
In Vitro Evaluation of the Anti-Proliferative and Anti-Diabetic Activities of Streptocaulon juventas Leaf Extracts
Abstract
Streptocaulon juventas, a medicinal plant traditionally used in Vietnam and other Southeast Asian countries, has been previously investigated for certain biological activities of its roots; however, studies on its leaves remain limited. In this study, leaf extracts were evaluated for their in vitro anti-proliferative activity against B16F10 (murine melanoma) and HepG2 (human hepatocellular carcinoma) cell lines, as well as for their α-glucosidase inhibitory potential. MTT assays revealed that the hexane (Sj-Hx) and dichloromethane (Sj-DCM) fractions exhibited the strongest cytotoxic effects on B16F10 cells, with IC₅₀ values of 36.09 µg/mL and 45.91 µg/mL, respectively. The hexane fraction (Sj-Hx) also demonstrated potent activity against HepG2 cells, with an IC50 value of 36.65 µg/mL. Notably, none of the extracts exhibited cytotoxicity toward Hs68 normal human fibroblasts, indicating favorable selectivity. DAPI staining further confirmed that both Sj-Hx and Sj-DCM induced nuclear fragmentation and chromatin condensation, consistent with apoptosis induction. Additionally, in vitro assays of anti-diabetic activity revealed that the hexane (Sj-Hx) and ethyl acetate (Sj-EA) extracts displayed the strongest α-glucosidase inhibition, with IC₅₀ values of 221.59 µg/mL and 265.73 µg/mL, respectively. Collectively, these findings suggest that S. juventas leaf extracts possess selective pro-apoptotic activity against cancer cells and represent a promising natural source for the discovery of anti-diabetic agents.
Keywords:
Streptocaulon juventas, Selective cytotoxicity, Apoptosis, α-GlucosidaseIntroduction
According to recent statistics, cancer and cardiovascular diseases are the leading causes of death in many countries.1 Type 2 diabetes mellitus (T2DM), which represents approximately 90% of all diabetes cases, has emerged as a major global health challenge in the twenty-first century. Cardiovascular complications associated with T2DM significantly increase mortality risk, highlighting the urgent need for effective preventive and therapeutic strategies.2
Despite significant advancements in cancer treatment, such as targeted therapies, hormone therapies, and monoclonal antibodies, patients, particularly those in developing countries, still struggle to access adequate treatment due to the prohibitive costs of these regimens. Conventional chemotherapy remains widely used; however, its severe side effects, caused by the drugs’ impact on healthy cells and tissues, pose major challenges for patients and hinder their recovery.3 Therefore, research aimed at discovering bioactive compounds with anticancer potential and fewer side effects remains an urgent priority. Medicinal plants have garnered significant interest due to their remarkable bioactive potential. Recent studies indicate that more than 60% of anticancer agents are derived from natural sources. Notable plant-derived compounds used in cancer treatment include vinca alkaloids from Catharanthus roseus and paclitaxel from Taxus brevifolia.4
In the ongoing efforts to discover effective treatments for type 2 diabetes, drugs such as glucagon-like peptide-1 (GLP-1) receptor agonists and sodium-glucose cotransporter-2 (SGLT-2) inhibitors have been successfully developed and applied. However, these medications do not provide optimal glycaemic control or complication reduction for all patients.5 Natural sources continue to be regarded as a promising avenue for identifying new agents that can offer effective and safe blood glucose management. Although numerous studies have reported potential antidiabetic activities of natural compounds, none have yet received FDA approval.6
Streptocaulon juventas (Lour.) Merr., a medicinal plant belonging to the Apocynaceae family, is widely distributed across Southeast Asian countries. For centuries, it has been used in Vietnamese traditional medicine to treat conditions such as fever, inflammation, wounds, and to promote overall health and vitality.7,8 From the root part of the plant, bioactive chemical constituents have been identified, including cardenolides such as periplogenin and digitoxigenin, which exhibit cytotoxic and antioxidant activities.9,10 Extracts from the roots of S. juventas have demonstrated anticancer activity against A549, HT-1080, and HeLa cell lines.11–13 Recently, corchorusoside C isolated from the roots of this plant has been reported to exhibit selective activity against prostate cancer cells.14
Although the roots of this medicinal plant have been reported in numerous studies, information regarding the bioactive potential of its leaves, stems, and branches remains limited. Moreover, no research to date has evaluated its antidiabetic potential. Therefore, this study was conducted to investigate the in vitro antidiabetic and antiproliferative activities of S. juventas extracts, thereby providing valuable insights into these underexplored plant parts as potential sources of therapeutic agents for cancer and diabetes.
Experimental
General experimental procedures – HepG2 (human hepatocellular carcinoma), B16-F10 (murine melanoma), and Hs68 (human foreskin fibroblast) cell lines were generously provided by the Department of Biochemistry and Molecular Biology, School of Medicine, Kyung Hee University (Republic of Korea). Dulbecco’s Modified Eagle Medium (DMEM) and fetal bovine serum (FBS) were obtained from Gibco®, Thermo Fisher Scientific (USA). Phosphate-buffered saline (PBS), trypsin-EDTA solution, and dimethyl sulfoxide (DMSO) were purchased from Merck (Germany). Vinblastine, α-glucosidase enzyme, and p-nitrophenyl-α-D-glucopyranoside (pNPG) were supplied by Sigma-Aldrich (USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and 4′,6-diamidino-2-phenylindole (DAPI) were purchased from Thermo Fisher Scientific (USA).
Plant material – The aerial parts (stems and leaves) of Streptocaulon juventas (Sj) were collected from Long Khanh, Dong Nai Province, Vietnam, and taxonomically authenticated by Dr. Huynh Loi (Binh Duong University). The plant material was shade-dried at 50°C and then ground into a fine powder. A voucher specimen (Sj-VN-0224) has been deposited in the Department of Pharmacognosy, University of Health Sciences, Vietnam National University, Ho Chi Minh City.
Extraction of plant material – Fifty grams of dried aerial parts were ground and sequentially extracted using an ultrasound-assisted hot extraction method with n-hexane (300 mL), dichloromethane (300 mL), ethyl acetate (300 mL), and methanol (300 mL). The obtained extracts were evaporated under reduced pressure to yield dry crude extracts, which were stored at 5°C until further use.
Cell culture – Cell lines were cultured in DMEM supplemented with 10% FBS and 1% antibiotic-antimycotic. Stocks were stored in liquid nitrogen vapor phase and thawed by gentle agitation in a 37°C water bath for approximately 2 minutes. All subsequent procedures were performed under aseptic conditions in a Class II biosafety cabinet. Cells were transferred to 60 mm petri dishes, diluted with culture medium, and incubated at 37°C in 5% CO2. To remove the cryoprotectant or concentrate cells, suspensions were centrifuged at 1000 rpm for 5 minutes, supernatants discarded, and pellets resuspended in fresh medium. Dilution ratios were adjusted for each cell line. For sub-culturing, the medium was removed, and cells were washed twice with PBS, followed by treatment with 500 µL trypsin-EDTA for 5 minutes. Cells were resuspended in fresh medium and seeded into new dishes at a 1:4 ratio. Sub-culturing was performed every 2–3 days.
Preparation of extracts – Extracts were freshly dissolved in 10% DMSO (Merck), then diluted with culture medium and used immediately. The samples were labeled as Sj-Hx (hexane), Sj-DCM (dichloromethane), Sj-EA (ethyl acetate), and Sj-MeOH (methanol).
Cell viability assay – Cell viability was assessed based on metabolic activity using the MTT assay. Cells were seeded into 96-well plates at a density of 5 × 103 cells/well in 100 μL of culture medium. After 24 hours of incubation, 5 μL of extract or control solution was added to achieve a final concentration of 50 μg/mL. Vinblastine (5 μM) and 10% DMSO were used as positive and negative controls, respectively. Following 48 hours of treatment, the medium was replaced with 100 μL of 10% MTT solution in DMEM, and the cells were incubated for 30 minutes. Formazan crystals were solubilized with 75 μL of DMSO:PBS (2:1), and absorbance was measured at 540 nm using a microplate reader (BioTek, USA).
To determine IC50 values, cells were treated with six concentrations of the extract (5–100 μg/mL). All experiments were performed in triplicate.
Apoptotic nuclear morphology assay – Apoptotic morphology was assessed using DAPI staining. Coverslips were placed in 24-well plates, and 0.05 × 106 cells/well were seeded and treated with 30 μg/mL of Sj-Hx or Sj-DCM extracts, while 10% DMSO served as control. After 24 hours, cells were fixed with 1% formalin, washed, and stained with DAPI (5 μL). Slides were sealed with nail polish and examined under a fluorescence microscope for nuclear fragmentation and apoptotic features.
α-Glucosidase inhibitory assay – The α-glucosidase inhibitory activity was evaluated with slight modifications based on the protocol described by Hua-Qiang Dong et al.15 Briefly, 60 μL of the test sample or acarbose (positive control) was mixed with 50 μL of 0.1 M phosphate buffer (pH 6.8) containing 0.2 U/mL α-glucosidase enzyme in a 96-well microplate. The reaction mixture was pre-incubated at 37°C for 10 minutes. Subsequently, 50 μL of p-nitrophenyl-α-D-glucopyranoside was added as the substrate, and the mixture was further incubated at 37°C for an additional 20 minutes. The enzymatic reaction was terminated, and the absorbance was measured at 405 nm using a microplate reader. All experiments were carried out in triplicate.
Statistical analysis – Data are expressed as mean ± SD from the indicated number of independent experiments. Cell viability (%) was calculated, and statistical significance was assessed using t-test and ANOVA (p < 0.05).
Results and Discussion
Preliminary cytotoxic screening of S. juventas extracts at 50 μg/mL demonstrated significant antiproliferative effects against both B16F10 melanoma and HepG2 hepatocellular carcinoma cell lines.
In the B16F10 cell line, the hexane extract (Sj-Hx) and dichloromethane extract (Sj-DCM) inhibited cell proliferation by 53% and 45%, respectively. The methanol extract (Sj-Me) exhibited moderate activity (25%), whereas the ethyl acetate extract (Sj-EA) demonstrated only minimal inhibition (3%) (Fig. 1). Remarkably, the cytotoxicity of Sj-Hx at this concentration was comparable to that of vinblastine (5 μM), a standard chemotherapeutic agent. Consequently, Sj-Hx and Sj-DCM were selected for further dose-response analysis.
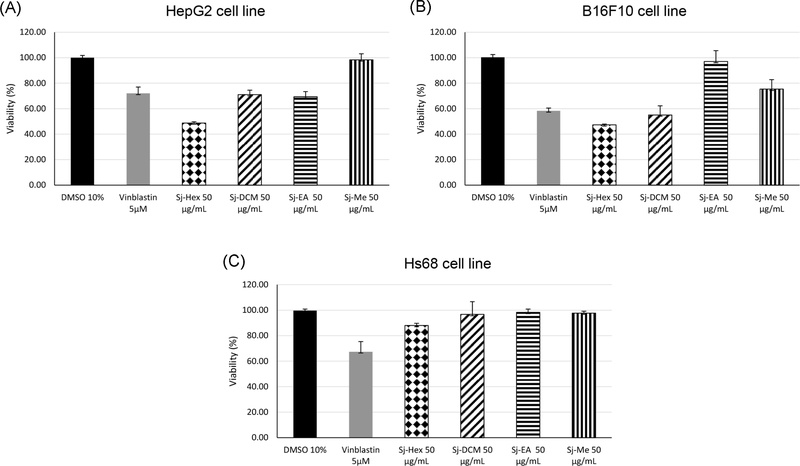
Cell viability of HepG2 (A), B16F10 (B) and Hs68 (C) cell lines after 48-hour treatment with Streptocaulon juventas extracts (50 μg/mL). The extracts showed selective cytotoxicity against cancer cells while sparing normal cells.
In B16F10 cells treated with Sj-Hx and Sj-DCM extracts across a concentration range of 5–100 μg/mL, dose-dependent cell death was observed. Nonlinear regression analysis yielded IC50 values of 36.09 ± 0.7 µg/mL (Sj-Hx) and 45.91 ± 0.63 µg/mL (Sj-DCM), indicating moderate cytotoxic potential according to established phytochemical screening standards.16
In the HepG2 cell line, the Sj-Hx extract exhibited the strongest activity, inhibiting cell proliferation by 51% at a concentration of 50 µg/mL, whereas Sj-DCM and Sj-EA demonstrated moderate effects of 29% and 31%, respectively, and the methanol extract (Sj-Me) showed negligible activity (2%) (Fig. 1). Accordingly, the Sj-Hx extract was selected for IC50 determination in the HepG2 cell line. The results revealed dose-dependent cytotoxicity when treated across the concentration range of 5–100 µg/mL.
The IC50 value for Sj-Hx was determined to be 36.65 ± 3.11 µg/mL (Table 1). According to the U.S. National Cancer Institute (NCI), crude extracts with an IC50 value around 30 µg/mL are considered promising candidates for drug development.17 Additionally, some authors suggest that plant extracts with IC50 values below 100 µg/mL may also be regarded as potential candidates.18 Therefore, based on the findings of this study, Streptocaulon juventas demonstrates strong potential as a promising candidate for the development of plant-derived anti-cancer agents.
Overall, these findings indicate that the low-polarity fractions of Streptocaulon juventas, particularly Sj-Hx and Sj-DCM, possess selective and moderate cytotoxic activity against both melanoma and hepatocellular carcinoma cell lines. The consistent dose-dependent inhibition and characteristic apoptotic morphology support the hypothesis that lipophilic compounds present in these fractions are likely responsible for the observed anticancer effects. These extracts are candidates for further studies aimed at chemical profiling and the isolation of potential bioactive compounds.
To evaluate whether these extracts exert cytotoxic effects on normal cells, human fibroblast Hs68 cells were used and treated with the extracts at a concentration of 50 µg/mL. Morphological assessments showed that the cells remained intact with an elongated shape across all treated groups. The MTT assay revealed no significant cell death (Fig. 1). These findings confirm that the extracts are non-toxic to normal cells, indicating a high degree of selectivity toward malignant cells.
In the Hs68 cell line, at the tested concentration of 50 µg/mL, all hexane, dichloromethane, ethyl acetate, and methanol extracts did not induce cell death, with cell viabilities remaining above 90%, comparable to the 10% DMSO control. This indicates that these extracts are non-toxic to normal human fibroblasts. The results are shown in Fig. 1.
In contrast, in cancer cell lines such as B16F10 and HepG2, these extracts markedly reduced cell viability. The hexane extract exhibited the strongest cytotoxicity, reducing cell viability to approximately 50% in B16F10 cells and even lower in HepG2 cells. The DCM and EtOAc extracts also demonstrated moderate cytotoxic effects on these cancer cell lines.
These findings indicate a selective cytotoxic effect of S. juventas extracts, particularly the hexane and DCM fractions on cancer cells while sparing normal cells. This selective behavior is a desirable feature for potential anti-cancer agents, minimizing side effects on healthy tissues. This result is consistent with recent studies highlighting the therapeutic potential of plant-derived compounds exhibiting selective cytotoxicity.
To evaluate whether the cytotoxic effects of the extracts were associated with programmed cell death, B16F10 and HepG2 cells were treated with the hexane (Sj-Hx) and dichloromethane (Sj-DCM) extracts at a concentration of 30 µg/mL, followed by DAPI staining. Nuclear morphological changes were then examined under a fluorescence microscope, and the findings are presented in Fig. 2.
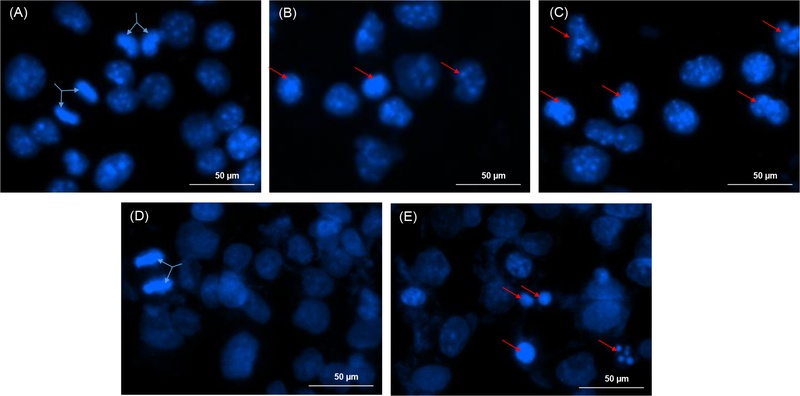
Nuclear morphology of B16F10 and HepG2 cells treated with Sj-Hex and Sj-DCM extracts (30 µg/mL) visualized by DAPI staining. (A, D) Negative control (10% DMSO) for B16F10 and HepG2 cells, respectively. (B, E) Cells treated with Sj-Hex extract. (C) B16F10 cells treated with Sj-DCM extract. Blue arrows indicate dividing cells; red arrows indicate apoptotic nuclei.
It is evident that B16F10 and HepG2 cells maintained intact nuclear morphology and uniform chromatin distribution when treated with negative control, 10% DMSO (Fig. 2A and 2D). The blue arrows indicate cells undergoing mitotic division, reflecting active proliferation and normal cellular development. However, upon treatment with the Sj-Hx extract, both B16F10 (Fig. 2B) and HepG2 (Fig. 2E) cells exhibited chromatin condensation, nuclear fragmentation, and reduced nuclear size (as indicated by the red arrows). These morphological alterations are characteristic of early and late stages of apoptosis, suggesting that the extracts likely induced programmed cell death in these cancer cells.
Similarly, treatment with the Sj-DCM extract (Fig. 2C) resulted in a greater reduction in cell number compared with the Sj-Hx-treated group, along with more pronounced nuclear disintegration and chromatin condensation. These observations indicate a more evident apoptotic process in cells treated with the Sj-DCM extract.
Thus, the observed results indicate that both extracts induce apoptosis in B16F10 and HepG2 cells, with this effect being particularly pronounced in the Sj-DCM extract.
The greater morphological damage and reduced cell number in the Sj-DCM-treated group may reflect a higher cytotoxic potential of this extract. These findings align with prior research highlighting the ability of plant-derived compounds to induce apoptosis in cancer cells via chromatin condensation and nuclear fragmentation.19–21
The results indicated that, except for the methanolic extract, all other extracts exhibited α-glucosidase inhibitory activity. Significantly, the hexane (Sj-Hx) and ethyl acetate (Sj-EA) extracts demonstrated the most pronounced inhibition, with IC50 values of 221.59 and 265.73 µg/mL, respectively, which were lower than that of the positive control, acarbose (IC50 = 303.10 µg/mL) under the same experimental conditions (Table 2). This study is the first to demonstrate the α-glucosidase inhibitory activity of Streptocaulon juventas extracts. These findings highlight S. juventas as a promising medicinal plant for further exploration in the discovery of novel antidiabetic compounds.
In conclusion, this study indicates that, in addition to the roots, which have been extensively investigated in previous research, the leaves of Streptocaulon juventas also warrant in-depth scientific exploration. Notably, in the evaluation of anti-proliferative activity, the extracts demonstrated selective cytotoxic effects. The hexane (SjHx) and dichloromethane (Sj-DCM) extracts exhibited minimal toxicity toward normal fibroblasts (Hs68) while exerting significant cytotoxic effects on cancer cell lines (B16F10 and HepG2). DAPI staining further revealed morphological and nuclear alterations consistent with programmed cell death (apoptosis). Additionally, the hexane extract displayed superior α-glucosidase inhibitory activity compared with the positive control, underscoring its therapeutic potential. Collectively, these findings highlight the leaves of S. juventas as a valuable phytochemical resource for the isolation of novel bioactive compounds, offering promising prospects for the discovery of anticancer and antidiabetic agents in future investigations.
Acknowledgments
This research was supported by the Vietnam National University (Project No. 36-2025-44-01).
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. Cancer 2021, 127, 3029–3030.
[https://doi.org/10.1002/cncr.33587]

-
Ma, C.-X.; Ma, X.-N.; Guan, C.-H.; Li, Y.-D.; Mauricio, D.; Fu, S.-B. Cardiovasc. Diabetol. 2022, 21, 74.
[https://doi.org/10.1186/s12933-022-01516-6]

-
Newman, D. J.; Cragg, G. M. J. Nat. Prod. 2020, 83, 770–803.
[https://doi.org/10.1021/acs.jnatprod.9b01285]

-
Cragg, G. M.; Newman, D. J. J. Ethnopharmacol. 2005, 100, 72–79.
[https://doi.org/10.1016/j.jep.2005.05.011]

-
Nauck, M. A.; Wefers, J.; Meier, J. J. Lancet Diabetes Endocrinol. 2021, 9, 525–544.
[https://doi.org/10.1016/S2213-8587(21)00113-3]

-
Alam, S.; Sarker, M. M. R.; Sultana, T. N.; Chowdhury, M. N. R.; Rashid, M. A.; Chaity, N. I.; Zhao, C.; Xiao, J.; Hafez, E. E.; Khan, S. A. Front. Endocrinol. 2022, 13, 800714.
[https://doi.org/10.3389/fendo.2022.800714]

- Nguyen, Q.-V.; Eun, J.-B. J. Med. Plants Res. 2011, 5, 2798–2811.
- Chi, V. V. Dictionary of Vietnamese medicinal plants; Publishing House Medicine: Vietnam 2012; p 2.
-
Ueda, J.-Y.; Tezuka, Y.; Banskota, A. H.; Tran, Q. L.; Tran, Q. K.; Saiki, I.; Kadota, S. J. Nat. Prod. 2003, 66, 1427–1433.
[https://doi.org/10.1021/np030177h]

-
Li, Y.; Feng, X.; Qiu, F. Chem. Biodivers. 2024, 21, e202301585.
[https://doi.org/10.1002/cbdv.202302050]

-
Han, N.; Yang, J.; Li, L.; Xiao, B.; Sha, S.; Tran, L.; Yin, J.; Wu, C. Planta Med. 2010, 76, 561–565.
[https://doi.org/10.1055/s-0029-1240600]

-
Xue, R.; Han, N.; Xia, M.; Ye, C.; Hao, Z.; Wang, L.; Wang, Y.; Yang, J.; Saiki, I.; Yin, J. Steroids 2015, 94, 51–59.
[https://doi.org/10.1016/j.steroids.2014.12.015]

-
Ueda, J.-Y.; Tezuka, Y.; Banskota, A. H.; Tran, Q. L.; Tran, Q. K.; Harimaya, Y.; Saiki, I.; Kadota, S. Biol. Pharm. Bull. 2002, 25, 753–760.
[https://doi.org/10.1248/bpb.25.753]

-
Anaya-Eugenio, G. D.; Addo, E. M.; Ezzone, N.; Henkin, J. M.; Ninh, T. N.; Ren, Y.; Soejarto, D. D.; Kinghorn, A. D.; Carcache de Blanco, E. J. J. Nat. Prod. 2019, 82, 1645–1655.
[https://doi.org/10.1021/acs.jnatprod.9b00140]

-
Dong, H.-Q.; Li, M.; Zhu, F.; Liu, F.-L.; Huang, J.-B. Food Chem. 2012, 130, 261–266.
[https://doi.org/10.1016/j.foodchem.2011.07.030]

- Atanasov, A. G.; Zotchev, S. B.; Dirsch, V. M.; Supuran, C. T. Nat. Rev. Drug Discov. 2021, 20, 200–216.
-
Kola, P.; Manjula, S. N.; Metowogo, K.; Madhunapantula, S. V.; Eklu-Gadegbeku, K. Heliyon 2023, 9, e13869.
[https://doi.org/10.1016/j.heliyon.2023.e13869]

-
Canga, I.; Vita, P.; Oliveira, A. I.; Castro, M. Á.; Pinho, C. Molecules 2022, 27, 4989.
[https://doi.org/10.3390/molecules27154989]

-
Elmore, S. Toxicol. Pathol. 2007, 35, 495–516.
[https://doi.org/10.1080/01926230701320337]

-
Carneiro, B. A.; El-Deiry, W. S. Nat. Rev. Clin. Oncol. 2020, 17, 395–417.
[https://doi.org/10.1038/s41571-020-0341-y]

-
Fulda, S. Int. J. Cell Biol. 2010, 2010, 370835.
[https://doi.org/10.1155/2010/370835]

