Quantitative HPLC-DAD Analysis of Gallic and Ellagic Acids and UVB-Protective Effects of Solvent Extracts of Quercus acuta Fruits
Abstract
Quercus acuta Thunb. (Q. acuta) is a subtropical, evergreen, broad-leaved tree with excellent carbon sequestration capacity and ecological adaptability. However, the phytochemical composition of fruits, particularly phenolic marker compounds, has not been sufficiently investigated. In this study, the biological activities of water and ethanol extracts of Q. acuta fruits were evaluated, and a high-performance liquid chromatography with diode array detection (HPLC-DAD)-based method for the quantification of marker compounds was established and validated. MTT assays showed no cytotoxicity at concentrations of 5–20 μg/mL, and the water extract significantly inhibited UVB-induced MMP-1 expression in HaCaT keratinocytes. Gallic acid and ellagic acid in Q. acuta fruits were confirmed as marker compounds, and the validated method demonstrated excellent linearity (R² > 0.999), recovery, and precision (RSD < 2.0%). Quantitative analysis revealed that the water extract contained higher levels of phenolic compounds compared with the 70% and 100% ethanol extracts. These findings support the stability and biological efficacy of phenolic marker compounds in aqueous environments and suggest the potential industrial applications of Q. acuta fruit extracts.
Keywords:
Quercus acuta Thunb., Gallic acid, Ellagic acid, HPLC-DAD, MMP-1Introduction
Climate change is expected to significantly alter the climatic suitability of arable land, affecting the spatial distribution and diversity of global food crops. Consequently, the ability to adapt to these changes and develop mitigation strategies is becoming increasingly important.1 Tree species in subtropical regions play a crucial role in climate mitigation due to their excellent carbon sequestration and storage capabilities.2 Q. acuta (Japanese evergreen oak, Quercus acuta Thunb.) is a subtropical evergreen broad-leaved tree native to Korea and Japan. It is primarily distributed along the coastal regions of Jeollanam-do Province and Jeju Island, with the Wando region accounting for approximately 18% of the domestic subtropical evergreen forest area.3–4 Plants of the genus Quercus have traditionally been used for the treatment of diarrhea, menorrhagia, and various inflammatory disorders, while galls have been applied for the treatment of wounds and burns.5–7 Various parts of Q. acuta have been reported to possess diverse bioactivities, including antioxidant, anti-inflammatory, antimicrobial, and antiviral effects.8-11 Major phytochemical constituents include catechin, isoquercitrin, taxifolin, fraxin, chlorogenic acid, galloyl protoquercetin, and gallic acid9,12–13 In our previous study, gallotannins and ellagic acid were identified as marker compounds in the water extract of Q. acuta fruits, and the extract was shown to inhibit the expression of matrix metalloproteinase-1 (MMP-1), a key enzyme involved in skin aging, thereby exhibiting anti-photoaging effects.14
Tannins are high-molecular-weight polyphenolic compounds widely found in Quercus fruits, exhibiting antioxidant, antimicrobial, anti-inflammatory, and UV-protective activities.14–16 They can be broadly classified into hydrolyzable and condensed tannins.17 However, due to their complex polymeric structure, direct quantification of tannins in Q. acuta fruits is challenging. Therefore, they are commonly hydrolyzed into low-molecular-weight compounds such as gallic acid for indirect quantification.18 At present, there is no validated analytical method available for the quantification of these marker compounds in Q. acuta fruits.
In this study, extraction conditions using various solvents were optimized, and the biological efficacy of Q. acuta fruit extracts was evaluated by assessing MMP-1 expression inhibition in UVB-irradiated HaCaT keratinocytes. In addition, a reliable high-performance liquid chromatography with diode array detection (HPLC-DAD) method was developed and validated for quantifying the marker compounds, gallic acid and ellagic acid. This approach provides scientific evidence for the quality standardization and industrial application of Q. acuta fruit extracts, while simultaneously offering foundational data supporting their potential as a natural source of bioactive tannins.
Experimental
Plant material – The fruits of Q. acuta were collected from Wando-gun, Jeollanam-do, Republic of Korea, where this species naturally regenerates. Voucher specimens of the fruit (JBF1201) and extract (JBF2202) were preserved at the Natural Resources Research Laboratory of the Jeonnam Bioindustry Foundation (JBF), Korea. The Q. acuta fruit was authenticated by Dr. Yonguk Kim. The fruits (1 kg) were washed to remove impurities, air-dried, and ground into fine powder for subsequent experiments. For extraction, 30 g of the powder was refluxed with 600 mL of distilled water at 100°C for 3 h. Similarly, 30 g of the powder was extracted with 600 mL of 70% (v/v) or 100% ethanol at 70°C for 3 h. The resulting extracts were freeze-dried at -45°C for three days. The extraction yields using water, 70% (v/v) ethanol, and 100% ethanol were 12.42%, 12.07%, and 7.45%, respectively.
Analytical instruments and equipment – The gallic and ellagic acid standards were purchased from Sigma-Aldrich (St. Louis, MO, USA). HPLC-grade acetonitrile and water were purchased from J. T. Baker (Phillipsburg, NJ, USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was obtained from Sigma-Aldrich (St. Louis, MO, USA). The Human MMP-1 ELISA kit was obtained from R&D Systems (Minneapolis, MN, USA). Dulbecco’s modified Eagle medium (DMEM), fetal bovine serum (FBS), phosphate-buffered saline (PBS), penicillin, and streptomycin were purchased from Gibco (Carlsbad, CA, USA). All additional reagents and chemicals used in the experiments were of analytical grade and were suitable for laboratory use.
Preparation of standard solutions – Gallic acid (100 mg) and ellagic acid (100mg) were accurately weighed and dissolved in 100% methanol by sonication for 30 min, and then diluted to a final volume of 100 mL in a volumetric flask. This stock solution was further diluted with 100% methanol to prepare working standard solutions in the concentration range of 1300 µg/mL. The solutions were filtered through a 0.22 µm membrane filter prior to analysis.
Preparation of sample solutions – The aqueous powder extract (200 mg) of Quercus acuta fruit was weighed and dissolved in 50% (v/v) methanol, followed by sonication for 60 min. The solution was brought to a final volume of 100 mL in a volumetric flask and initially filtered through a 0.45 µm membrane to remove insoluble particles. To ensure reproducibility and consistency with the method validation, all samples, including the extracts, were filtered through a 0.22 µm membrane immediately prior to HPLC analysis.
HPLC analytical conditions – Chromatographic separation was performed on a Waters HPLC system (e2695) equipped with a 2998 photodiode array detector (Waters Corporation, Milford, MA, USA) and a YMC Triart C18 column (4.6 × 250 mm, 5 µm, YMC, Kyoto, Japan). The mobile phase consisted of solvent A (acetonitrile) and solvent B (0.1% (v/v) formic acid in water). The gradient program was as follows: 98–95% B (0–3 min), 95% B (3–15 min), 95–89% B (15–40 min), 89–77% B (40–50 min), 77–57% B (50–65 min), 57–51% B (65–68 min), and 0% B (68–70 min), followed by re-equilibration at 98% B (70–75 min). The flow rate was 1.0 mL/min, injection volume 10 µL, and the column temperature 30°C. Detection was carried out at 254 nm, and data were acquired and processed using Empower 3 software.
HaCaT keratinocyte culture – HaCaT keratinocytes were cultured in Dulbecco’s modified DMEM medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% FBS and 1% penicillin-streptomycin (Gibco). The cells were maintained at 37°C in a humidified incubator with 5% CO2 to ensure optimal growth conditions.
UVB irradiation – The cells were exposed to UV-B using a UVM-225D MineraLight UV lamp (UVP Inc., Phoenix, AZ, USA) at a wavelength of 302 nm. The irradiation dose was quantified using a UV meter (HD2102-2; Delta OHM, Italy) and the cells were subjected to a UVB dose of 30 mJ/cm.
Cell viability assay – To evaluate the cytotoxicity and cell viability, an MTT assay was performed. HaCaT keratinocytes were seeded in 96-well plates at a density of 0.5 × 105 cells per well and incubated for 24 h. Cells were then treated with extracts of Q. acuta fruits at concentrations of 5, 10, 20, and 50 µg/mL. After 24 h of treatment, MTT solution (0.5 mg/mL in DMEM) was added to each well and incubated for 4 h at 37°C. After incubation, the supernatant was carefully removed, and dimethyl sulfoxide (DMSO) was added to dissolve the resulting formazan crystals. The absorbance was measured at 540 nm using a microplate reader.
ELISA for MMP-1 quantification – The concentration of MMP-1 released into the culture medium was quantified using a commercial ELISA kit (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions.
Statistical analysis – All results are presented as the mean ± standard deviation (SD). Statistical comparisons between groups were conducted using one-way analysis of variance (ANOVA), followed by Dunnett’s post-hoc test, using GraphPad Prism software (version 5.0; GraphPad, USA). The statistical significance was set at p < 0.05. The significance relative to untreated control cells is indicated as #p < 0.05, ##p < 0.01, and ###p < 0.001, and the significance relative to UVB-treated cells is denoted as *p < 0.05, **p < 0.01, and ***p < 0.001.
Specificity – To evaluate the specificity of the marker compounds, gallic acid and ellagic acid, in the water extract of Q. acuta fruit, HPLC chromatograms of the standard and sample solutions were analyzed. The specificity was assessed by comparing the separation (resolution), retention times, and UV spectra corresponding to the peaks.
Linearity, limit of detection (LOD), and limit of quantification (LOQ) – The linearity of the method was evaluated using six concentrations of each standard compound: gallic acid (1–300 µg/mL) and ellagic acid (1–300 µg/mL). Each concentration was measured in triplicate to confirm the reproducibility and calculate the coefficient of determination (R2). The LOD and LOQ were determined based on the standard deviation of the response (σ) for the y-intercept and the mean slope (S) of the calibration curve obtained from three measurements, as follows.
Accuracy and recovery – Accuracy refers to the closeness of a measured value obtained using a test method to the true or standard value. It was assessed using the standard addition method, in which known amounts of standard solutions were added to the test samples, and evaluated by calculating the recovery rate (%) from repeated analyses. Specifically, gallic acid was added to solutions of the water and 100% ethanol extracts of Q. acuta fruits (1 mg/mL) at 25, 50, and 100 µg/mL, and to the 70% ethanol extract (1 mg/mL) at 12, 25, and 50 µg/mL. Ellagic acid was added to all three extracts at 5, 10, and 20 µg/mL. The resulting mixtures were filtered through a 0.22 µm membrane filter and analyzed in triplicate (n = 3).
Precision – The precision was evaluated by assessing intra-day (repeatability) and inter-day (intermediate precision) variations. Repeatability was determined by analyzing each sample solution in triplicate within a single day, while reproducibility was assessed over three consecutive days, with three replicates per day, performed by different analysts. The sample solutions of the water, 70% ethanol, and 100% ethanol extracts of Q. acuta fruits were prepared at low, medium, and high concentrations and filtered through a 0.22-µm membrane filter prior to analysis. (n = 3).
Quantification of marker compounds in extracts – A validated analytical method was used to determine and compare the gallic acid and ellagic acid contents in the water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta fruit powder. Each extract was dissolved to a final concentration of 1 mg/mL and each sample was analyzed in triplicate (n = 3).
Results and Discussion
We previously evaluated the protective effects of water extract of Q. acuta fruit against UVB-induced cytotoxicity in HaCaT keratinocytes.14 In this study, the investigation is extended to include 70% (v/v) and 100% (v/v) ethanol extracts of Q. acuta fruit. The cytotoxic effects of UVB and the water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta on HaCaT keratinocytes were evaluated via MTT assay. Treatment with Q. acuta extract did not cause marked cytotoxicity for 24 h (Fig. 1A). Thus, for further cell-based experiments, the extracts were used at concentrations of 5–20 μg/mL. As shown in Fig. 1B, exposure to UVB radiation (30 mJ/cm2) markedly decreased the viability of HaCaT keratinocytes to 77.67% ± 1.75% relative to the untreated control. Treatment with the water extract significantly restored the cell viability in a concentration-dependent manner to 81.28% ± 0.93%, 85.11% ± 1.80%, and 91.87% ± 1.21% at 5, 10, and 20 μg/mL, respectively. Similarly, the 70% (v/v) ethanol extract improved the cell viability to 79.41% ± 1.46%, 83.09% ± 1.48%, and 86.07% ± 1.03% at the respective concentrations, whereas the 100% ethanol extract restored it to 78.70% ± 1.43%, 80.10% ± 1.70%, and 83.45% ± 0.72%, respectively. The water extract exhibited pronounced protective effects against UVB-induced cytotoxicity. The effect of the extract on UVB-induced MMP-1 expression in HaCaT keratinocytes was also investigated. UVB irradiation markedly elevated the MMP-1 levels to 209.23% ± 3.71% relative to that of the control. According to the ELISA results, treatment with the water extract of Q. acuta significantly suppressed this increase in a dose-dependent manner, reducing the MMP-1 expression to 196.96% ± 4.14%, 184.05% ± 5.23%, and 164.14% ± 5.71% at 5, 10, and 20 μg/mL, respectively. The 70% (v/v) ethanol extract reduced the expression of MMP-1 to 206.02% ± 4.17%, 197.91% ± 5.11%, and 183.35% ± 6.22%, whereas the 100% ethanol extract led to levels of 203.45% ± 4.51%, 198.31% ± 4.67%, and 190.16% ± 3.42% at the corresponding concentrations (Fig. 1C). Among the tested extracts, the aqueous extract most effectively inhibited the UVB-induced expression of MMP-1 (Fig. 1).
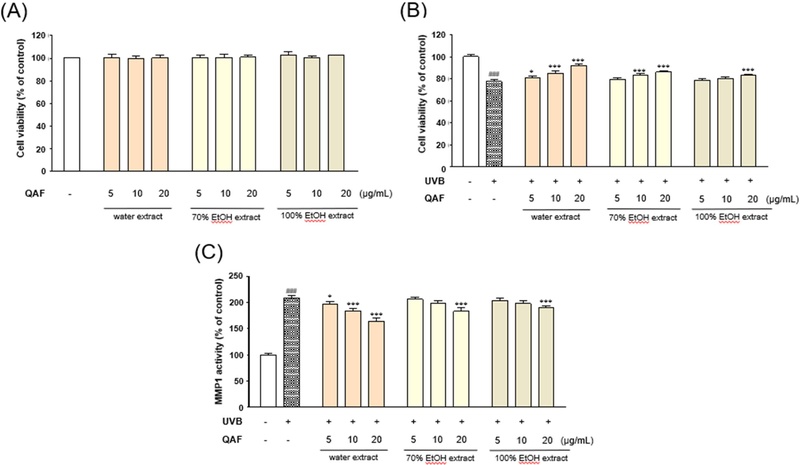
Comparison of effects of water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta fruit on cell viability, proliferation, and MMP-1 production in UVB-irradiated HaCaT keratinocytes. (A) HaCaT keratinocytes were treated with extracts of Q. acuta fruit (water extract, 70% (v/v) ethanol extract, and 100% ethanol extract) at the indicated concentrations for 24 h, and cell viability was assessed using the MTT assay. (B) After UVB irradiation, the cells were treated with Q. acuta fruit extracts (water extract, 70% (v/v) ethanol extract, and 100% ethanol extract) at the specified concentrations for 24 h, followed by evaluation of the cell viability using the MTT assay. (C) Cells were seeded and pretreated with Q. acuta extracts at concentrations of 5, 10, and 20 µg/mL, then exposed to UVB and cultured for an additional 24 h. The concentration of MMP-1 released into the culture medium was quantified using an ELISA assay. All data are presented as the mean ± standard deviation (SD). n = 3 independent experiments, *p < 0.05, **p < 0.01, ***p < 0.001 versus control group; #p < 0.05, ##p < 0.01, ###p < 0.001 versus UVB-treated group.
In our previous study, the water extract of Q. acuta fruits was found to contain gallotannin and ellagic acid as the main marker compounds.14 Gallotannin is a polymeric compound formed by the esterification of gallic acid with a sugar moiety.19 For quantification, the analytical method was modified based on previous procedures. Gallotannin is generally quantified after conversion to gallic acid, a low-molecular-weight compound.18 According to Rashid,20 gallic acid is a heat-sensitive, naturally occurring aromatic carboxylic acid that exhibits reduced stability at elevated temperatures. Therefore, the column temperature was lowered from 35°C to 30°C.21 Based on the UV absorbance characteristics of gallic acid, the mobile phase was changed from methanol to acetonitrile, and the analytical conditions were optimized accordingly. Accordingly, gallic acid was employed as a surrogate standard in this study, while ellagic acid was selected as the indicator compound. Both gallic acid and ellagic acid were consistently detected in the water extract. Furthermore, comparison of the peak areas revealed that the water extract contained higher concentrations of both compounds than the 70% (v/v) and 100% ethanol extracts. These findings are consistent with previous reports indicating that water extraction provides better recovery and reproducibility than methanol for hydrolyzable compounds such as gallic and ellagic acids from chestnut wood, even if the total polyphenol content is lower.17 Overall, the results of this study confirmed that water extraction is more effective than ethanol extraction for isolating water-soluble polyphenols from Q. acuta fruits (Fig. 2).
Specificity refers to the ability of an analytical method to measure a target analyte accurately in the presence of other components.22 Gallic and ellagic acid standards were detected at retention times of approximately 10.58 and 40.98 min, respectively (Fig. 3). These retention times are consistent with those observed for the sample extracts. Furthermore, the UV spectra of the sample solutions matched those of the corresponding standards (Fig. 4). Comparison of the chromatograms of the standard solutions and the water extract of Q. acuta fruit demonstrated that the peaks corresponding to gallic and ellagic acids were well separated and were not affected by co-eluting or interfering substances, thereby verifying the specificity of the method. The linearity was assessed using standard solutions at six concentration levels: 2.5–300 µg/mL for gallic acid and 2.5–300 µg/mL for ellagic acid. Each measurement was performed in triplicate to assess the margin of error. According to the ICH guidelines, the correlation coefficient (R²) of the calibration curve must exceed 0.99.22 The calibration curve for gallic acid was described by the equation y = 20,026x – 2,564 with an R² value of 0.9997, indicating excellent linearity. Similarly, ellagic acid afforded a calibration curve of y = 115,525x + 75,824 with an R² value of 0.9999. The results demonstrate high linearity, with R² values greater than or equal to 0.999. Based on the linearity results, the LOD and LOQ for gallic acid were determined to be 0.53 and 1.59 μg/mL, respectively, whereas the corresponding values for ellagic acid were 0.48 and 1.45 μg/mL. These results confirmed that both compounds could be reliably detected and quantified, even at trace levels (Table 1). The accuracy reflects the degree of agreement between the experimental value and the true or accepted reference values. In this study, the accuracy was assessed by calculating the recovery rate and relative standard deviation (RSD) based on replicate measurements of the samples spiked with a known amount of analyte.22 The concentrations of gallic and ellagic acids in the water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta fruits were used to define low, medium, and high spike levels. Each extract was spiked with standard solutions of gallic and ellagic acids and analyzed in triplicate. Recovery rates of gallic acid ranged from 98.62% to 100.36%, and those of ellagic acid ranged from 98.39% to 100.13% across all extracts and spike levels, with RSDs below 2%. According to AOAC guidelines, acceptable recovery rates range from 90–107%.22 These results demonstrate that the developed HPLC method is reliable and accurate for the simultaneous quantification of gallic and ellagic acids in Q. acuta fruit extracts (Table 2). Precision refers to the degree of agreement between measured values when multiple measurements are obtained from a homogeneous sample under specified conditions. It assesses the repeatability (intra-day precision) of results from repeated measurements of the same sample under identical conditions, as well as the intermediate precision (inter-day precision) of measurements within the same laboratory, considering factors such as different analysts, experimental days, instruments, and equipment.22–24 The intra- and inter-day precision for the quantification of gallic and ellagic acids in all extracts was evaluated at low, medium, and high concentrations through at least three replicate experiments. Intra-day precision, expressed as RSD, ranged from 0.22% to 1.09% for gallic acid and from 0.12% to 1.88% for ellagic acid (Table 3). Inter-day RSD values ranged from 0.22% to 1.91% for gallic acid and from 0.19% to 1.76% for ellagic acid (Table 4–6). As RSD values below 2% are generally considered acceptable for analytical precision,23 these results confirm that the method exhibits excellent precision for the quantification of both compounds (Table 3–6).
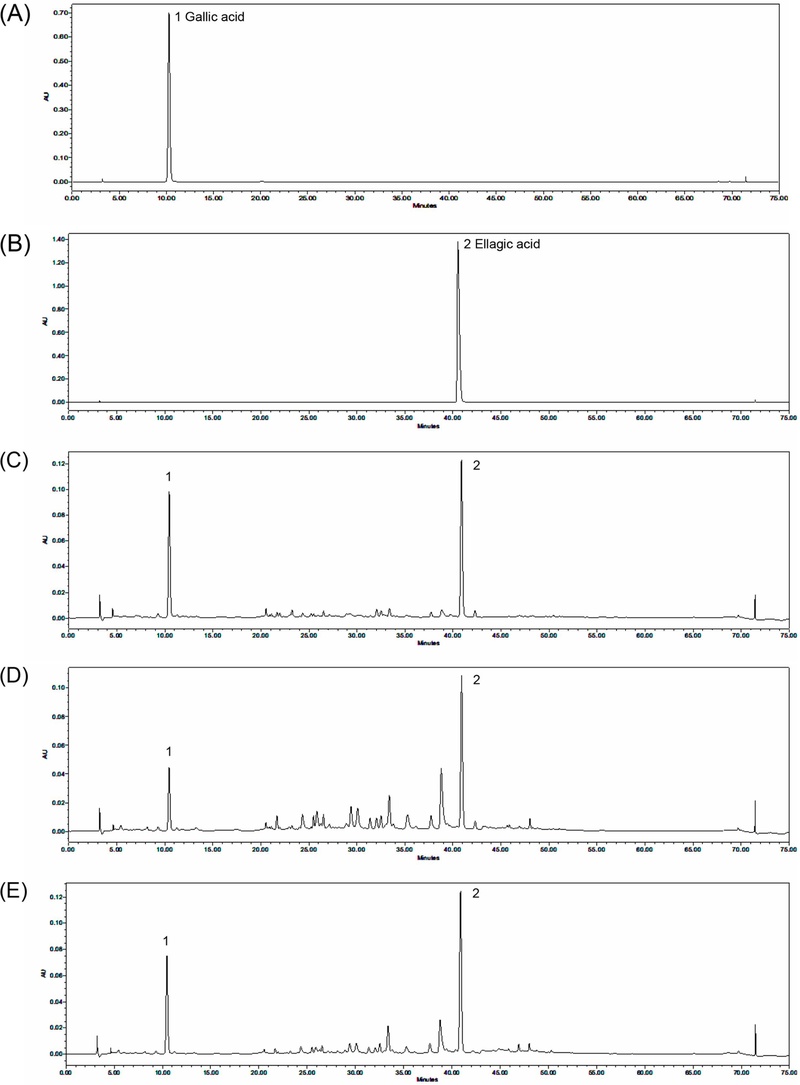
Representative HPLC-DAD chromatograms of Q. acuta fruit extracts using (A) gallic acid and (B) ellagic acid standards, and extracts obtained with (C) water, (D) 70% (v/v) ethanol, and (E) 100% ethanol. The retention times of the gallic acid and ellagic acid standards were 10.28 min and 40.53 min, respectively. In the extracts, the retention times were 10.45 and 40.88 min for the water extract, 10.46 and 40.91 min for the 70% ethanol extract, and 10.46 and 40.89 min for the 100% ethanol extract.
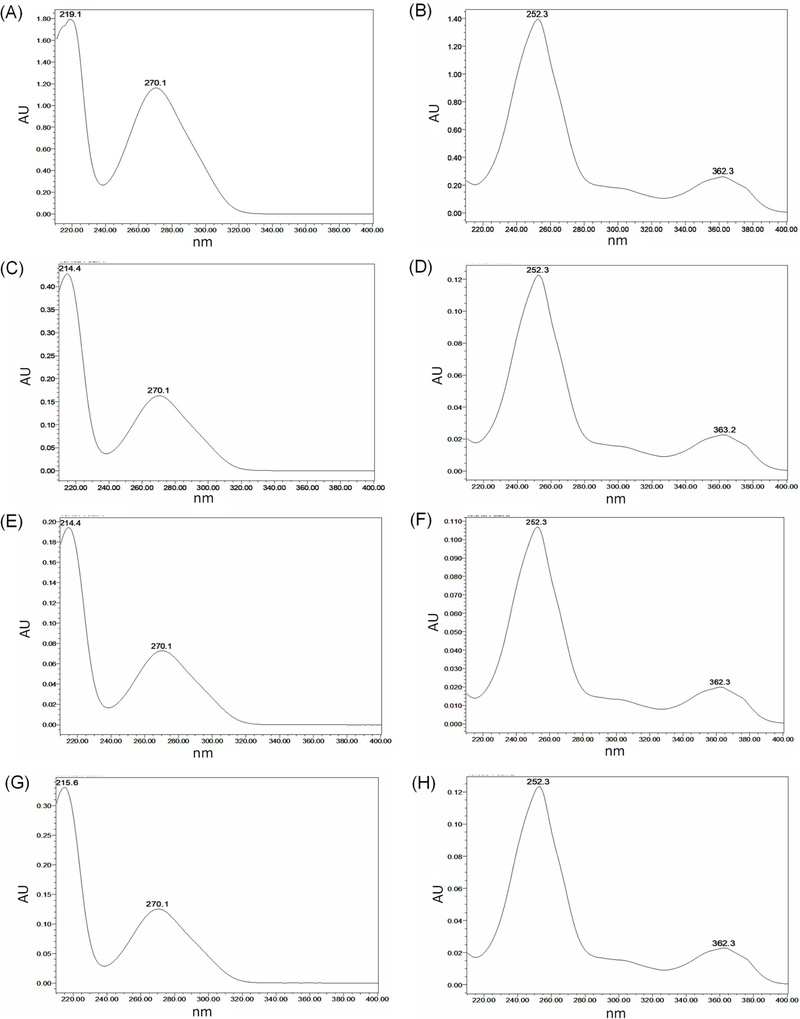
Ultraviolet-visible spectrum (UV spectrum) of gallic acid and ellagic acid standards and Q. acuta fruit extracts. (A) gallic acid standard, (B) ellagic acid standard, (C) gallic acid in water extract, (D) ellagic acid in water extract, (E) gallic acid in 70% ethanol extract, (F) ellagic acid in 70% ethanol extract, (G) gallic acid in 100% ethanol extract, (H) ellagic acid in 100% ethanol extract.

Linearity, limit of detection (LOD), and limit of quantification (LOQ) of gallic acid and ellagic acid(n = 3)

Accuracy of gallic acid and ellagic acid quantification in the water, 70% ethanol, and 100% ethanol extracts of Q. acuta fruit, expressed as recovery(n = 3)

Evaluation of repeatability in the quantification of gallic acid and ellagic acid in all extracts of Q. acuta fruit(n = 3)

Evaluation of intermediate precision (inter-day precision) in the quantification of gallic acid and ellagic acid in the water extract of Q. acuta fruit(n = 3)

Evaluation of intermediate precision (inter-day precision) in the quantification of gallic acid and ellagic acid in the 70% ethanol extract of Q. acuta fruit(n = 3)

Evaluation of intermediate precision (inter-day precision) in the quantification of gallic acid and ellagic acid in the 100% ethanol extract of Q. acuta fruit(n = 3)

Comparison of gallic acid and ellagic acid contents in water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta fruit (n = 3) and RSD (%) values
Using the validated analytical method, the gallic and ellagic acid contents in the water, 70% (v/v) ethanol, and 100% ethanol extracts of Q. acuta fruits were quantified. The concentrations of gallic acid and ellagic acid were 55.49 and 12.53 mg/g in the water extract, 24.65 and 10.57 mg/g in the 70% (v/v) ethanol extract, and 42.67 and 12.48 mg/g in the 100% ethanol extract, respectively. The RSD values for gallic acid and ellagic acid were 0.56% and 0.35% in the water extract, 0.44% and 0.30% in the 70% (v/v) ethanol extract, and 0.48% and 0.89% in the 100% ethanol extract, respectively (Table 5). These results confirmed that gallic acid content was significantly higher in the water extract than in the ethanolic extracts, while ellagic acid showed no significant difference among the water, 70% (v/v) ethanol, and 100% ethanol extracts. In addition, a supplementary time-dependent experiment of high-temperature water extraction showed that the recovery of both marker compounds increased with extraction time, although the rate of increase slowed around 5 hours. These findings support that water extraction is effective for obtaining key phenolic compounds and may have practical implications for future applications.
Gallic acid, a low-molecular-weight compound derived from gallotannin, was identified as a marker alongside ellagic acid, and, despite being common compounds, they were selected as suitable markers for industrial quality control purposes. A stable analytical method was successfully developed and validated. Comparative analysis revealed that water-soluble compounds showed good recovery and reproducibility in the aqueous extract. Furthermore, the water extract significantly inhibited UVB-induced MMP-1 expression in HaCaT keratinocytes, supporting its biological relevance. Although gallic acid and ellagic acid are the major phenolic compounds in Quercus acuta extracts, overall bioactivity, including cell viability and MMP-1 inhibition, may be influenced by other bioactive constituents such as flavonoids and tannins. Therefore, the observed activity is likely attributable to synergistic effects among multiple compounds rather than to individual phenolics alone.
While various unidentified peaks were observed in the chromatograms, most are likely hydrolysis products of hydrolyzable tannins, making structural elucidation of individual secondary metabolites challenging. Notably, the water extract provided reliable quantitative data for gallic acid and ellagic acid. The purpose of this study was to quantify marker compounds under practical extraction conditions rather than to achieve complete hydrolysis of tannins. Although partial hydrolysis may occur during high-temperature water extraction, the extraction temperature of 100°C was selected to simulate the real preparation process of hot-water extracts, which are widely used in functional food and cosmetic formulations.
These findings support the potential application of Q. acuta fruit water extracts in further biological and industrial studies. Further investigations employing advanced analytical techniques are warranted to explore the complete metabolic profile of the fruit.
Acknowledgments
This study was supported by the R&D Program for Forest Science Technology (Project No. RS-2023-KF002467) through the Korea Forest Service (Korea Forestry Promotion Institute, Korea).
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Heikonen, S.; Heino, M.; Jalava, M.; Siebert, S.; Viviroli, D.; Kummu, M. Nat. Food 2025, 6, 331–342.
[https://doi.org/10.1038/s43016-025-01135-w]

-
Yang, S. H.; Lee, Y. J.; Kang, H. S.; Shin, H. J.; Choi, M. H. Kor. Soc. Biotechnol. Bioeng. J. 2024, 39, 25–33.
[https://doi.org/10.7841/ksbbj.2024.39.1.25]

- Shin, M.-S.; Seo, C.; Park, S.-U.; Hong, S.-B.; Kim, J.-Y.; Jeon J.-Y.; Lee M. J. Environ. Impact Assess. 2018, 27, 291‒306.
- Kim, S.; Park, I. H. Korean J. Environ. Ecol. 2014, 35, 621‒631.
-
Huang, J.; Wang, Y.; Li, C.; Wang, X., He, X. Molecules 2016, 21, 669.
[https://doi.org/10.3390/molecules21050669]

-
Sheu, S.-Y.; Tsuang, Y.-H.; Hsu, F.-L.; Lu, F.-J.; Chiang, H.-C. Am. J. Chin. Med. 1997, 25, 307–315.
[https://doi.org/10.1142/S0192415X97000342]

-
Umachigi, S. P.; Jayaveera, K. N.; Ashok Kumar, C. K.; Kumar, G. S.; Vrushabendra Swamy, B. M.; Kishore Kumar, D. V.; Trop J Pharm Res. 2008, 7, 913–919.
[https://doi.org/10.4314/tjpr.v7i1.14677]

-
Oh, M. H.; Park, K. H.; Kim, M. H.; Kim, H. H.; Kim, S. R.; Park, K. J.; Heo, J. H.; Lee, M. W. Asian J. Chem. 2014, 26, 4582‒4586.
[https://doi.org/10.14233/ajchem.2014.16116]

-
Kim, B.; Kim, Y. S.; Hwang, Y.-H.; Yang, H. J.; Li, W.; Kwon, E.-B.; Kim, T. I.; Go, Y. H.; Choi, J.-G. Antioxidants 2021, 10, 1638.
[https://doi.org/10.3390/antiox10101638]

-
Kim, J. K.; Yang, H. J.; Go, Y. H. Antioxidants 2022, 11, 1851.
[https://doi.org/10.3390/antiox11101851]

-
Kim, M. H.; Park, D.-H.; Bae, M.-S.; Song, S.-H.; Seo, H.-J.; Han, D.-G.; Oh, D.-S.; Jung, S.-T.; Cho, Y.-C.; Park, K.-M.; Bae, C.-S.; Yoon, I.-S.; Cho, S.-S. Molecules 2018, 23, 1772.
[https://doi.org/10.3390/molecules23071772]

-
Kim, Y.; Kim, S. W.; Lee, H.-K.; Cho, J.-Y.; Moon, J.-H. J. Kor. Tea Soc. 2023, 29, 63‒71.
[https://doi.org/10.29225/jkts.2023.29.2.63]

-
Serit, M.; Okubo, T.; Su, R.-H.; Hagiwara, N.; Kim, M.; Iwagawa, T.; Yamamoto, T. Agric. Biol. Chem. 1991, 55, 19‒23.
[https://doi.org/10.1080/00021369.1991.10870529]

-
Hong, J.-A.; Bae, D.; Oh, K.-N.; Oh, D.-R.; Kim, Y.; Kim, Y.; Im, S. J.; Choi, E.-J.; Lee, S.-G.; Kim, M.; Jeong, C.; Choi, C. Y. BMC Complement. Med. Ther. 2022, 22, 6.
[https://doi.org/10.1186/s12906-021-03473-1]

-
Gharzouli, K.; Khennouf, S.; Amira, S.; Gharzouli, A. Phytother Res. 1999, 13, 42–45.
[https://doi.org/10.1002/(SICI)1099-1573(199902)13:1<42::AID-PTR383>3.0.CO;2-2]

-
Ozogul, Y.; Ucar, Y.; Tadesse, E. E.; Rathod, N.; Kulawik, P.; Trif, M.; Esatbeyoglu, T.; Ozogul, F. Applied Food Research. 2025, 5, 100738.
[https://doi.org/10.1016/j.afres.2025.100738]

-
Richard-Dazeur, C.; Jacolot, P.; Niquet-Léridon, C.; Goethals, L.; Barbezier, N.; Anton, P. M. Heliyon 2023, e18993.
[https://doi.org/10.1016/j.heliyon.2023.e18993]

-
Newsome, A. G.; Yongchao, L.; Breemen, R. B. J. Agric. Food Chem. 2016, 64, 1326–1334.
[https://doi.org/10.1021/acs.jafc.5b04966]

-
Yan, J.; Yoo, D. I.; Shin, Y. Text. Color. and Finish. 2015, 27, 281‒287.
[https://doi.org/10.5764/TCF.2015.27.4.281]

-
Rashid, M. A. Geochemistry of marine humic compouns; Sptinger Verlag, 1985; p 35.
[https://doi.org/10.1007/978-1-4615-7098-1_2]

-
Lee, S.-Y.; Jeong, H.-S.; Lee, M.-Y.; Kim, K.-H.; Choi, J.-H.; Kwon, K.-I.; Lee, H.-Y. J. Korean Soc. Food Sci. Nutr. 2024, 53, 53‒62.
[https://doi.org/10.3746/jkfn.2024.53.1.53]

- International Council for Harmonisation (ICH). Validation of Analytical Procedures Q2(R2). ICH, 2023; Geneva, Switzerland
- AOAC International. Appendix F: Guidelines for Standard Method Performance Requirements. 2016.
-
Kwon, J. G.; Kim, J. K.; Seo, C.; Hong, S. S.; Ahn, E.-K.; Seo, D.-W.; Oh, J. S. Korean Soc. Food Sci. Nutr. 2016, 45, 61‒67.
[https://doi.org/10.3746/jkfn.2016.45.1.061]