
Herbal Intervention Against Atherosclerosis through the Dual Lipid-Lowering and Antioxidant Actions of Caesalpinia bonducella
Abstract
This study evaluated the cardioprotective potential of Caesalpinia bonducella (L.) Roxb. seed extracts in obese rats fed a high-fat diet (HFD), aiming to improve fat metabolism, reduce oxidative stress, and prevent harmful lipid accumulation in blood vessels. Thirty-six adult albino rats were divided into six groups: normal control, HFD control, HFD with simvastatin, and HFD with petroleum ether (PTCB), ethyl acetate (EACB), or ethanolic (ETCB) seed extracts (100 mg/kg) for 30 days. Body weight, lipid profile (total cholesterol, LDL, HDL, triglycerides), and oxidative stress markers (catalase and malondialdehyde) were measured. EACB and ETCB extracts significantly lowered total cholesterol, LDL, triglycerides, and malondialdehyde while increasing HDL and catalase, with ETCB showing superior effects. The extracts also reduced HFD-induced weight gain, indicating improved fat metabolism. These results suggest that C. bonducella seed extracts, particularly ETCB, protect against atherosclerosis and oxidative stress, supporting their potential as natural cardioprotective agents and warranting further studies to clarify their underlying molecular mechanisms.
Keywords:
Caesalpinia bonducella, Atherosclerosis, High-fat diet, Lipid profile, Oxidative stressIntroduction
Cardiovascular diseases (CVDs) remain the leading cause of death globally, often referred to as “the disease of the centuries” due to their high prevalence and impact.1 In 2015 alone, CVDs accounted for approximately 17 million deaths and affected over 422 million people, representing 31% of global mortality. Alarmingly, 75% of these deaths occur in low- and middle-income countries, mainly due to limited healthcare access.2,3 Atherosclerosis is the primary contributor to CVD mortality. It is a chronic, progressive condition marked by lipid and fibrous plaque build-up in arterial walls, leading to reduced blood flow.4 While age, sex, and genetics are non-modifiable risk factors, modifiable ones include hypertension, diabetes, dyslipidemia, obesity, smoking, and physical inactivity.5 Elevated LDL-cholesterol plays a key role in atherogenesis, especially through the peroxidation of polyunsaturated fatty acids in LDL and VLDL, which promotes plaque formation.6
Conventional lipid-lowering drugs like statins, though effective, often cause adverse effects such as muscle pain and limited mobility. Other agents like colestipol and cholestyramine cause gastrointestinal issues and drug interactions, while bococizumab was withdrawn due to neutralizing antibodies.7 Plant-derived antioxidants such as flavonoids and polyphenols show promise in managing atherosclerosis by reducing oxidative stress, inflammation, LDL, and triglycerides.8 With 80% of the global population relying on herbal remedies, especially in India, plant-based therapies offer a safer alternative.
Caesalpinia bonducella (L.) Roxb., traditionally used for various ailments, exhibits antioxidant, antihyperlipidemic, antidiabetic, and anti-inflammatory properties.9–14 Although its hypolipidemic activity is documented.15–20 its anti-atherogenic potential in high-fat diet models remains unexplored. This study investigates the ability of different seed extracts of C. bonducella to mitigate atherosclerosis in obese rats.
Experimental
Plants material collection and extraction – The seeds of Caesalpinia bonducella (L.) Roxb. were collected from Mangalore and authenticated. About 500 g of powdered material was successively extracted using petroleum ether, ethanol, and ethyl acetate in a Soxhlet apparatus at 40–60°C. The marc was oven-dried and re-extracted with solvents of increasing polarity. Each extract was concentrated under reduced pressure and lyophilized (Lyodel).21
Preliminary phytochemical screening of plant extracts – Preliminary phytochemical screening of the extracts of Caesalpinia bonducella (L.) Roxb seed was carried out according to standard procedures described in the literature.22,23
Experimental design, animal grouping, and diet preparation – Healthy male Wistar rats (Rattus norvegicus domestica) were used in the study after approval from the Institutional Animal Ethics Committee (Approval No: AKCP/IAEC/01/19-20), in accordance with CPCSEA guidelines. Thirty-six rats were randomly divided into six groups (n = 6). Group I received normal saline (negative control); Group II received a high-fat diet (HFD, positive control); Group III received simvastatin (1.8 mg/kg b.w., orally) with HFD; Groups IV, V, and VI received petroleum ether, ethyl acetate, and ethanolic extracts of Caesalpinia bonducella, respectively, at 100 mg/kg b.w., all with HFD. Extracts and simvastatin were suspended in normal saline and administered orally once daily for 30 days.
The HFD was formulated by mixing 20.5% wheat flour, 52.6% roasted Bengal gram, 5% skim milk powder, 4% refined oil, 4% casein, 4% starch with salt mixture, 9% coconut oil, 0.5% choline-vitamin mix, and 0.4% cholesterol. After the treatment period and overnight fasting, all animals were euthanized by cervical dislocation. Blood samples were collected in heparinized tubes for plasma separation. The heart, liver, and aorta were excised, cleared of adipose tissue, weighed, and homogenized for further biochemical analysis. All procedures strictly followed animal welfare and ethical protocols.
Lipid extraction from tissues and plasma – All analytical methods used in this study are standard and validated, as described in the cited references. Lipid extraction from tissues and plasma was carried out following the method of Folch et al. (1957), using ethanol-ether and chloroform-methanol mixtures, with sequential heating and centrifugation steps to isolate lipid fractions for analysis of triglycerides, phospholipids, free fatty acids, and cholesterol forms.24
Estimation of free and ester cholesterol – Cholesterol fractions were quantified using the method by Varley (1954), involving digitonin complexation, selective extraction with petroleum ether, and colorimetric analysis via the Liebermann–Burchard reaction at 620 nm.25
Estimation of free fatty acids – Free fatty acids were extracted, purified using silicic acid, and reacted with diphenyl carbazide after complexation with copper soaps, based on the method by Falholt et al. (1973), with absorbance read at 540 nm.26
Estimation of phospholipids – Phospholipid content was determined as inorganic phosphorus using the Zilversmit and Davis (1950) method involving digestion with acids, molybdate complex formation, and color development using ANSA reagent measured at 680 nm.27
Estimation of triglycerides – Triglycerides were estimated via isopropanol extraction, alumina deproteinization, and subsequent color development with metaperiodate and acetyl acetone as per Foster and Dunn (1973), read at 405 nm.28
Estimation of HDL cholesterol – HDL cholesterol was quantified enzymatically using a cholesterol oxidase method after apo B-containing lipoprotein precipitation by phosphotungstic acid and magnesium (Demacker et al., n.d.).29
Calculation of LDL cholesterol – LDL cholesterol was calculated using the Friedewald formula:
Calculation of VLDL cholesterol – VLDL cholesterol was derived by the formula:
Calculation of atherogenic index – Atherogenic index was calculated using the log ratio of triglycerides to HDL cholesterol as per Sun et al. (2024).32
Calculation of cardiac risk ratio – Cardiac risk ratio was calculated by:
Calculation of atherogenic coefficient – Atherogenic coefficient was calculated using:
Estimation of TBARS (lipid peroxidation marker) – Thiobarbituric acid reactive substances (TBARS) were estimated by reaction with TBA in acidic medium, forming a pink chromophore measured at 535 nm as per Niehaus and Samuelsson (1968).34
Estimation of conjugated dienes – Conjugated dienes, formed by lipid peroxidation of PUFAs, were extracted and measured at 233 nm using a method adapted from Buege and Aust (1978), indicating oxidative stress levels in tissues.35
Statistical analysis – All data are presented as mean ± standard deviation (SD) for six animals per group. Each sample was analyzed in triplicate, and the mean of triplicates was used for statistical analysis to minimize analytical variation. Statistical significance was determined using one-way analysis of variance (ANOVA) followed by Scheffe’s multiple comparisons test. A p-value < 0.05 was considered statistically significant.
Results and Discussion
A high-fat diet (HFD) led to a significant increase in body weight in Group II rats compared to normal controls (Group I). This confirms diet-induced obesity. Treatment with various extracts of Caesalpinia bonducella—especially the ethanolic extract (ETCB) at 200 mg/kg—significantly attenuated this weight gain. The reduction observed in extract-treated groups was comparable to the Simvastatin standard group (Group III), indicating the anti-obesity potential of the plant (Table 1). These results align with previous findings demonstrating the role of phytochemicals in lipid metabolism regulation.36
The HFD-fed rats (Group II) showed a significant increase in total cholesterol, triglycerides, and free fatty acids compared to controls. Treatment with ETCB and ethyl acetate extract (EACB), especially at 200 mg/kg, resulted in marked reductions in these parameters. Among the extracts, ETCB exhibited the most pronounced hypolipidemic effect. This can be attributed to flavonoids and sterols, which have known lipid-lowering activity.37–39 Furthermore, phospholipid levels also decreased significantly in extract-treated groups compared to HFD rats (Table 2), supporting improved lipid mobilization.

Effect of Caesalpinia bonducella extracts on plasma lipid profile parameters including total cholesterol, triglycerides, free fatty acids, and phospholipids in high-fat diet-fed rats
Group II rats showed a notable decrease in HDL-cholesterol and an increase in LDL and VLDL-cholesterol, reflecting a typical dyslipidemic profile. Post-treatment with extracts restored HDL levels and significantly decreased LDL and VLDL fractions (Table 3). The effect was dose-dependent, with ETCB at 200 mg/kg showing the best modulation. These outcomes suggest the extracts improve reverse cholesterol transport and reduce atherogenic indices.40,41

Effect of various extracts of Caesalpinia bonducella on plasma lipoprotein levels (HDL, LDL, VLDL) in high-fat diet-fed rats
The liver and heart lipid content in HFD rats showed substantial accumulation of cholesterol and triglycerides. Extract treatments markedly reversed this lipid deposition in both tissues (Table 4). The efficacy was again most pronounced in high-dose ETCB groups, indicating strong lipid-regulatory effects in peripheral organs. This might be due to improved lipid oxidation and reduced synthesis, as suggested in previous phytotherapeutic studies.42

Effect of various extracts of Caesalpinia bonducella on tissue lipid content (cholesterol and triglycerides) in high-fat diet-fed rats
Lipid peroxidation, measured via thiobarbituric acid reactive substances (TBARS), increased significantly in the liver and heart of HFD-fed rats (Group II). Administration of both ETCB and EACB led to a significant decrease in TBARS levels (Table 5), indicating reduced oxidative stress. These findings support the antioxidant potential of C. bonducella, possibly attributed to its polyphenolic content.42

Effect of various extracts of Caesalpinia bonducella on Tissue Lipid Peroxidation (cholesterol and triglycerides) in high-fat diet—fed rats
A high-fat diet caused a substantial decline in enzymic antioxidants such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) in liver and heart tissues. Extract treatments significantly enhanced the activity of these enzymes, with the highest restoration seen in the ETCB 200 mg/kg group (Table 6). These results reinforce the role of C. bonducella in re-establishing redox balance through enzymic defense.43,44

Effect of various extracts of Caesalpinia bonducella on Tissue Enzymic Antioxidants (TBARS levels) in high-fat diet-fed rats
The concentration of glutathione (GSH)—a critical non-enzymic antioxidant—was significantly depleted in the aorta, heart, and liver of HFD rats (Group II), as shown in Table 7. Treatment with C. bonducella extracts, particularly ETCB at both 100 and 200 mg/kg, restored GSH levels across all tissues. This may be due to the activation of glutathione reductase or enhanced GSH synthesis.45,46 Restoration of GSH reflects improved antioxidant status, which is essential for combating HFD-induced oxidative injury.47,48

Effect of various extracts of Caesalpinia bonducella on Tissue Non-Enzymic Antioxidants in high-fat diet-fed rats
Histopathological evaluation confirmed biochemical findings. The liver of HFD-fed rats showed microvesicular steatosis and congested blood vessels. Treatment with ETCB and EACB at both doses markedly reduced steatosis and vascular congestion, showing restoration of hepatic architecture (Fig. 1). Similarly, aortic sections from HFD rats displayed prominent fat deposition, which was significantly reduced in extract-treated groups (Fig. 2). These improvements are consistent with the lipid-lowering and antioxidant effects observed in earlier parameters.
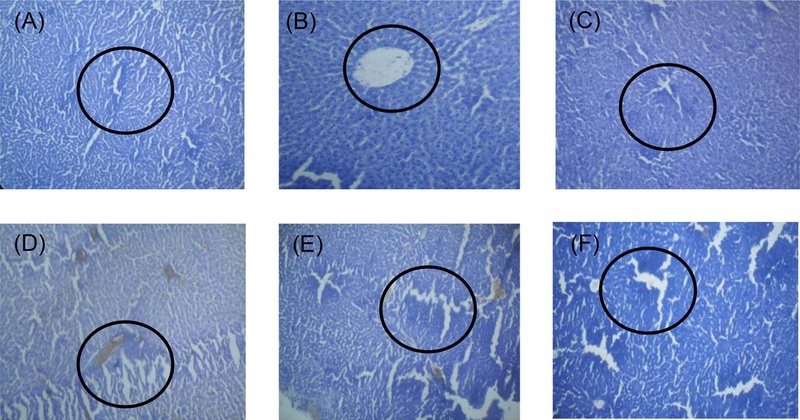
Histopathological studies for liver [(A–F) Group I–VI]; (A) Group I received normal saline (negative control); (B) Group II received a high-fat diet (HFD, positive control); (C) Group III received simvastatin (1.8 mg/kg b.w., orally) with HFD; (D)–(F) Groups IV, V, and VI received petroleum ether, ethyl acetate, and ethanolic extracts of Caesalpinia bonducella, respectively, at 100 mg/kg b.w., all with HFD.
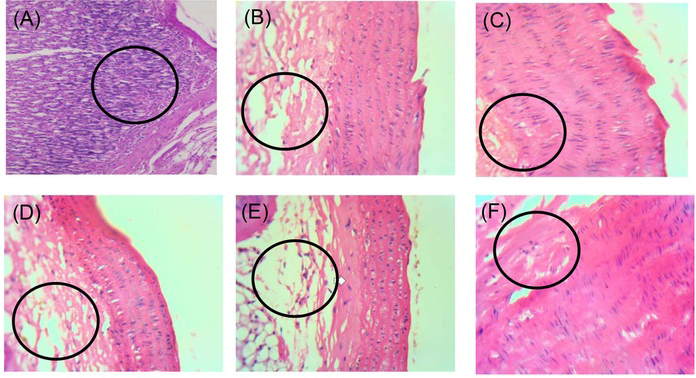
Histopathological studies for aorta [(A–F) Group I–VI)]; (A) Group I received normal saline (negative control); (B) Group II received a high-fat diet (HFD, positive control); (C) Group III received simvastatin (1.8 mg/kg b.w., orally) with HFD; (D–F) Groups IV, V, and VI received petroleum ether, ethyl acetate, and ethanolic extracts of Caesalpinia bonducella, respectively, at 100 mg/kg b.w., all with HFD.
The ethanolic extract had the most comprehensive range of phytoconstituents, including alkaloids, sterols, flavonoids, tannins, phenolic compounds, saponins, glycosides, and amino acids. These compounds are strongly linked in literature to the prevention and treatment of atherosclerosis due to their antioxidant, anti-inflammatory, and lipid-lowering effects. Consistent with previous reports, extracts rich in flavonoids, sterols, saponins, and phenolics exhibit potent anti-atherosclerotic activities by improving lipid profiles and reducing plaque formation. Our in vivo biochemical results, showing significant reductions in total cholesterol, LDL-C, triglycerides, and improvements in HDL-C, support this.49,50 Together, the findings indicate that the phytochemically rich ethanolic extract is largely responsible for the observed cardiovascular-protective effects, confirming its strong anti-atherosclerotic potential.
In conclusion, the present study demonstrates that Caesalpinia bonducella extracts, particularly the ethyl acetate and ethanolic fractions at a dose of 100 mg/kg body weight, exhibit significant anti-atherogenic and antioxidant effects in HFD-induced hyperlipidemic rats. Treatment led to a notable reduction in plasma and tissue levels of free and esterified cholesterol, triglycerides, phospholipids, LDL, and VLDL, while restoring HDL levels and reducing the atherogenic index—effects comparable to the standard drug simvastatin. Additionally, the extracts significantly attenuated lipid peroxidation markers such as TBARS and conjugated dienes and enhanced both enzymatic (GPX, GRX, GST, SOD) and non-enzymatic (glutathione) antioxidant defenses in vital organs. The use of validated and standardized biochemical assays ensured high analytical precision, which is evident from the minimal variation among replicates, thereby supporting the reliability of the experimental findings. These findings highlight the therapeutic potential and novelty of C. bonducella in managing diet-induced atherosclerosis through dual lipid-lowering and antioxidative mechanisms. The promising outcomes support its further exploration through molecular studies to elucidate the underlying pathways and potential development as a plant-based alternative in cardiovascular disease management.
Acknowledgments
The authors thank the Arulmigu Kalasalingam College of Pharmacy, for providing tremendous opportunities accomplish this work.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Burlutskaya, A. V.; Tril, V. E.; Polischuk, L. V.; Pokrovskii, V. M. Rev. Cardiovasc. Med. 2021, 22, 817–834.
[https://doi.org/10.31083/j.rcm2203088]

-
Mozaffarian, D. J. Am. Coll. Cardiol. 2017, 70, 26–28.
[https://doi.org/10.1016/j.jacc.2017.05.007]

- Roth, G. A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S. F.; Abyu, G.; Ukwaja, K. N. J. Am. Coll. Cardiol. 2017, 70, 1-25.
-
Nedkoff, L.; Briffa, T.; Zemedikun, D.; Herrington, S.; Wright, F. L. Clin. Ther. 2023, 45, 1087–1091.
[https://doi.org/10.1016/j.clinthera.2023.09.020]

-
Luca, A. C.; David, S. G.; David, A. G.; Țarcă, V.; Pădureț, I.-A.; Mîndru, D. E.; Roșu, S. T.; Roșu, E. V.; Adumitrăchioaiei, H.; Bernic, J.; Cojocaru, E.; Țarcă, E. Life 2023, 13, 2056.
[https://doi.org/10.3390/life13102056]

-
Devi, J. A. I.; Muthu, A. K. Afr. J. Pharm. Pharmacol. 2015, 9, 1080–1087.
[https://doi.org/10.5897/AJPP2015.4447]

-
Bergheanu, S. C.; Bodde, M. C.; Jukema, J. W. Neth. Heart J. 2017, 25, 231–242.
[https://doi.org/10.1007/s12471-017-0959-2]

-
Singh, S. Curr. Atheroscler. Rep. 2019, 21, 12.
[https://doi.org/10.1007/s11883-019-0774-x]

- Manikandaselvi, S.; Vadivel, V.;Brindha, P. J. Chem. Pharm. Res. 2015, 7, 137-142.
- Pandey, D. D.; Jain, A. P.; Kumar, A. Pharma Innov. 2018, 7, 190-193.
- Sundar, J. S.; Christobel, R. G. J.; Selvi, N. K.; Abirami, M. P.; Samuel, S. Pharma Innov. 2018, 7, 95-102.
-
Sayyed, F. J.; Wadkar, G. H. Ann. Pharm. Fr. 2018, 76, 44–49.
[https://doi.org/10.1016/j.pharma.2017.09.004]

- Deepika, K. S. N.; Muthuraman, M. S.; Natesan, R.; Pemaiah, B. Int. J. Pharm. Pharm. Sci. 2014, 311–314.
-
Kandasamy, V.; Balasundaram, U. J. Ethnopharmacol. 2021, 279, 114–375.
[https://doi.org/10.1016/j.jep.2021.114375]

-
Musa, W. J.; Bialangi, N.; Kilo, A. K.; Situmeang, B.; Susparini, N. T.; Rusydi, I. D. Pharmacia 2023, 70, 97–103.
[https://doi.org/10.3897/pharmacia.70.e96817]

-
Parameshwar, S.; Srinivasan, K. K.; Rao, C. M. Pharm. Biol. 2002, 40, 590–595.
[https://doi.org/10.1076/phbi.40.8.590.14656]

- Jana, K.; Chatterjee, K.; Bera, T. K.; Maiti, S.; De, D.; Ali, K. M.; Ghosh, D. Int. J. Pharm. Tech. Res. 2010, 2, 2234–2242.
-
Musa, W. J. A.; Bialangi, N.; Kilo, A. K.; Situmeang, B.; Susparini, N. T.; Rusydi, I. D. Pharmacia 2023, 70, 97–103.
[https://doi.org/10.3897/pharmacia.70.e96817]

-
Kannur, D. M.; Hukkeri, V. I.; Akki, K. S. Fitoterapia 2006, 77, 546–549.
[https://doi.org/10.1016/j.fitote.2006.06.013]

-
Sharma, S. R.; Dwivedi, S. K.; Swarup, D. J. Ethnopharmacol. 1997, 58, 39–44.
[https://doi.org/10.1016/S0378-8741(97)00079-2]

- Harborne, J. B. In Phytochemical methods: a guide to modern techniques of plant analysis; Springer: Netherlands, 1954, pp 1–36.
- Trease, G. E.; Evans, W. C. Textbook of Pharmacognosy, 12th ed.; Billiere Tindall and W. B; London, 1983; p 444.
- Harborne, J. B. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis; Springer Science & Business Media: Germany, 1998, pp 49–188.
-
Folch, J.; Lees, M.; Stanley, G. S. J. Biol. Chem. 1957, 226, 497–509.
[https://doi.org/10.1016/S0021-9258(18)64849-5]

- Varley, H. Practical clinical biochemistry; William Heinemann Medical Books Ltd; 1974, pp 186–190.
-
Falholt, K.; Lund, B.; Falholt, W. Clin. Chim. Acta. 1973, 46, 105–111.
[https://doi.org/10.1016/0009-8981(73)90016-8]

- Zilversmit, D. B.; Davis, A. K. J. Lab. Clin. Med. 1950, 35, 155–160.
-
Foster, L. B.; Dunn, R. T. Clin. Chem. 1973, 19, 338–340.
[https://doi.org/10.1093/clinchem/19.3.338]

-
Demacker, P. N.; Hessels, M.; Toenhake-Dijkstra, H.; Baadenhuijsen, H. Clin. Chem. 1997, 43, 663–668.
[https://doi.org/10.1093/clinchem/43.4.663]

- Lee, Y.; Siddiqui, W. J. Cholesterol Levels; StatPearls Publishing: Treasure Island, FL, 2023, pp 1–18.
-
DeLong, D. M.; DeLong, E. R.; Wood, P. D.; Lippel, K.; Rifkind, B. M. JAMA 1986, 256, 2372–2377.
[https://doi.org/10.1001/jama.1986.03380170088024]

-
Sun, Y.; Li, F.; Zhou, Y.; Liu, A.; Lin, X.; Zou, Z.; Lv, X.; Zhou, J.; Li, Z.; Wu, X.; Dou, S.; Zhang, M.; Zhu, J.; Chen, Y.; Xiao, X.; Hu, Y.; Li, H.; Li, Y. Cardiovasc. Diabetol. 2024, 23, 226.
[https://doi.org/10.1186/s12933-024-02330-y]

- Sujatha, R.; Kavitha, S. J. Neurol. 2017, 16, 78–82.
-
Niehaus Jr, W. G.; Samuelsson, B. Eur. J. Biochem. 1968, 6, 126–130.
[https://doi.org/10.1111/j.1432-1033.1968.tb00428.x]

-
Kong, X.; Chen, H.; Li, D.; Ma, L. Cell. Mol. Biol. 2021, 67, 144–150.
[https://doi.org/10.14715/cmb/2021.67.5.20]

-
Buege, J. A.; Aust, S. D. Methods Enzymol. 1978, 52, 302–310.
[https://doi.org/10.1016/S0076-6879(78)52032-6]

-
Zhang, S.; Hong, F.; Ma, C.; Yang, S. Endocr. Metab. Immune Disord. Drug Targets. 2022, 22, 590–600.
[https://doi.org/10.2174/1871530322666211220110810]

-
Wan, J.; Feng, Y.; Du, L.; Veeraraghavan, V. P.; Mohan, S. K.; Guo, S. J. Environ. Pathol. Toxicol. Oncol. 2020, 39, 61–65.
[https://doi.org/10.1615/JEnvironPatholToxicolOncol.2020031478]

-
Hu, N.; Chen, C.; Wang, J.; Huang, J.; Yao, D.; Li, C. Int. J. Mol. Sci. 2021, 22, 11107.
[https://doi.org/10.3390/ijms222011107]

-
Çelik, E.; Çora, A. R.; Karadem, K. B. J. Saudi Heart Assoc. 2021, 33, 244.
[https://doi.org/10.37616/2212-5043.1266]

- Onuchukwu, I. C.; Etim, E. U.; Chimdike, O. G.; Oguzie, B. C.; Emuebie, H.; Orji, K. N.; Ifionu, O. V. J. Drug Deliv. Ther. 2022, 12214.
-
Duan, H.; Song, P.; Li, R.; Su, H.; He, L. Front. Pharmacol. 2023, 14, 1161657.
[https://doi.org/10.3389/fphar.2023.1161657]

-
Poznyak, A. V.; Grechko, A. V.; Orekhova, V. A.; Chegodaev, Y. S.; Wu, W.-K.; Orekhov, A. N. Biology 2020, 9, 60.
[https://doi.org/10.3390/biology9030060]

-
Liu, Y.-G.; Yan, J.-L.; Ji, Y.-Q.; Nie, W.-J.; Jiang, Y. Food Funct. 2020, 11, 2997–3005.
[https://doi.org/10.1039/C9FO02736J]

- Sethupathy, S.; Elanchezhiyan, C.; Vasudevan, K.; Rajagopal, G. Indian J. Exp. Biol. 2002, 40, 1169–1172.
-
Neumann, C. A.; Krause, D. S.; Carman, C. V.; Das, S.; Dubey, D. P.; Abraham, J. L.; Van Etten, R. A. Nature 2003, 424, 561–565.
[https://doi.org/10.1038/nature01819]

-
Ramos, L. P. A.; Justino, A. B.; Tavernelli, N.; Saraiva, A. L.; Franco, R. R.; Souza, A. V.; Silva, H. C. G.; Moura, F. B. R.; Botelho, F. V.; Espindola, F. S. Biomed. Pharmacother. 2021, 142, 112049.
[https://doi.org/10.1016/j.biopha.2021.112049]

-
Meeran, M. F. N.; Azimullah, S.; Al Ahbabi, M. M.; Jha, N. K.; Lakshmanan, V.-K.; Goyal, S. N.; Ojha, S. Molecules 2020, 25, 5656.
[https://doi.org/10.3390/molecules25235656]

-
Yap, N. A. L.; Khan, Z.; He, X.; Lee, J.-G.; Maung, S.; Morgan, K. R.; Zhou, T.; Precht, H.; Serruys, P. W.; Garcia-Garcia, H. M.; Onuma, Y.; Hynes, S.; Kelle, S.; Mathur, A.; Baumbach, A.; Bourantas, C. V. Front. Cardiovasc. Med. 2025, 12, 1507892.
[https://doi.org/10.3389/fcvm.2025.1507892]

-
Salvamani, S.; Gunasekaran, B.; Shukor, M. Y.; Bakar, M. Z. A.; Ahmad, S. A. RSC Adv. 2016, 6, 32685–32696.
[https://doi.org/10.1039/C6RA04827G]
