common application in volatile compound isolation, a range of polarities to compare extraction efficiency, and immiscibility with water. All the chemicals used were of analytical grade.
Plant collection and sample preparation – The plant specimens (Flowers) of T. erecta L. and T. patula L. were collected from Tansen, Palpa, located at an altitude of 1350 m above sea level (msl) (27° 52’ 2.32” N latitude and 83° 32’ 48.12” E longitude). The collected samples were identified by the botanist at the Central Department of Chemistry, Kritipur, Nepal. The accession codes for these specimens are Palpa 101 and Palpa 201, respectively. The samples were shade-dried for two weeks to achieve a moisture content of less than 10%, ground into a powder, and kept in an airtight container for further use.
Extraction of essential oil – The essential oil from two species of Tagetes was extracted using two distinct techniques, SD and SDE, following the reported protocol.19,22 In the process of extracting hexane, diethyl ether, and DCM were used as solvents.
Steam distillation – 200 g of Tagetes flower (T. erecta and T. patula) were kept in a perforated flask positioned above a round-bottom flask containing 2.5 litres of milli-Q water, and the condenser was connected to the other open end. The apparatus was heated to boil the water, initially reaching the boiling temperature at 100℃, and maintained for 4 hours. The steam formed travels through the sample into the condenser, where it condenses along with the essential oil. The resulting oil and water mixture was collected in a specially designed separating vessel. The two layers were then separated based on their density. Thus, collected oil was transferred into vials containing sodium sulfate until further use for GC-MS analysis.
Simultaneous distillation extraction – For SDE, a modified Likens-Nickerson apparatus was employed. A 5 L round-bottom flask containing 200 g of plant material and 2.5 L of Milli-Q water was connected to the left arm. The right arm was connected to a 500 mL round-bottom flask containing 200 mL of organic solvent (solvent-to-sample ratio of 1:1 v/w). The chilled circulating coolant (isopropanol) was maintained at −5°C. Both flasks were heated independently to their boiling points using heating mantles. The vapours from both flasks met in the central condensation chamber, where they condensed and formed two immiscible layers. The design of the apparatus allowed continuous recirculation of both phases; the aqueous phase returned to the plant flask, and the organic phase returned to the solvent flask, ensuring continuous extraction over the 4 hours. To minimize solvent loss, all joints were securely sealed with PTFE sleeves, the condenser efficiency was verified, and the system was maintained under slight positive pressure. After extraction, the solvent phase was collected, dried over anhydrous sodium sulfate, and concentrated under reduced pressure (40°C) using a rotary evaporator.
GC-MS analysis – The composition of the concentrated essential oils of Tagetes species was analyzed using gas chromatography-mass spectroscopy. An Agilent 7890A GC system (Agilent, USA) equipped with an Agilent HP-5 (5% phenylmethylsiloxane) MS fused silica capillary column (30 m × 250 µm × 0.25 µm) was used to chromatograph the concentrated oil, and an Agilent MS 5975C was used to continue the qualitative examination of the essential oil. The spectrometer was operated in electron impact mode at 70 eV utilizing helium as a carrier gas with a flow rate made constant at 1.0 mL/min, split ratio of 75:1, and head pressure of 6.777 psi.
1 µL of the extracted essential oil was diluted in hexane at a ratio of 1:10 v/v for GC analysis. The column temperature was initially set at 35℃ for 5 minutes and then ramped at 5℃/min to 100℃, 7℃/min to 150℃, and 10℃/min to 205℃ for a total time of 33.64 min. The sample was fed into a GC inlet, maintaining a column flow rate of 1 mL/min and purge flow of 3 mL/min. The ion source temperature was maintained at 220℃ and 235℃ as the contact temperature. Mass spectra were scanned at a speed of 666, from m/z 25–500. The retention indices were calculated by using a standard hydrocarbons mixture (C5-C32) from LGC group Dr Ehrenstorfer, United States. Lastly, by comparing retention indices and mass spectral fragmentation patterns (with over 90% similarity index). The constituent present in the essential oil was identified by cross-referencing MS with the NIST library database.
Preparation of microbial culture media – 13 g of LB powder (Sisco Research Laboratories Pvt. Ltd., India) were dissolved in one liter of distilled water to create the liquid broth (LB) medium. The mixture was autoclaved for 25 minutes at 121℃ and 15 psi pressure. After cooling to 40–50°C, the sterilized medium was transferred into 15 mL falcon tubes that had been previously sterilized (5 mL each). Bacterial seed was co-cultured in each tube independently using the prepared medium, and the tubes were incubated for a full day.
Preparation of MH media plates – One liter of distilled water was used to dissolve 39 g of Mueller-Hinton (MH) agar powder (Sisco Research Laboratories Pvt. Ltd., India) to create the MH agar plates. The mixture was autoclaved for 25 minutes at 121°C and 15 psi pressure. After cooling to 40–50°C, the sterilized medium was transferred onto 25 mL sterile petri dishes.
Antibacterial assay – The antibacterial activity of the essential oils was evaluated using the agar well diffusion method. Briefly, 100 µL of a freshly prepared bacterial suspension was spread evenly onto the surface of Mueller-Hinton agar plates. Wells (6 mm in diameter) were punched aseptically into the agar. Subsequently, 100 µL of each essential oil sample, dissolved in dimethyl sulfoxide (DMSO) at a concentration of 100 mg/mL, was introduced into the designated wells. Appropriate controls were included: DMSO and deionized water (DW) served as the negative control, and kanamycin (5 µg/well) served as the positive antibiotic control. All assays were performed in triplicate (n = 3). The plates were incubated at 37°C for 24 h, after which the zones of inhibition (ZOI) were measured in millimetres.
Results and Discussion
Variations in the percentage yield of essential oil were obtained from SD and SDE for two species of Tagetes, as shown in Table 1. For both species, SD offers higher yields, with 0.05% for T. erecta and 0.12% for T. patula. However, in SDE, dichloromethane offers a higher yield compared to the other two solvents. Similarly for T. patula. Diethyl ether offers a higher yield of essential oil.
The yellow-colored oil with different % yields was obtained from T. erecta. The GC-MS technique was used for the identification of chemical constituents by comparing the MS using the NIST library. A total of 24 compounds were identified in four different essential oils (Table 2). However, the percentage composition varied with the technique and solvent used. In SD, β-caryophyllene (37.25%) and germacrene D (19.17%) were identified as major compounds, whereas in SDE, in all three solvents, β-caryophyllene (20.94% average) stood out as a major compound. Out of the three solvents used in SDE, DCM and diethyl ether showed a higher yield of β-caryophyllene (21.96%). Also, the higher yield of β-linalool (23.75%) and piperitone (6.09%) was obtained using DCM as a solvent. Similarly, α-terpineol (7.46%), caryophyllene oxide (9.02%), and Z-geraniol (6.59%) were in higher amounts when diethyl ether was used as a solvent, as shown in Fig. S1.
Chemical profiling of T. erecta flower essential oil obtained from steam distillation and simultaneous distillation extraction using GC-MS
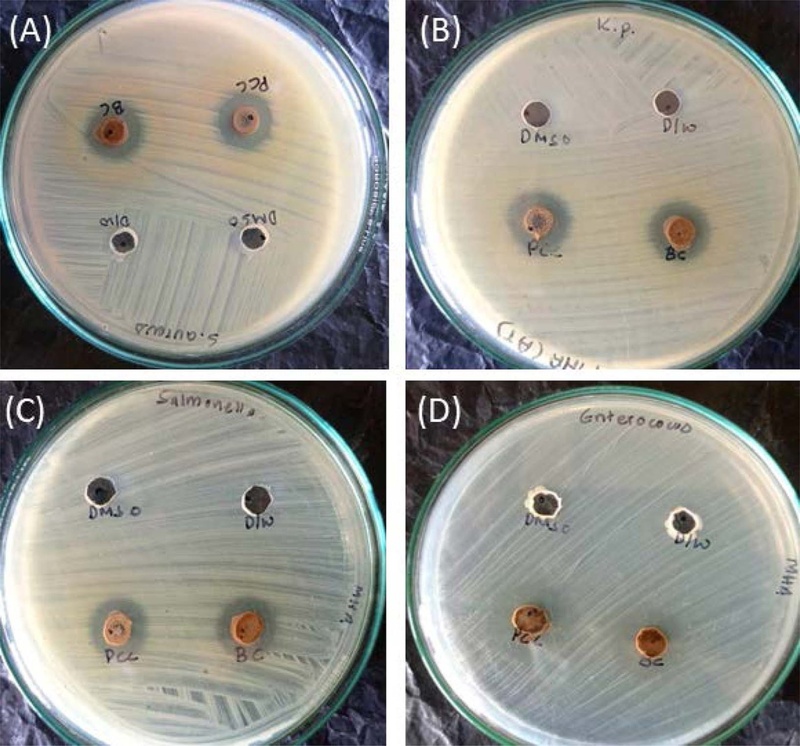
Antimicrobial activity of the T. erecta and T. patula against (A) S. aureus, (B) K. pneumoniae, (C) S. typhi, and (D) E. faecalis.
GC-MS showed that the yellow-colored essential oil of T. patula obtained from two extraction techniques consists of 27 compounds (Table 3). The present study showed that more major compounds can be extracted using the SD. Compounds like β-caryophyllene (65.47%), β-cubebene (9.35%), caryophyllene oxide (6.86%), and (Z)-β-farnesene (4.09%) were obtained at higher yields when SD was used for oil extraction. The identified compounds were cross-checked with the standard retention indices of the NIST Library database Fig. S1.

Chemical profiling of T. patula flower essential oil obtained from steam distillation and simultaneous distillation extraction using GC-MS
The essential oil obtained from steam distillation was evaluated for its antibacterial activity against two gram-positive bacteria, S. aureus and E. faecalis and two gram-negative bacteria, K. pneumoniae, and S. typhi, as shown in Fig. 1. PCC represents the T. erecta, and BC represents the T. patula. The antibacterial efficacy, measured by the zone of inhibition, is summarized in Table 4. T. erecta exhibited the 11.0 mm and 0.0 mm ZOI against S. aureus and E. faecalis, whereas T. patula demonstrated 10.0 mm and 0.0 mm, respectively. Against gram-negative bacteria, T. erecta exhibits a zone of inhibition of 12.0 mm and 11.0 mm for K. pneumoniae and S. typhi, respectively. In comparison, T. patula exhibits a zone of inhibition of 9.0 mm and 10.0 mm for the same bacteria.
The present study demonstrates that the chemical composition and yield of essential oils from T. erecta and T. patula are significantly influenced by the extraction technique, with SD proving superior for overall yield and recovery of key terpenoids such as β-caryophyllene. This aligns with earlier findings that SD better preserves thermally stable, non-polar volatiles compared to solvent-based methods, which may selectively extract or alter certain compounds due to solvent polarity and boiling point effects.13,14 In our study, SD of T. erecta and T. patula yielded 0.05% and 0.12% oil, respectively, which is consistent with yields reported for other Tagetes species using hydro distillation.8,23 However, the notably high β-caryophyllene content in SD extracts (37.25% in T. erecta, 65.47% in T. patula) exceeds levels commonly reported in literature for these species,7,24 possibly due to genotypic variation, environmental factors, or the avoidance of solvent-induced compound loss.
From T. erecta and T. patula, a total of 24 and 27 chemical constituents were discovered. T. erecta contained significant amounts of β-caryophyllene, β-linalool, and germacrene D, while T. patula included significant amounts of β-caryophyllene, β-cubebene, and caryophyllene oxide. Diethyl ether can be used as a solvent in SDE to extract large amounts of caryophyllene oxide for both species. DCM solvent was thought to be the most effective for extracting piperitone from T. erecta. However, it is less prevalent in T. patula, and hexane was considered the most effective solvent for its extraction. It is interesting to note that, although using solvent-based extraction might be preferential to extracting some oxygenated monoterpenes, it has been indicated in a study25 that using hydrodistillation, high yields of piperitone and piperitenone were obtained using T. erecta. This ostensible inconsistency can be explained by the fact that plant chemotype, growing environment, or minor differences in distillation parameters may also cause it, and it shows how the results of extractions can be affected by both methodological and biological factors.
While the essential oils of T. erecta and T. patula share key compounds such as β-caryophyllene, their profiles exhibit significant interspecific variation. T. erecta oil was distinguished by the presence of germacrene D and higher yields of β-linalool, whereas T. patula oil contained greater proportions of caryophyllene oxide and (Z)-β-farnesene. Notably, piperitenone, a compound reported in prior studies of T. patula, was absent in our extracts, underscoring the impact of chemotypic variation. While the major compounds identified here align with previous literature, their relative abundances differ considerably, a divergence likely attributable to a combination of genetic factors, environmental conditions, and extraction methodology. The prevalence of β-caryophyllene in both oils is of particular relevance, given its well-documented bioactivity, including antimicrobial, anti-inflammatory, and neuroprotective properties mediated through pathways such as CB2 receptor interaction.26,27
The higher antibacterial activity of T. erecta oil, especially against K. pneumoniae, can be correlated with its unique chemical profile. The SD oils of both species were rich in β-caryophyllene, while T. erecta oil included a wider range of significant components such as piperitone and germacrene D, which were absent or minimal in T. patula. β-Caryophyllene is a well-known antimicrobial sesquiterpene capable of disrupting bacterial membranes and potentiating the activity of other agents. Its high concentration provides a baseline of activity in both oils. However, the broader-spectrum efficacy of T. erecta oil suggests a possible synergistic interaction.28–29 For example, the combination of β-caryophyllene with germacrene D and oxygenated monoterpenes like piperitone may create a multi-target effect, compromising bacterial cell integrity, efflux pumps, and enzymatic functions more effectively than β-caryophyllene alone.30 This aligns with previous studies on Tagetes oils, where antimicrobial potency was attributed to the combined activity of the terpenoid ensemble rather than a single compound.29,31 The absence of piperitenone in our samples further emphasizes how chemotypic diversity and extraction technique influence not only composition but also biological activity, which contrasts with several reports highlighting piperitenone as a key antibacterial agent in T. patula.12 Consequently, the enhanced activity of T. erecta oil is likely the result of a specific, synergistic compositional matrix optimally produced by steam distillation, rather than the concentration of a single component.
It is important to note that this study employed an agar well diffusion assay, which provides an initial indication of antibacterial potential through zone of inhibition (ZOI) measurements. While the ZOI data clearly differentiate the activity profiles of the two oils, the quantitative biological relevance cannot be fully ascertained without determining Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) values. Future work should include broth dilution assays to establish MIC/MBC and further explore the mechanisms of synergy among the bioactive constituents.
In conclusion, this comparative study demonstrates that while steam distillation (SD) is optimal for maximizing overall yield and producing essential oils with broad-spectrum antibacterial activity, simultaneous distillation extraction (SDE) provides superior selectivity for enriching specific target compounds. Therefore, the choice of extraction method depends on the intended application; SD is recommended for maximizing total oil recovery and bioactivity, whereas SDE is better suited for targeted compound isolation. These findings offer practical guidance for selecting extraction protocols based on desired outcomes. Future studies incorporating statistical validation, MIC/MBC determinations, and expanded bioactivity assessments will further clarify the therapeutic potential of Tagetes essential oils.
Acknowledgments
The authors are thankful to the Central Department of Chemistry and Nepal Academy of Science and Technology for providing the necessary facilities to carry out this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
-
Salehi, B.; Valussi, M.; Morais-Braga, M. F. B.; Carneiro, J. N. P.; Leal, A. L. A. B.; Coutinho, H. D. M.; Vitalini, S.; Kręgiel, D.; Antolak, H.; Sharifi-Rad, M.; Silva, N. C. C.; Yousaf, Z.; Martorell, M.; Iriti, M.; Carradori, S.; Sharifi-Rad, J. Molecules2018, 23, 2847.
[https://doi.org/10.3390/molecules23112847]

-
Regmi, M.; Ghimire, N.; Subedi, S. J. Inst. Agric. Anim. Sci. 2025, 39, 121–130.
[https://doi.org/10.3126/jiaas.v39i1.88281]

-
Rahman, M. H.; Roy, B.; Chowdhury, G. M.; Hasan, A.; Saimun, M. S. R. Environ. Sustain. 2022, 5, 207–241.
[https://doi.org/10.1007/s42398-022-00230-z]

-
Aati, H. Y.; Emam, M.; Al-Qahtani, J.; Aati, S.; Aati, A.; Wanner, J.; Seif, M. M. Molecules2022, 27, 7242.
[https://doi.org/10.3390/molecules27217242]

-
Khalil, M.; Raila, J.; Ali, M.; Islam, K. M. S.; Schenk, R.; Krause, J.-P.; Schweigert, F. J.; Rawel, H. J. J. Funct. Foods 2012, 4, 602–610.
[https://doi.org/10.1016/j.jff.2012.03.006]

-
Alves-Rodrigues, A.; Shao, A. Toxicol. Lett. 2004, 150, 57–83.
[https://doi.org/10.1016/j.toxlet.2003.10.031]

-
Krishna, A.; Kumar, S.; Mallavarapu, G. R.; Ramesh, S. J. Essent. Oil Res. 2004, 16, 520–522.
[https://doi.org/10.1080/10412905.2004.9698786]

-
Moghaddam, M.; Pirbalouti, A. G.; Babaei, K.; Farhadi, N. J. Essent. Oil Bear. Plants 2021, 24, 990–997.
[https://doi.org/10.1080/0972060X.2021.2005691]

-
Martínez, R.; Diaz, B.; Vásquez, L.; Compagnone, R. S.; Tillett, S.; Canelón, D. J.; Torrico, F.; Suárez, A. I. J. Essent. Oil Bear. Plants 2009, 12, 476–481.
[https://doi.org/10.1080/0972060X.2009.10643747]

-
Kafaltiya, M.; Lohani, H.; Haider, S. Z.; Chauhan, N.; Joshi, N. J. Chem. Pharm. Sci. 2019, 12, 117–122.
[https://doi.org/10.30558/jchps.20191204002]

-
Sagar, P. K.; Khan, A. S.; Sonali. S. Int. J. Pharm. Pharm. Sci 2022, 4, 57–66.
[https://doi.org/10.33545/26647222.2022.v4.i2a.90]

-
Scandiffio, R.; Geddo, F.; Cottone, E.; Querio, G.; Antoniotti, S.; Gallo, M. P.; Maffei, M. E.; Bovolin, P. Nutrients 2020, 12, 3273.
[https://doi.org/10.3390/nu12113273]

-
Cruz, E. M. O.; Pinto, J. A. O.; Fontes, S. S.; Arrigoni-Blank, M. de F.; Bacci, L.; Jesus, H. C. R.; Santos, D. A.; Alves, P. B.; Blank, A. F. ScientificWorldJournal 2014, 2014, 314626.
[https://doi.org/10.1155/2014/314626]

-
Santos, C. P.; Pinto, J. A. O.; Santos, C. A.; Cruz, E. M. O.; Arrigoni-Blank, M. F.; Andrade, T. M.; Santos, D. A.; Alves, P. B.; Blank, A. F. Ind. Crops. Prod. 2016, 79, 205–210.
[https://doi.org/10.1016/j.indcrop.2015.11.015]

-
Stratakos, A. C.; Koidis, A. In Essential Oils in Food Preservation, Flavor and Safety; Preedy, V. R. Ed. Preedy, Victor R.; Academic Press: San Diego, 2016; pp 31–38.
[https://doi.org/10.1016/B978-0-12-416641-7.00004-3]

-
Ribeiro, B. S.; Ferreira, M. F.; Moreira, J. L.; Santos, L. Cosmetics 2021, 8, 117.
[https://doi.org/10.3390/cosmetics8040117]

-
Aziz, Z. A. A.; Ahmad, A.; Setapar, S. H. M.; Karakucuk, A.; Azim, M. M.; Lokhat, D.; Rafatullah, M.; Ganash, M.; Kamal, M. A.; Ashraf, G. M. Curr. Drug Metab. 2018, 19, 1100–1110.
[https://doi.org/10.2174/1389200219666180723144850]

-
Souza Junior, E. T. de; Siqueira, L. M.; Almeida, R. N.; Lucas, A. M.; Silva, C. G. F. da; Cassel, E. Braz. arch. biol. technol. 2020, 63, e20190213.
[https://doi.org/10.1590/1678-4324-2020190213]

-
Liao, P.-H.; Yang, H.-H.; Wu, P.-C.; Abu Bakar, N. H.; Urban, P. L. Anal. Chem. 2020, 92, 1228–1235.
[https://doi.org/10.1021/acs.analchem.9b04380]

-
Cai, J.; Liu, B.; Ling, P.; Su, Q. J. Chromatogr. A 2002, 947, 267–275.
[https://doi.org/10.1016/S0021-9673(02)00015-8]

-
Forehand, J. B.; Dooly, G. L.; Moldoveanu, S. C. J. Chromatogr. A 2000, 898, 111–124.
[https://doi.org/10.1016/S0021-9673(00)00827-X]

-
Pollien, P.; Chaintreau, A. Anal. Chem. 1997, 69, 3285–3292.
[https://doi.org/10.1021/ac9701905]

-
Aati, H.; Emam, M.; Alqahtani, J.; Aati, S.; Aati, A.; Wanner, J. K. R.; Seif, M. Mol. 2022, 27, 7242.
[https://doi.org/10.3390/molecules27217242]

-
Héthélyi, E.; Dános, B.; Tétényi, P.; Koczka, I. Flavour Fragr. J. 1986, 1, 169–173.
[https://doi.org/10.1002/ffj.2730010408]

-
Laosinwattana, C.; Wichittrakarn, P.; Teerarak, M. Ind. Crop. Prod. 2018, 126, 129–134.
[https://doi.org/10.1016/j.indcrop.2018.10.013]

-
Francomano, F.; Caruso, A.; Barbarossa, A.; Fazio, A.; La Torre, C.; Ceramella, J.; Mallamaci, R.; Saturnino, C.; Iacopetta, D.; Sinicropi, M. S. Appl. Sci. 2019, 9, 5420.
[https://doi.org/10.3390/app9245420]

-
Fidyt, K.; Fiedorowicz, A.; Strzadala. L.; Szumny, A.; Cancer Med. 2016, 5, 3007–3017.
[https://doi.org/10.1002/cam4.816]

-
Dahham, S. S.; Tabana, Y. M.; Iqbal, M. A.; Ahamed, M. B. K.; Ezzat, M. O.; Majid, A. S. A.; Majid, A. M. S. A. Molecules 2015, 20, 11808–11829.
[https://doi.org/10.3390/molecules200711808]

-
Marques, M. M. M.; Sobrinho, A. C. N.; Morais, S. M.; Fontenelle, R. O. S.; Guedes, M. I. F. Arch. Curr. Res. Int. 2025, 25, 506–514.
[https://doi.org/10.9734/acri/2025/v25i91515]

- Lateef, R.; Bhat, K. A.; Chandra, S.; Banday, J. A. Am. J. Essent. Oil. Nat. Prod. 2018, 6, 35–41.
-
Bassolé, I. H. N.; Juliani, H. R. Molecules 2012, 17, 3989–4006.
[https://doi.org/10.3390/molecules17043989]
