
Poria cocos Extract Protects Feline Kidney Epithelial Cells from Oxidative Damage and Promotes Cell Growth
Abstract
Reactive oxygen species (ROS), particularly hydrogen peroxide (H2O2), are critical mediators of oxidative stress, which can lead to cellular damage and contribute to various kidney diseases. Poria cocos, a traditional medicinal fungus, has demonstrated various health benefits, particularly in gastrointestinal and neurological health. This study investigates the potential of P. cocos extract (PCE) to reduce H2O2-induced oxidative injury and promote cell proliferation in feline kidney epithelial cells. PCE treatment significantly improved cell viability in H2O2-injured cells in a dose-dependent manner. PCE reduced intracellular ROS levels, including superoxide anions, hydroxyl radicals, and intracellular H2O2, while enhancing the activities of antioxidant enzymes such as superoxide dismutase and catalase. Furthermore, PCE treatment promoted cell proliferation, as evidenced by increased Ki-67 expression, and enhanced the phosphorylation of ERK1/2 and AKT, which are key mediators of cell survival and proliferation. PCE effectively mitigates oxidative stress and promotes cell proliferation in kidney epithelial cells through the activation of the ERK1/2 and AKT pathways, suggesting its potential as a therapeutic agent for oxidative stress-related kidney diseases.
Keywords:
Poria cocos, Reactive oxygen species, Kidney epithelial cell, Cell proliferation, Antioxidant enzymeIntroduction
Reactive oxygen species (ROS) are reactive molecules that are naturally generated during cellular metabolism, particularly in the mitochondria.1 They play a vital role in cell signaling and maintaining homeostasis.1 However, excessive levels of ROS can lead to oxidative stress, resulting in damage to proteins, lipids, and DNA, which contributes to the development of various diseases.2 Among the different types of ROS, hydrogen peroxide (H2O2) is notable for its stability and ability to permeate cell membranes.3 It is formed through the enzyme-catalyzed or spontaneous dismutation of superoxide anions.4 H2O2 has been extensively utilized to investigate mechanisms of tissue damage and cell death.4 Both exogenous and endogenous H2O2 serve as critical mediators of kidney tubular injury in conditions such as ischemia and reperfusion injury,5 cisplatin nephrotoxicity,6 and ureteral obstruction.7 In these scenarios, kidney parenchymal cells in the tubules are more susceptible to injury and cell death compared to tubulointerstitial cells.8,9 Consequently, exogenous H2O2 is frequently employed to induce ROS-mediated oxidative damage in kidney tubule epithelial cells.9 Understanding the mechanisms underlying H2O2-induced tubular injury is essential for advancing research and treatment of kidney diseases.
Chronic kidney disease (CKD) represents one of the leading causes of illness and death in domestic cats, particularly in aging populations, yet effective curative therapies remain limited.10,11 The progressive and irreversible nature of feline CKD underscores the urgent need for safe and innovative therapeutic approaches, including interventions derived from natural products with renoprotective potential. Poria cocos, a medicinal fungus with a long history in traditional Chinese medicine, is renowned for its diverse pharmacological properties, including anti-inflammatory and immunomodulatory effects.12,13 Recent studies highlight its potential to protect various cell types from oxidative stress-related damage,14 suggesting a role in enhancing kidney health. P. cocos extract (PCE) has demonstrated beneficial effects in hyperuricemic nephropathy, with research indicating their ability to improve kidney function.15 Notably, P. cocos nanoparticles have shown effectiveness in reducing oxidative stress and apoptosis in kidney tissues, while bioactive compounds from the PCE, including poricoic acid A, have been found to inhibit kidney fibrosis.16,17 Although these findings are promising, further research is necessary to fully elucidate the ROS-dependent mechanisms by which PCE exerts its protective effects and to optimize its therapeutic applications in kidney diseases. Therefore, the objective of this study was to determine whether treatment with PCE could influence H2O2-induced cell death in kidney tubular cells and whether treatment with PCE following H2O2 injury would promote cell proliferation.
Experimental
Extraction of P. cocos – Dried P. cocos was collected from Jecheon, Chungbuk, South Korea, in March 2024. A voucher specimen (JNUPe-2024-21) is maintained at the College of Pharmacy, Jeju National University. The P. cocos powder was extracted three times using 70% ethanol with an ultrasonicator (Powersonic 520; Hwashintech, Seoul, Republic of Korea) for 30 minutes each time. After extraction, the mixture was centrifuged at 4,500 rpm for 5 minutes using a benchtop centrifuge (Avanti J-15R; Beckman Coulter, Brea, CA, USA) and filtered through filter paper. This procedure was repeated three times. The combined supernatants were concentrated using a reduced-pressure rotary evaporator (Heidolph, Schwabach, Germany) and stored at 4°C until analysis.
Cell culture and treatment – The Crandell-Rees feline kidney (CRFK) cell line (KCLB, Seoul, Korea; product no. 10094) was cultured in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum at 37°C in a 5% CO2 atmosphere. Upon reaching 80% confluence in the culture dishes, the culture medium was replaced with a serum-free medium. Subsequently, the cells were treated with 10 to 100 μg of PCE per mL of culture medium (vehicle) for 60 minutes. They were then exposed to 400 μM H2O2 (Sigma-Aldrich, St. Louis, MO, USA; product no. H1009) or distilled water (control) for 6 hours. To investigate the role of PCE during recovery following H2O2-induced injury, the cells were treated with PCE for 18 hours after the 6-hour exposure to H2O2.
Cell viability – A yellow water-soluble tetrazolium dye, thiazolyl blue tetrazolium bromide (MTT; Biosesang, Seongnam, Korea; product no. M1029), was used to assess cell viability, as previously described.18,19 Following the removal of the culture medium, CRFK cells in 24-well plates were incubated at 37°C with 300 μL of 5 mg/mL MTT in phosphate-buffered saline (PBS) for 30 minutes. After discarding the MTT solution, 300 μL of dimethyl sulfoxide (DMSO) was added to each well and incubated for an additional 5 minutes. To quantify the purple-colored formazan product, the absorbance of 100 μL of the solution from the wells treated with DMSO was measured on a 96-well plate at a wavelength of 595 nm, with a reference wavelength of 620 nm, using the SpectraMax i3x multi-mode microplate reader (Molecular Devices, San Jose, CA, USA) in the Bio-Health Materials Core-Facility at Jeju National University.
Superoxide anion and hydroxyl radical levels – The fluorescent dyes dihydroethidium (DHE; Sigma-Aldrich; product no. 37291) and 2ʹ,7ʹ-dichlorofluorescin diacetate (DCFDA; Sigma-Aldrich; product no. D6883) were used to evaluate the production of intracellular superoxide anions and hydroxyl radicals, respectively.20,21 CRFK cells cultured in 60 mm dishes were detached using 0.05% trypsin-ethylenediaminetetraacetic acid and subsequently incubated with 1 μM DHE and 10 μM DCFDA for 30 minutes. Following isolation into a single-cell population, the fluorescences of DHE and DCFDA were detected through the phycoerythrin-area (PE-A) and fluorescein isothiocyanate-area (FITC-A) channels, respectively, using the CytoFLEX flow cytometer and CytExpert software obtained from Beckman Coulter (Indianapolis, IN, USA). The single-cell population was defined by side scatter-height versus (vs.) side scatter-width, followed by forward scatter-height vs. forward scatter-width in the flow cytometer.
Intracellular H2O2 concentration – The intracellular H2O2 concentration was measured using a commercial kit (Northwest Life Science Specialties; product no. NWK-HYP01) in accordance with the manufacturer’s instructions.9,22 This assay relies on the formation of a complex between xylenol orange and ferric iron, which is generated through the peroxide-dependent oxidation of ferrous iron. The results were normalized to micromoles of hydrogen peroxide per milligram of protein.
Enzyme activity – CRFK cells were maintained on ice during sample preparation and assay. The protein concentration in the homogenates was determined using Bradford reagent (Sigma-Aldrich; product no. B6916) and subsequently normalized to units per milligram of protein for enzyme activity assessment. Catalase and superoxide dismutase (SOD) activities were measured using commercial kits (Northwest Life Science Specialties, Vancouver, WA, USA; product no. NWK-CAT01 and Cayman Chemical, Ann Arbor, MI, USA; product no. 706002, respectively) in accordance with the manufacturer’s instructions.23,24 These assays are based on the methodologies established by Beers25 and Marklund,26 with modifications implemented to enhance robustness and convenience. One unit (U) of catalase is defined as the amount of enzyme that decomposes 1.0 mmol of hydrogen peroxide per minute at 25°C. One unit (U) of SOD is defined as the amount of enzyme required to achieve 50% dismutation of the superoxide radical, as measured by the change in absorbance per minute at 25°C. The absorbances in the assays were measured using a Cole-Parmer spectrophotometer at 240 nm and 450 nm for catalase and SOD activities, respectively.
Cell proliferation – The proliferation of CRFK cells was quantified by the mean value of Ki-67-positive cells per normalized cell count (10,000 events per experiment) using flow cytometry analysis.27 CRFK cells were detached from 60 mm dishes using 0.05% trypsin-EDTA, fixed in ice-cold 70% ethanol, and stained with Alexa Fluor 488 anti-Ki-67 antibody (Abcam, Cambridge, MA, USA; product no. ab206633). The cells were then isolated into a single-cell population and gated in FITC-A using the CytoFLEX flow cytometer and CytExpert software, both obtained from Beckman Coulter (Indianapolis, IN, USA).
Western blot analysis – Electrophoresis of proteins in cell lysate was conducted using 7.5% polyacrylamide gels prepared from the TGX FastCast acrylamide kit (Bio-Rad Laboratories, Hercules, CA, USA; product no. 1610171) in running buffer (Bio-Rad Laboratories; product no. TR2015-100-00) and subsequently transferred to a polyvinylidene fluoride membrane in transfer buffer (Bio-Rad Laboratories; product no. TR2028-100-00), as previously described.28,29 Following the transfer, the membrane was blocked with 5% skim milk and then incubated with antibodies against phosphorylated extracellular signal-regulated kinase 1/2 (p-ERK1/2; 1:2,500 dilution; Cell Signaling; product no. 4370), ERK1/2 (1:2,500 dilution; Santa Cruz Biotechnology, Santa Cruz, CA, USA; product no. sc-93), phosphorylated protein kinase B (p-AKT; 1:2,500 dilution; ABclonal, Woburn, MA, USA; product no. AP0637), AKT (Santa Cruz Biotechnology; product no. sc-8312), and β-actin (1:5,000 dilution; Santa Cruz Biotechnology; product no. sc-47778). Subsequently, either peroxidase-conjugated anti-rabbit IgG antibody (1:5,000 dilution; Vector Laboratories, Burlingame, CA, USA; product no. WB-1000) or peroxidase-conjugated anti-mouse IgG antibody (1:5,000 dilution; Vector Laboratories; product no. WB-2000) was applied. Protein expression bands were detected using the Azure c300 imaging system (Azure Biosystems, Dublin, CA, USA) and quantified with the AzureSpot analysis software (Azure Biosystems).
Chemical profiling of PCE by LC-QToF mass spectrometry – The molecular formulas and chemical constituents of PCE were analyzed using a high-resolution QToF mass spectrometer (G6546A; Agilent Technologies, Santa Clara, CA, USA) equipped with an electrospray ionization (ESI) source incorporating Jet Stream technology. The mobile phases consisted of water with 0.1% formic acid (A) and acetonitrile with 0.1% formic acid (B) (Merch, Darmstadt, Germany). The gradient program was set as follows: 5% B initially, increased to 35% B over 5 minutes, then to 75% B over the next 50 minutes, followed by 95% B over 5 minutes, and maintained at 95% B for the final 8 minutes. Analytical conditions were as follows: flow rate, 0.8 mL/minute; injection volume, 5 µL; drying gas, nitrogen; gas temperature, 300°C; gas flow rate, 8 L/minute; nebulizer pressure, 40 psi; sheath gas temperature, 350°C; sheath gas flow rate, 11 L/minute; capillary voltage, 3,500 V; fragmentor voltage, 150 V; skimmer voltage, 65 V; and octopole radiofrequency (Oct RF), 750 V. Internal mass calibration was performed using m/z 112.9856 (deprotonated trifluoroacetic acid, TFA) and m/z 1033.9881 (TFA-adducted HP-921) in negative ion mode to ensure accurate mass measurements. Tentative identification of the constituents was based on the accurate mass data. Data acquisition and analysis were conducted using Agilent MassHunter Acquisition Software (v. A.05.00), and each sample was analyzed in negative ion mode over an m/z range of 100–1100.
Statistical analysis – Numeric data were analyzed using Sigma Plot 14.0 (Systat Software, San Jose, CA, USA), as previously described.28 The normality of the data was assessed using the Shapiro-Wilk test, followed by the Brown-Forsythe test to evaluate equal variance. Parametric data were analyzed using one-way or two-way analyses of variance (ANOVA), followed by the Holm-Sidak post hoc test. Non-parametric data were analyzed using the Kruskal-Wallis H test, followed by the Student-Newman-Keuls post hoc test. For comparisons between two groups, an unpaired Student’s t test was used when both normality and equal variance assumptions were met. In graphical representations, parametric data are presented as means ± standard error of the mean with individual data points, while non-parametric data are shown as median values with quartiles. A significance level of p < 0.05 was considered statistically significant. The data availability of all raw numeric data and corresponding statistical results is ensured.
Results and Discussion
The MTT assay revealed that H2O2 exposure for 6 hours significantly decreased CRFK cell viability to 58.1% (Fig. 1). Treatment with PCE significantly mitigated this reduction in a dose-dependent manner, with 30 and 100 μg/mL of PCE showing higher viability compared to lower concentrations (p = 0.019 and 0.026, respectively). This finding indicates a dose-dependent protective effect of PCE against H2O2-induced injury. Notably, treatment with 100 μg/mL of PCE also enhanced cell viability in the non-H2O2-exposed control group. These findings align with previous studies demonstrating the protective effects of PCE in neuronal cells exposed to β-amyloid-induced injury30 and in mouse kidney collecting duct cells subjected to hypertonic stress,31 suggesting that PCE may promote kidney epithelial cell health under both stressed and normal conditions.
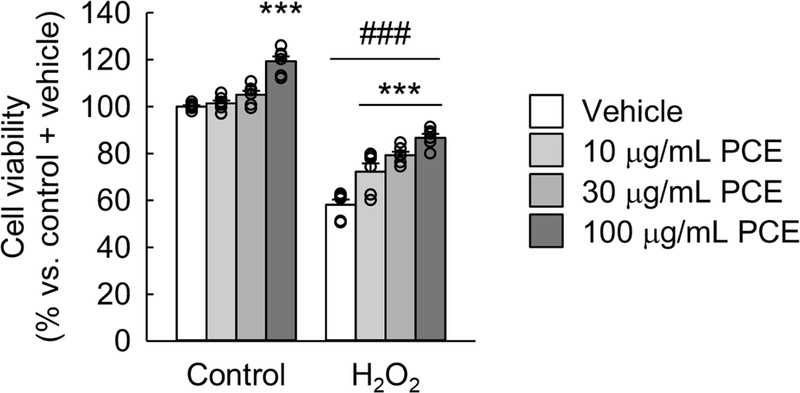
Treatment with PCE enhances cell viability in feline kidney epithelial cells. CRFK cells were pre-treated with PCE at concentrations of 10, 30, and 100 μg/mL for 60 minutes, followed by a 6-hour exposure to 400 μM H2O2 (n = 6). Cell viability was assessed using the MTT assay. A two-way ANOVA revealed significant effects of H2O2 (F = 505.621, p < 0.001), PCE treatment (F = 48.888, p < 0.001), and their interaction (F = 5.800, p = 0.002). ***p < 0.001 vs. vehicle; ###p < 0.001 vs. control, as determined by the Holm-Sidak post hoc test.
Flow cytometric analyses showed that H2O2 exposure significantly increased superoxide anion and hydroxyl radical levels by 1.9-fold and 1.8-fold, respectively, compared to control (Fig. 2A, and 2B). PCE treatment markedly attenuated these increases. Furthermore, PCE significantly reduced hydroxyl radical and intracellular H2O2 levels even in non-exposed cells (Fig. 2B, and 2C), indicating that it may contribute to maintaining redox homeostasis under physiological conditions. Similar antioxidant effects of PCE have been reported in other cell types, including reduction of hydroxyl radical production in high glucose-exposed rat mesangial cells,32 ox-LDL-induced ROS in vascular smooth muscle cells,33 and ROS in alcohol-exposed liver34 and sepsis-exposed spleen tissues.35
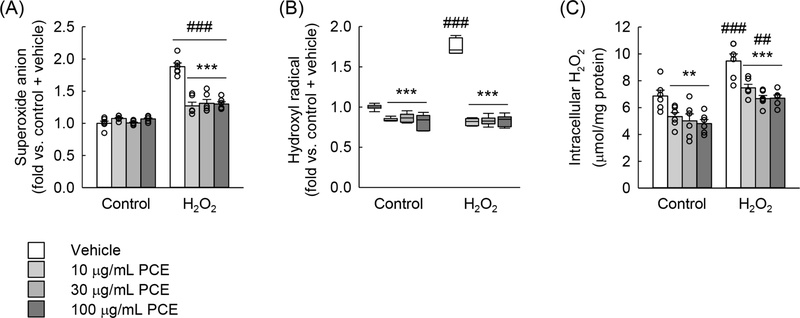
Treatment with PCE reduces oxidative stress induced by H2O2 exposure in feline kidney epithelial cells. CRFK cells were pre-treated with PCE at concentrations of 10, 30, and 100 μg/mL for 60 minutes, followed by a 6-hour exposure to 400 μM H2O2 (n = 6). (A) Superoxide anion levels were assessed using DHE and flow cytometry. A two-way ANOVA revealed significant effects of H2O2 (F = 215.184, p < 0.001), PCE treatment (F = 14.737, p < 0.001), and their interaction (F = 26.045, p < 0.001). (B) Hydroxyl radical levels were determined with DCFDA and flow cytometry. The Kruskal-Wallis H test showed a significant effect among groups (H = 23.138, p ≤ 0.001). (C) Intracellular H2O2 concentrations were quantified using a commercial kit. Significant effects were observed in a two-way ANOVA for H2O2 (F = 62.217, p < 0.001) and PCE treatment (F = 18.184, p < 0.001), but no significant interaction between the two factors (F = 0.614, p = 0.610) was found. **p < 0.01, ***p < 0.001 vs. vehicle; ## p < 0.01, ###p < 0.001 vs. control, as determined by the Holm-Sidak (A, C) or the Student-Newman-Keuls (B) post hoc test.
PCE treatment significantly mitigated the reduction in SOD and catalase activities caused by H2O2 exposure, restoring them to near-control levels (Fig. 3A, and 3B). Additionally, catalase activity was increased by PCE even in the control group, further supporting its role in enhancing intrinsic antioxidant defenses. Previous studies similarly reported that PCE polysaccharides elevate catalase and SOD activities in cryopreserved spermatozoa36 and human dermal fibroblasts exposed to H2O2,37 highlighting its broad antioxidative potential.
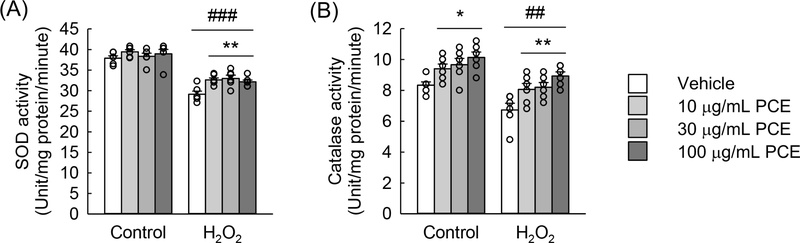
Treatment with PCE enhances the activity of antioxidant enzymes in feline kidney epithelial cells. CRFK cells were pre-treated with PCE at concentrations of 10, 30, and 100 μg/mL for 60 minutes, followed by a 6-hour exposure to 400 μM H2O2 (n = 6). The activities of SOD (A) and catalase (B) were assessed using commercial kits. A two-way ANOVA on the SOD activity data revealed significant effects of H2O2 (F = 198.752, p < 0.001) and PCE treatment (F = 5.338, p = 0.003), but no significant interaction between the two factors (F = 1.996, p = 0.130) was observed. Similarly, a two-way ANOVA on the catalase activity data indicated significant effects of H2O2 (F = 33.988, p < 0.001) and PCE treatment (F = 12.197, p < 0.001), with no significant interaction between the two factors (F = 0.128, p = 0.943). *p < 0.05, **p < 0.01 vs. vehicle; ##p < 0.01, ###p < 0.001 vs. control, as determined by the Holm-Sidak post hoc test.
To assess whether PCE promotes proliferation, we measured Ki-67 expression. Following treatment with 100 μg/mL of PCE for 7 hours, Ki-67 expression was significantly elevated (Fig. 4), suggesting increased proliferative activity. Additionally, after 6 hours of H2O2 exposure and subsequent 18-hour recovery, PCE treatment significantly enhanced cell viability compared to vehicle (Fig. 5). While PCE enhances proliferation in normal and injured kidney epithelial cells, it inhibits proliferation in various cancer cells38-42 and high glucose-exposed mesangial cells,32 suggesting that its effects are highly cell type-dependent.
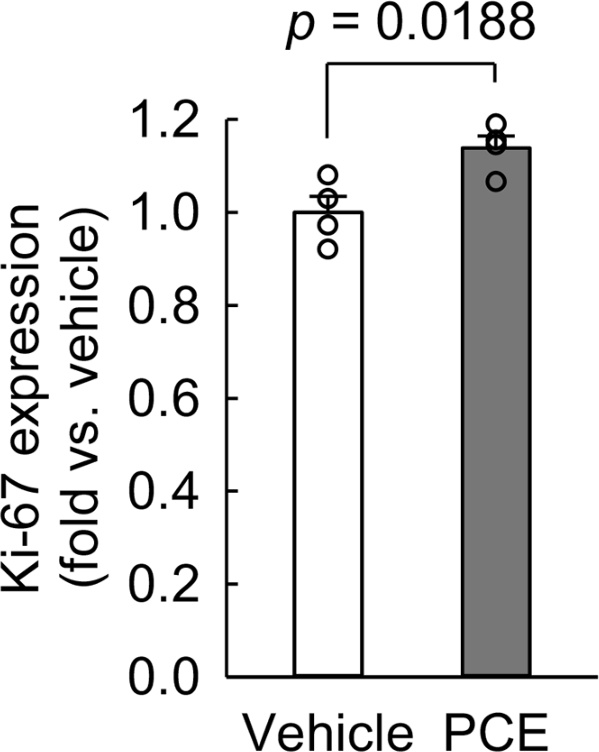
Treatment with PCE enhances the proliferation of feline kidney epithelial cells. CRFK cells were treated with PCE at a concentration of 100 μg/mL for 7 hours (n = 4). Cell proliferation was assessed using an anti-Ki-67 antibody and flow cytometry. The student’s t test revealed a significant difference between the two groups (t = -3.192, p = 0.0188).
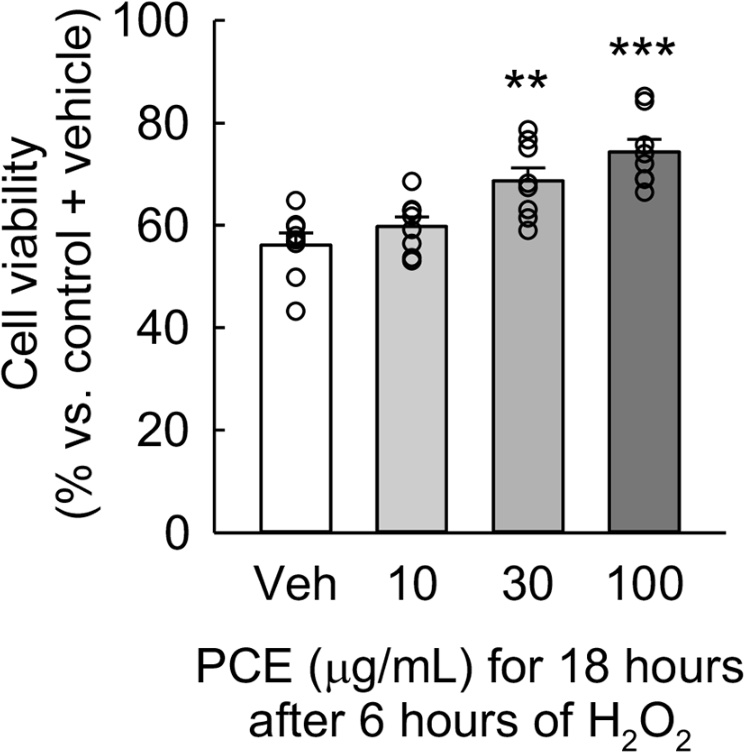
Treatment with PCE enhances the viability of damaged feline kidney epithelial cells during recovery after H2O2 exposure. CRFK cells were exposed to 400 μM H2O2 for 6 hours, followed by treatment with PCE at concentrations of 10, 30, and 100 μg/mL for 18 hours (n = 8). Cell viability, assessed using the MTT assay, demonstrated a significant effect among the groups, as indicated by a one-way ANOVA (F= 12.577, p < 0.001). **p < 0.01, ***p < 0.001 vs. vehicle, as determined by the Holm-Sidak post hoc test.
Western blot analyses demonstrated that treatment with 100 μg/mL of PCE for 7 hours significantly increased the phosphorylation of ERK1/2 and AKT by 1.3-fold and 1.4-fold, respectively (Fig. 6A–C). Activation of these pathways is essential for cell survival, proliferation, and recovery from oxidative injury.43,44 Previous studies have shown that PCE polysaccharides activate ERK/Nrf2/HO-1 signaling in vascular smooth muscle cells,33 modulate ERK1/2 in intestinal barrier function,45 and activate AKT in human keratinocytes.46 Conversely, PCE inhibits ERK1/2 and AKT in certain cancer cells,38,41 further emphasizing its context-dependent activity. The observed activation of ERK1/2 and AKT in CRFK cells suggests that these pathways contribute to the enhanced proliferation and survival following PCE treatment.
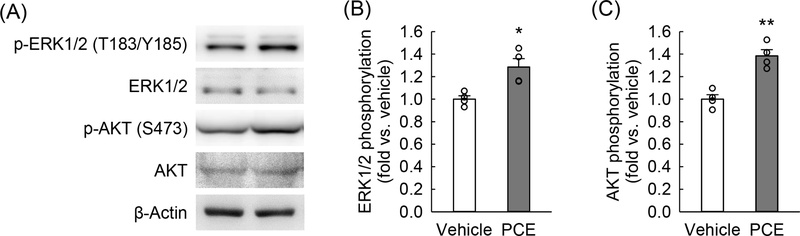
Treatment with PCE enhances the phosphorylation of ERK1/2 and AKT proteins in feline kidney epithelial cells. CRFK cells were treated with PCE at a concentration of 100 μg/mL for 7 hours (n = 4). The phosphorylation levels of ERK1/2 and AKT, as well as the total expressions of these proteins, were assessed using Western blot analysis. β-Actin expression was utilized as a loading control. Student’s t test revealed a significant difference between the two groups in ERK1/2 phosphorylation (t = −3.574, p = 0.0117) and a significant difference in AKT phosphorylation (t = −5.636, p = 0.00134).
UHPLC-QToF-MS analysis of PCE identified five major triterpenoids, and the corresponding MS data are presented in Supplementary Fig. S1. Peaks 1–5 were tentatively assigned as poricoic acids A, BM, or dehydroporicoic acid H; poricoic acids H or GM; 6α-hydroxypolyporenic acid C or 25-hydroxyporicoic acid C; 16α-hydroxytrametenolic acid; and pachymic acid, respectively (Fig. 7, and Table 1), by comparison of their MS data with previously reported literature.47 All five peaks exhibited quasi-molecular ions [M-H]- and [2M-H]-, and their observed molecular formulas were consistent with those of compounds previously reported in P. cocos. These compounds possess documented antioxidant,48 anti-inflammatory,49 and antifibrotic activities,50,51 supporting the observed protective effects. Notably, similar lanostane-type triterpenes are known to activate ERK1/2 and AKT signaling pathways,16,51–53 suggesting that these identified compounds are the bioactive constituents mediating PCE’s cytoprotective and proliferative effects.
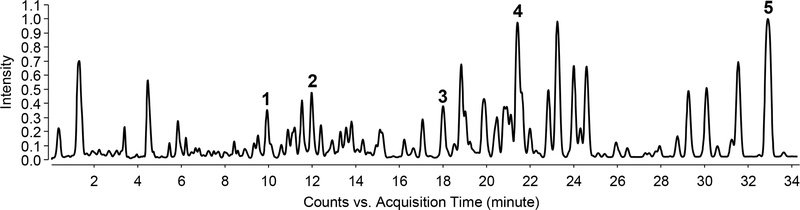
The representative base peak chromatogram of PCE in ESI negative ion mode. The peak number referred to Table 1.
In summary, PCE protects feline kidney epithelial cells from H2O2-induced oxidative damage by reducing ROS, enhancing antioxidant enzyme activities, activating ERK1/2 and AKT signaling pathways, and promoting cell proliferation. Given the pivotal role of oxidative stress in kidney injury and fibrosis, PCE may represent a promising therapeutic candidate for kidney diseases. Further in vivo studies are warranted to confirm its protective effects and elucidate the specific triterpenoids responsible for its bioactivity.
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (RS-2023-00274853, to DM; 2021R1I1A3056002, to JK; RS-2022-NR072037, to JYB).
Conflicts of Interest
The authors declare that they have no competing interests.
References
-
Zorov, D. B.; Juhaszova, M.; Sollott, S. J. Physiol. Rev. 2014, 94, 909–950.
[https://doi.org/10.1152/physrev.00026.2013]

-
Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxid. Med. Cell. Longev. 2017, 2017, 8416763.
[https://doi.org/10.1155/2017/8416763]

-
Bienert, G. P.; Schjoerring, J. K.; Jahn, T. P. Biochim. Biophys. Acta 2006, 1758, 994–1003.
[https://doi.org/10.1016/j.bbamem.2006.02.015]

-
van der Vliet, A.; Janssen-Heininger, Y. M. W. J. Cell. Bioche. 2014, 115, 427–435.
[https://doi.org/10.1002/jcb.24683]

-
Kim, J.; Kim, K. Y.; Jang, H.-S.; Yoshida, T.; Tsuchiya, K.; Nitta, K.; Park, J.-W.; Bonventre, J. V.; Park, K. M. Am. J. Physiol. Renal Physiol. 2009, 296, F622–633.
[https://doi.org/10.1152/ajprenal.90566.2008]

-
Kim, J.; Long, K. E.; Tang, K.; Padanilam, B. J. Kidney Int. 2012, 82, 193–203.
[https://doi.org/10.1038/ki.2012.64]

-
Jung, K.-J.; Kim, J.; Park, Y.-K.; Yoon, Y.-R.; Park, K. M. Food Chem. Toxicol. 2010, 48, 522–529.
[https://doi.org/10.1016/j.fct.2009.11.006]

-
Qi, R.; Yang, C. Cell Death Dis. 2018, 9, 1126.
[https://doi.org/10.1038/s41419-018-1157-x]

-
Kim, J.; Jang, H.-S.; Park, K. M. Am. J. Physiol. Renal Physiol. 2010, 298, F158–F166.
[https://doi.org/10.1152/ajprenal.00474.2009]

-
Polzin, D. J. Vet. Clin. North Am. Small Anim. Pract. 2011, 41, 15–30.
[https://doi.org/10.1016/j.cvsm.2010.09.004]

-
Chakrabarti, S.; Syme, H. M.; Elliott, J. J. Vet. Intern. Med. 2012, 26, 275–281.
[https://doi.org/10.1111/j.1939-1676.2011.00874.x]

-
Qiu, W.-L.; Chao, C.-H.; Lu, M.-K. Int. J. Biol. Macromol. 2024, 279, 135483.
[https://doi.org/10.1016/j.ijbiomac.2024.135483]

-
Sun, M.; Yao, L.; Yu, Q.; Duan, Y.; Huang, J.; Lyu, T.; Yu, N.; Peng, D.; Chen, W.; Wang, Y.; Wang, L.; Zhang, Y. Int. J. Biol. Macromol. 2024, 273, 132931.
[https://doi.org/10.1016/j.ijbiomac.2024.132931]

-
Li, Y.; Wang, P.; Yang, H.; He, J.; Yang, Y.; Tao, Y.; Zhang, M.; Zhang, M.; Yu, J.; Yang, X. Heliyon 2024, 10, e35645.
[https://doi.org/10.1016/j.heliyon.2024.e35645]

-
Liang, D.; Yong, T.; Diao, X.; Chen, S.; Chen, D.; Xiao, C.; Zuo, D.; Xie, Y.; Zhou, X.; Hu, H. Pharm. Biol. 2021, 59, 275–286.
[https://doi.org/10.1080/13880209.2021.1885450]

-
Li, Q.; Ming, Y.; Jia, H.; Wang, G. Exp. Ther. Med. 2021, 21, 289.
[https://doi.org/10.3892/etm.2021.9861]

-
Chen, D.-Q.; Chen, L.; Guo, Y.; Wu, X.-Q.; Zhao, T.-T.; Zhao, H.-L.; Zhang, H.-J.; Yan, M.-H.; Zhang, G.-Q.; Li, P. Acta Pharmacol. Sin. 2023, 44, 1038–1050.
[https://doi.org/10.1038/s41401-022-01026-x]

-
Yu, J.-B.; Padanilam, B. J.; Kim, J. Cells 2024, 13, 1475.
[https://doi.org/10.3390/cells13171475]

-
Yu, J.-B.; Lee, D.-S.; Padanilam, B. J.; Kim, J. Cells 2022, 11, 3472.
[https://doi.org/10.3390/cells11213472]

-
Moon, D.; Yoon, S.-P.; Jang, H.-S.; Noh, M. R.; Ha, L.; Padanilam, B. J.; Kim, J. Anat. Biol. Anthropol. 2019, 32, 121–128.
[https://doi.org/10.11637/aba.2019.32.4.121]

-
Moon, D.; Kim, J. Anat. Cell Biol. 2019, 52, 312–323.
[https://doi.org/10.5115/acb.18.192]

-
Kim, J.; Park, J.-W.; Park, K. M. Am. J. Physiol. Renal Physiol. 2009, 296, F1202–F1211.
[https://doi.org/10.1152/ajprenal.90592.2008]

-
Kim, J.; Seok, Y. M.; Jung, K.-J.; Park, K. M. Am. J. Physiol. Renal Physiol. 2009, 297, F461–F470.
[https://doi.org/10.1152/ajprenal.90735.2008]

-
Kim, J. Arch. Pharm. Res. 2017, 40, 1197–1208.
[https://doi.org/10.1007/s12272-017-0957-3]

-
Beers Jr, R. F.; Sizer, I. W. J. Biol. Chem. 1952, 195, 133–140.
[https://doi.org/10.1016/S0021-9258(19)50881-X]

-
Marklund, S.; Marklund, G. Eur. J. Biochem. 1974, 47, 469–474.
[https://doi.org/10.1111/j.1432-1033.1974.tb03714.x]

-
Moon, D.; Padanilam, B. J.; Park, K. M.; Kim, J. Int. J. Mol. Sci. 2024, 25, 4610.
[https://doi.org/10.3390/ijms25094610]

-
Moon, D.; Padanilam, B. J.; Jang, H.-S.; Kim, J. Pharmacol. Rep. 2022, 74, 1041–1053.
[https://doi.org/10.1007/s43440-022-00403-x]

-
Kim, M.-J.; Moon, D.; Jung, S.; Lee, J.; Kim, J. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 318, R843–R854.
[https://doi.org/10.1152/ajpregu.00130.2019]

- Park, Y.-H.; Son, I. H.; Kim, B.; Lyu, Y.-S.; Moon, H.-I.; Kang, H.-W. Pharmazie 2009, 64, 760–764.
-
Lee, S. M.; Lee, Y. J.; Yoon, J. J.; Kang, D. G.; Lee, H. S. J. Ethnopharmacol. 2012, 141, 368–376.
[https://doi.org/10.1016/j.jep.2012.02.048]

-
Yoon, J. J.; Lee, Y. J.; Lee, S. M.; Jin, S. N.; Kang, D. G.; Lee, H. S. Am. J. Chin. Med. 2013, 41, 71–83.
[https://doi.org/10.1142/S0192415X13500067]

-
Zhao, J.; Niu, X.; Yu, J.; Xiao, X.; Li, W.; Zang, L.; Hu, Z.; Siu-Po Ip, P.; Li, W. Int. Immunopharmacol. 2020, 80, 106173.
[https://doi.org/10.1016/j.intimp.2019.106173]

-
Jiang, Y.-H.; Wang, L.; Chen, W.-D.; Duan, Y.-T.; Sun, M.-J.; Huang, J.-J.; Peng, D.-Y.; Yu, N.-J.; Wang, Y.-Y.; Zhang, Y. Front. Nutr. 2022, 9, 963598.
[https://doi.org/10.3389/fnut.2022.963598]

-
Wu, Y.; Li, D.; Wang, H.; Wan, X. Front. Microbiol. 2022, 13, 887949.
[https://doi.org/10.3389/fmicb.2022.887949]

-
Zhou, J.; Zhang, K.; Gao, J.; Xu, J.; Wu, C.; He, M.; Zhang, S.; Zhang, D.; Dai, J.; Sun, L. Cells 2023, 12, 1456.
[https://doi.org/10.3390/cells12111456]

-
Fang, C.-L.; Paul, C. R.; Day, C. H.; Chang, R.-L.; Kuo, C.-H.; Ho, T.-J.; Hsieh, D. J.-Y.; Viswanadha, V. P.; Kuo, W.-W.; Huang, C.-Y. Environ. Toxicol. 2021, 36, 729–736.
[https://doi.org/10.1002/tox.23075]

-
Yang, H.; Zhao, Y.; Ren, B.; Wu, Y.; Qiu, Z.; Cheng, Y.; Qiu, B. Heliyon 2024, 10, e31106.
[https://doi.org/10.1016/j.heliyon.2024.e31106]

-
Chen, Y.-Y.; Chang, H.-M. Food Chem. Toxicol. 2004, 42, 759–769.
[https://doi.org/10.1016/j.fct.2004.01.018]

-
Jiang, Y.; Fan, L. J. Ethnopharmacol. 2020, 257, 112851.
[https://doi.org/10.1016/j.jep.2020.112851]

-
Xie, Z.; Zeng, H.; He, D.; Luo, J.; Liu, T.; Shen, B.; Qin, Y.; Zhang, S.; Jin, J J. Ethnopharmacol. 2024, 320, 117417.
[https://doi.org/10.1016/j.jep.2023.117417]

-
Wang, H.; Luo, Y.; Chu, Z.; Ni, T.; Ou, S.; Dai, X.; Zhang, X.; Liu, Y. Molecules 2022, 27, 3629.
[https://doi.org/10.3390/molecules27113629]

-
Albeck, J. G.; Mills, G. B.; Brugge, J. S. Mol. Cell 2013, 49, 249–261.
[https://doi.org/10.1016/j.molcel.2012.11.002]

-
New, D. C.; Wu, K.; Kwok, A. W. S.; Wong, Y. H. FEBS J. 2007, 274, 6025–6036.
[https://doi.org/10.1111/j.1742-4658.2007.06116.x]

-
Jiang, Y.; Fan, L. J. Ethnopharmacol. 2021, 266, 113456.
[https://doi.org/10.1016/j.jep.2020.113456]

-
Park, S.-G.; Jo, I.-J.; Park, S.-A.; Park, M.-C.; Mun, Y.-J. Clin. Cosmet. Investig. Dermatol. 2022, 15, 1919–1931.
[https://doi.org/10.2147/CCID.S378545]

-
Zhu, L.; Xu, J.; Zhang, S.; Wang, R.; Huang, Q.; Chen, H.; Dong, X.; Zhao, Z. J. Pharm. Biomed. Anal. 2018, 150, 278–286.
[https://doi.org/10.1016/j.jpba.2017.11.066]

-
Bao, T.-R.-G.; Long, G.-Q.; Wang, Y.; Wang, Q.; Liu, X.-L.; Hu, G.-S.; Gao, X.-X.; Wang, A.-H.; Jia, J.-M. J. Agric. Food Chem. 2022, 70, 4418–4433.
[https://doi.org/10.1021/acs.jafc.2c00823]

-
Lee, S. R.; Lee, S.; Moon, E.; Park, H.-J.; Park, H. B.; Kim, K. H. Bioorg. Chem. 2017, 70, 94–99.
[https://doi.org/10.1016/j.bioorg.2016.11.012]

-
Chen, D.-Q.; Wang, Y.-N.; Vaziri, N. D.; Chen, L.; Hu, H.-H.; Zhao, Y.-Y. Phytomedicine 2020, 72, 153232.
[https://doi.org/10.1016/j.phymed.2020.153232]

- Chen, W.; Fan, Z.; Huang, C.; Liu, J. Evid. Based Complement. Alternat. Med. 2022, 2022, 8644353.
-
Yue, S.; Feng, X.; Cai, Y.; Ibrahim, S. A.; Liu, Y.; Huang, W. Nutrients 2023, 15, 4360.
[https://doi.org/10.3390/nu15204360]

-
Ding, X.; Li, S.; Huang, H.; Shen, J.; Ding, Y.; Chen, T.; Ma, L.; Liu, J.; Lai, Y.; Chen, B.; Wang, Y.; Tan, Q. J. Ethnopharmacol. 2024, 325, 117812.
[https://doi.org/10.1016/j.jep.2024.117812]

