Quantitative Analysis of Bioactive Compounds in a Mixture of the Lindera glauca Leaves Extract and Water-soluble Mastic Gum using an HPLC/UV Method
Abstract
This study aimed to establish and validate an HPLC method for the quantitative analysis of bioactive compounds in a mixture of the L. glauca leaves extract and water-soluble mastic gum (MLM). MLM has shown potential as an effective agent for preventing hair loss in the previous study. For the development of the quality evaluation of MLM, quercitrin (1), isoquercitrin (2), and oleanonic acid (3) were selected as analytical markers. The separation was achieved using a reverse-phase column with a gradient solvent system of 0.1% formic acid aqueous-0.1% formic acid acetonitrile at a flow rate of 1.0 mL/min. Detection was carried out at 210 nm and 254 nm. The calibration curves for all three markers exhibited good linearity (R2 > 0.999). Recoveries of the three markers ranged from 100 ± 15%. The concentrations of compounds 1, 2, and 3 in MLM was determined to be 25.73 ± 1.38, 8.36 ± 0.05, and 212.24 ± 12.88 μg/mL, respectively. The validated method will facilitate further compositional investigations in MLM.
Keywords:
Lindera glauca leaves, water-soluble mastic gum, HPLC method validation, quercitrin, isoquercitrin, oleanonic acidIntroduction
Lindera glauca Sieb. & Zucc. (Lauraceae) is a deciduous tree commonly found in the mountainous regions of Korea, Japan, China, and Taiwan.1 L. glauca has been found to contain flavonoids, lignans, and alkaloids, and has exhibited various physiological activities such as antibacterial, antioxidant, and anticancer effects.2-4 On the other hand, mastic gum is an aromatic resin produced by the evergreen shrub Pistacia lentiscus var. Chia (Anacardiaceae) and has been used as a traditional medicine for over 2500 years.5 Today, mastic gum is widely used in the production of phytotherapeutic products with antibacterial, antioxidant, anti-ulcer, anti-diabetic, anti-inflammatory, and anti-cancer properties.6,7
Alopecia, a chronic dermatological disease that leads to hair loss from the body, is a significant health concern, particularly for the scalp.8 Despite extensive research, the exact cause of alopecia remains unknown; however, stress, genetic factors, and environmental factors have been suggested as potential causes.9,10 Dermal papilla cells, which are specialized dermal fibroblasts located within the hair bulb, play a vital role in the formation of hair follicles, hair regeneration, and hair growth by interacting with various types of epithelial cells within the hair follicle.11 Thus, the growth and proliferation of dermal papilla cells, as well as the inhibition of apoptosis, have become important targets for the development of drugs that maintain the growth phase of hair and inhibit hair loss.11
In recent research, we confirmed that a mixture of the L. glauca leaves extract and water-soluble mastic gum (MLM) can potentially be used as a new agent for hair loss treatment by promoting the proliferation of dermal papilla cells.12 Hence, in the present study, we developed an efficient HPLC analysis method for the quality control and standardization of MLM.
Experimental
General experimental procedures – HPLC grade water and acetonitrile were purchased from TEDIA (Fairfield, USA). Formic acid (FA) was obtained from Daejung (Siheung, Korea). Three standard compounds (1-3) were purchased from ChemFaces (Hubei, China), and were HPLC analytical grade with a more than 98% purity. Each extract of L. glauca leaves and water-soluble mastic gum were provided by FromBio. Co., Ltd (Yongin-si, Korea), and its preparation methods are patented (Patent No. KR2328824).
HPLC apparatus and chromatographic conditions – The chromatographic system for quantitative analysis consisted of a 1260 quaternary pump, an autosampler, and a multiple wavelength detector (Agilent Technologies, Santa Clara, CA, USA). The chromatographic separation of analyses was performed using a Hector M C18-M51002546 (RSTech, Korea; 5 μm, 4.6 × 250 mm). The mobile phase was a gradient solvent system consisting of solvent A (0.1% FA in water) and solvent B (0.1% FA in acetonitrile) as follows: linear gradient 15-25-75-80-100% B (0-20-25-50-51 min) and isocratic 100% B (51-60 min). The flow rate was 1.0 mL/min, and injection volume was 10 μL (Table 1). UV absorption was monitored at 210 and 254 nm. The column temperature was maintained at 30oC.
Preparation of standard solutions – Based on the solubility of each component in water, a stock standard solution was prepared by dissolving 1.00 mg of each compound in 1 mL methanol. Seven and six additional calibration levels (compound 1 and 2; 5, 10, 25, 50, 100, 250 and 500 μg/mL, compound 3; 50, 100, 200, 250, 500 and 1000 μg/mL) were prepared by diluting this stock solution with methanol. These solutions were stored at 4oC.
Sample preparation – To prepare the samples, 1.0 mg of L. glauca leaves extract was accurately weighed and dissolved in 5 mL of methanol to obtain a solution with a final concentration of 0.2 mg/mL (solution A). Next, 5 mg of water-soluble mastic gum was accurately weighed and dissolved in 1 mL of water (5 mg/mL). It was then diluted 200 times to obtain a solution with a final concentration of 0.025 mg/mL (solution B). Solutions A and B were mixed at a 1:9 ratio, and the resulting mixture was concentrated. Finally, the concentrated mixture was dissolved in methanol to prepare a final concentration of 100 mg/mL. After filtration using a 0.45 μm filter membrane, the solution was utilized for HPLC method validation.
Specificity – The specificity of the marker compounds was evaluated by comparing the retention time (tR) of the standard compounds 1-3 in the HPLC chromatogram and the tR of the standard substances in the sample.
Linearity – The stock standard solutions of each compound 1-3 were prepared and diluted with methanol to appropriate concentrations (compound 1 and 2; 5, 10, 25, 50, 100, 250 and 500 μg/mL, compound 3; 50, 100, 200, 250, 500 and 1000 μg/mL) for constructing calibration curves. Seven and six concentration levels of the standard solutions were injected, and the area of each standard peak was represented by a regression line. Linearity (correlation coefficient values R2 > 0.999) was evaluated based on the calibration curves.
Precision – Standard solutions were prepared at three concentrations (100, 250 and 500 μg/mL) and measured three times within a day to determine the coefficient of variation of the area value for intraday precision. Standard solutions for storage of three concentrations were measured once a day, and interday precision was measured by obtaining the coefficient of variation of the value measured at 3-day intervals. In each case, it was evaluated whether the coefficient of variation was within 10%.
Accuracy – The accuracy of the method was assessed by a recovery test. Accurate amounts of three concentrations (25, 50 and 100 μg/mL) of standard solutions 1-3, which was quantified previously, were added into a sample solution. The concentration of the standard material was calculated based on the calibration curve, and this was repeated three times to obtain the recovery rate of the sample. At each concentration, it was evaluated whether the recovery rate was within 100 ± 15% and the coefficient of variation was within 10%.
Result and Discussion
An HPLC method was developed for quantitative analysis of bioactive compounds in MLM. Three standards compounds, quercitrin (1), isoquercitrin (2), and oleanonic acid (3) (Fig. 1), were chosen for the development of method validation for MLM based on the relevant information from literature.2,13
The specificity of the three compounds in MLM was confirmed by comparing their retention times and chromatographic peaks. Fig. 2 showed representative chromatograms of three markers and MLM. Compounds 1-3 were detected at 19.51, 15.71 and 49.18 mins, respectively, in the HPLC chromatogram, and these peaks showed good resolution and symmetry. Calibration curves for each compound were prepared using seven and six concentrations of methanol dilution, as shown in Table 2, demonstrating good linearity with R2 > 0.999. The precision of the method was evaluated by measuring the three concentrations of compounds 1-3 three times within a single day to determine intraday precision. Additionally, measurements were taken once daily over a span of three days to establish interday precision. The results indicated that both intraday and interday precisions were within 10% relative standard deviation (Table 3). To confirm the recovery rate of compounds 1-3, they were spiked at three concentrations into MLM. The accuracy results are shown in Table 3, showing that the recovery rate of each compound was within 100 ± 15%. These findings provided strong evidence that the quantification study of compounds 1-3 in MLM were valid and reliable.
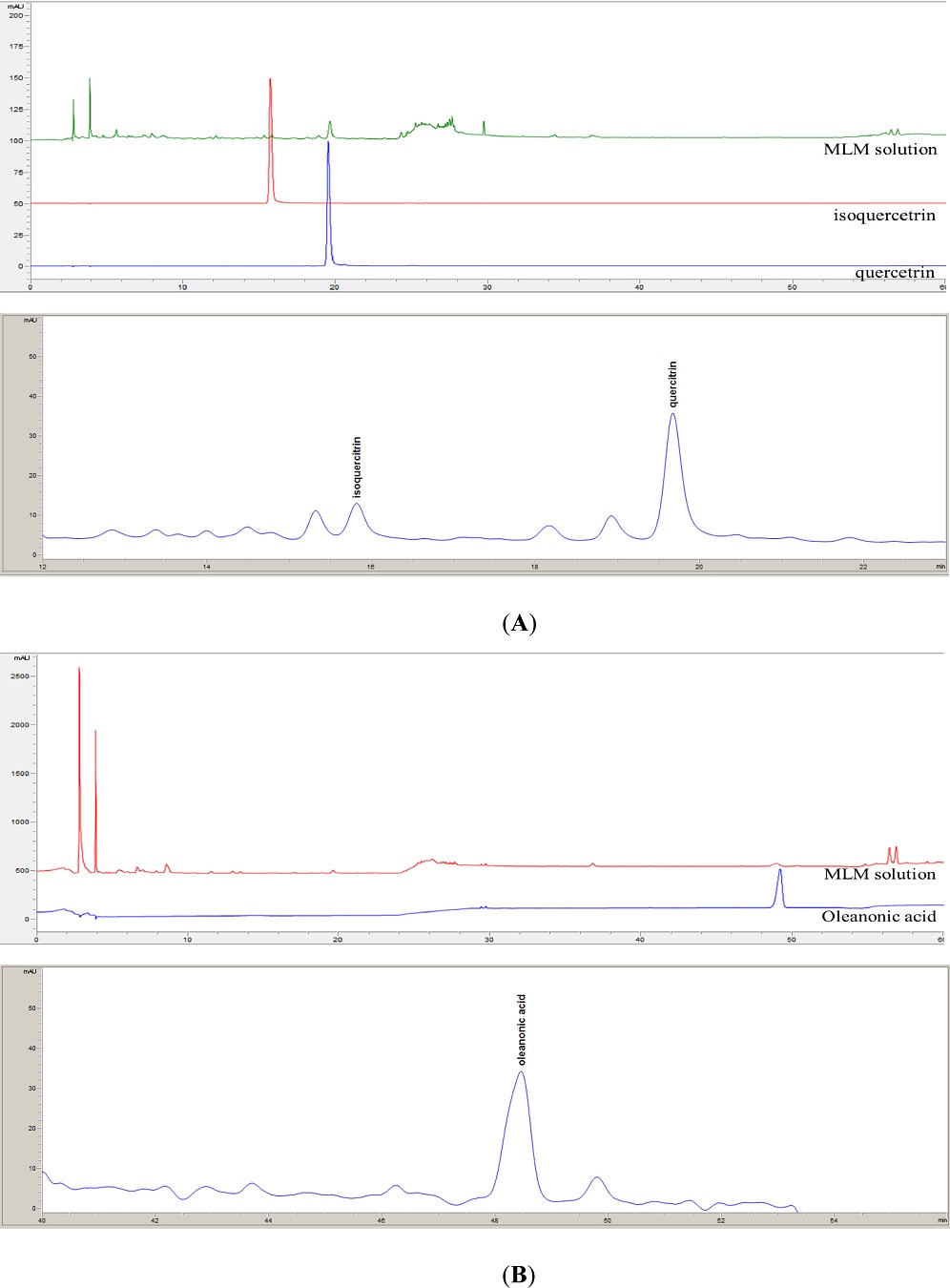
HPLC chromatogram of quercitrin (1), isoquercitrin (2), and oleanonic acid (3) isolated from MLM at 254 nm (A) and 210 nm (B).
The established analytical method was applied to quantitatively analyze the MLM for three compounds 1-3 using the developed regression equation. The concentrations of each compound in MLM samples were as follows: 1 (25.73 ± 1.38 μg/mL), 2 (8.36 ± 0.05 μg/mL), and 3 (212.24 ± 12.88 μg/mL) (Table 4).
The potential of MLM as a promising material for preventing hair loss was attributed to its ability to promote the proliferation of dermal papilla cells.12 In this study, we had developed, validated, and presented a reliable quantitative analysis method for three standard compounds in MLM. Although the specific mechanism of the three compounds in relation to hair loss improvement research has not yet been identified, the results from our study were expected to be valuable as it has identified and quantified the markers present in MLM. It was anticipated that our proposed analysis method will facilitate the efficient development of MLM compositions and support further research on improving hair loss.
Acknowledgments
This study was supported by the research grant of Kangwon National University in 2023
Conflict of Interest
The authors declare a potential conflict of interest in relation to the research reported in this paper. The investigated MLM, which were examined for their efficacy in hair loss prevention, are the subject of a patent held by FromBio. Co., Ltd (Yongin-si, Korea). The authors have a cooperative relationship with FromBio and have received support for their research. The collaboration and research were conducted in accordance with ethical guidelines, and the authors have made efforts to ensure the impartiality and accuracy of the presented results. FromBio had no direct involvement in the design, data collection, analysis, or interpretation of the study.
References
-
Zhu, B.; Hou, X.; Niu, J.; Li, P.; Fang, C.; Qiu, L.; Ha, D.; Zhang, Z.; Sun, J.; Li, Y.; Lin, S. J. Essent. Oil-Bear. Plants 2016, 19, 926-935.
[https://doi.org/10.1080/0972060X.2014.981598]

-
Chang, Y. C.; Chang, F. R.; Wu, Y. C. J. Chin. Chem. Soc. 2000, 47, 373-380.
[https://doi.org/10.1002/jccs.200000050]

-
Kim, K. H.; Moon, E.; Ha, S. K.; Suh, W. S.; Kim, H. K.; Kim, S. Y.; Choi, S. U.; Lee, K. R. Chem. Pharm. Bull (Tokyo). 2014, 62, 1136-1140.
[https://doi.org/10.1248/cpb.c14-00381]

- Wang, S.; Liu, J.; Gao, Q.; Mo, J.; Wang, X.; Chen, X.; Jiang, H. Gen. Chem. 2021, 7, 210005.
-
Pachi, V. K.; Mikropoulou, E. V.; Gkiouvetidis, P.; Siafakas, K.; Argyropoulou, A.; Angelis, A.; Mitakou, S.; Halabalaki, M. J. Ethnopharmacol. 2020, 254, 112485.
[https://doi.org/10.1016/j.jep.2019.112485]

- Dimas, K. S.; Pantazis, P.; Ramanujam, R. In Vivo 2012, 26, 777-785.
-
Soulaidopoulos, S.; Tsiogka, A.; Chrysohoou, C.; Lazarou, E.; Aznaouridis, K.; Doundoulakis, I.; Tyrovola, D.; Tousoulis, D.; Tsioufis, K.; Vlachopoulos, C.; Lazaros, D. Nutrients 2022, 14, 590.
[https://doi.org/10.3390/nu14030590]

-
Hunt, N.; McHale, S. BMJ 2005, 331, 951-953.
[https://doi.org/10.1136/bmj.331.7522.951]

-
Paus, R.; Arck, P. J. Invest. Dermatol. 2009, 129, 1324-1326.
[https://doi.org/10.1038/jid.2009.111]

-
Simakou, T.; Butcher, J. P.; Reid, S.; Henriquez, F. L. J. Autoimmun. 2019, 98, 74-85.
[https://doi.org/10.1016/j.jaut.2018.12.001]

-
Driskell, R. R.; Clavel, C.; Rendl, M.; Watt, F. M. J. Cell Sci. 2011, 124, 1179-1182.
[https://doi.org/10.1242/jcs.082446]

- FromBio Co., Ltd. Composition for Hair Growth Stimulation or Hair Loss Prevention Using Solubilized Mastic Gum and an Extract of Lindera glauca. KR Patent. 2021, KR10-2328824.
- Dimas, K. S.; Pantazis, P.; Ramanujam, R. In vivo 2012, 26, 777-785.