
Protective Effect of Secang Wood (Caesalpinia sappan L.) on Wistar Rats’ Trachea upon Exposure to Cigarette Smoke
Abstract
Continuous exposure to cigarette smoke causes oxidative stress, leading to diseases in the respiratory tract. Secang (Caesalpinia sappan L.) wood is a popular herbal drink in Indonesia due to its antioxidant properties. To date, the protective effects of Secang on the respiratory system have not been widely studied. Ethanolic extract of Secang was prepared by maceration. An in vivo experiment was conducted on 24 male Wistar rats divided into four groups: Control (K), Negative control (KN), low-dose treatment of the extract (P1, 300 mg/kg), and a high-dose one (P2, 600 mg/kg) for 30 days of treatment. After finishing treatments, rats were sacrificed for histological analysis. Rats' average bodyweight showed a consistent increase, indicating tolerable toxicity of the extract, which does not interfere with rats' general health. Serum glutamic pyruvic transaminase (SGPT) and serum glutamic oxaloacetic transaminase (SGOT) depicted consistent results, where P1 and P2 groups significantly decreased (p < 0.001) compared to KN, indicating hepatoprotective activity of Secang. Histologically, tracheal lumen diameter in P2 significantly (p < 0.05) increased compared to KN, suggesting Secang’s potent antioxidant activity. The number of goblet cells increased in KN compared to K, indicating irritation and inflammatory conditions from cigarette smoke. Secang-treated groups exhibited improvement in the lumen diameter, goblet cells, and ciliated epithelium. The findings suggest protective potential of Secang against cigarette smoke-induced tracheal damage.
Keywords:
Cigarette smoke, Secang, Antioxidant, TracheaIntroduction
Smoking is a prevalent unhealthy habit that links to many non-communicable diseases and mortalities all over the world. Despite its harmful effects on health, smoking is still a common practice for people who predominantly live in urban areas rather than in rural areas in Indonesia.1 Recent studies have shown that more than 60% of Indonesian men are active smokers, and only less than 14% of them are willing to quit smoking.2 According to a 2023 WHO report on the global tobacco epidemic, over 8 million people annually die from diseases related to smoking or exposure to secondhand smoke.3 More than 4000 toxicants, including polycyclic aromatic hydrocarbons, metals, aldehydes, and nitrosamines, are found in cigarette smoke, which are harmful, addictive, and known as cancer-causing agents.4 Exposure to free radicals in cigarette smoke can cause oxidative stress and lead to various diseases. Preclinical studies have shown that cigarette smoke causes adverse physiological effects on Wistar rats’ respiratory and excretory systems.5
The body’s natural antioxidants can neutralize free radical reactions, thereby preventing diseases due to these oxidative reactions.6 However, this natural ability decreases as we age and accumulate exposure to free radicals. Therefore, as an alternative solution, people commonly consume herbal drinks that contain various phytochemicals rich in antioxidants, such as Secang (Caesalpinia sappan L.) from the Leguminosae family.7 While Secang wood is widely recognized for its antioxidant properties and potential health benefits,8 there appears to be limited research investigating its protective effects on the respiratory system. Most studies focus on its antioxidant, anti-inflammatory, and antibacterial properties, as well as its applications in treating other conditions like cancer or gastrointestinal diseases.9
As a popular herbal plant frequently utilized as a traditional drink, Secang is commonly found in many tropical Asian countries like Indonesia. Traditionally, Secang wood has been employed by communities as a medicinal treatment and dye. It is particularly noted for its effectiveness in reducing inflammatory conditions, treating various ailments such as diarrhea, dysentery, hemoptysis due to tuberculosis, hematemesis, syphilis, malaria, tetanus, tumors, and pain resulting from blood circulation disorders.7
Secang is rich in various chemical constituents, including gallic acid, tannins, alkaloids, saponins, flavonoids, phenolics, glycosides, resorcinol, brazilin, brazilein, sappanchalcone, and essential oils.9 Brazilin is a compound that gives Secang wood its red color and belongs to the group of flavonoids, as homoisoflavonoids which shows potent antioxidant properties.9,10
As part of the rat’s respiratory system, the trachea serves as an airway located between the larynx and the bronchi. The transverse cross-section of the trachea consists of several layers: the mucosal layer (which includes ciliated and lamina propria-layered cylindrical epithelial cells), the submucosal layer (the glandular tip region), the cartilage ring, the muscle layer (musculus transversus trachealis), and the adventitia layer.11 Pulmonary histology of rats consists of the alveolar system, which is located on the walls of the respiratory bronchioles, alveolar ducts, and sacs. The alveoli are lined by a thin layer of epithelial cells (primarily type I pneumocytes) resting on the basal membrane which is closely apposed to the basal membrane of the capillary endothelium, forming a thin air-blood barrier separated by interalveolar septa.12
Ample studies suggest that cigarette smoke exposure adversely affects the respiratory organs; however, the effect of Secang on tracheal histology and goblet cells is still underexplored. Hence, this study aimed to investigate the protective effects of Secang wood on rats’ trachea.
Experimental
General experimental procedures – The materials used in the experiments include commercially-obtained Secang wood, 24 male Wistar rats aged 3–4 months with an average weight of 250 g, husks, filter paper (Whatman), distilled water (dH2O), aluminum foil (Klinpak), glass slides (Sailing Boat), NaCl 0.9% (Merck), picric acid (SigmaAldrich), paraffin (Merck), toluol (Sigma-Aldrich), xylol (Sigma-Aldrich), formalin 10% v/v (Warchem), ethanol 96% v/v (Merck), hematoxylin (Merck), eosin (Merck), PBS (Merck), ether (Warchem), ethanol series of 70%, 80%, 90%, and 100% (v/v) (Merck). The tools used in the experiments were: analytical scales (Mettler toredo), pocket scales (Acis), hand counters, gloves, masks, haemocytometers, dropper pipettes, dark reagent bottles, blenders (Philips), oven (Memmert), micropipettes and tips (Eppendorf), 500 ml erlenmeyer (Duran), glass funnels (Herma), stirring rods, porcelain pestles, rotary evaporators (IKA), pharmaceutical sieves number 100, maintenance cages, laboratory inhalation chamber, embedding sets, rat gastric probe with 1 cc syringe, microscope camera (Optilab), object glass (Sail brand), flannel bottle, cover slip (One Med), paraffin block, water bath (Memmert), microscope (Olympus), dissecting set, and petri dish.
Preparation of Secang wood ethanol extract – Secang identification was carried out in the Plant Structure Laboratory, Faculty of Applied Science and Technology, Universitas Ahmad Dahlan (UAD) (voucher number 470/Lab.Bio/B/VII/2025). This plant was dried in the oven at 60°C overnight and ground in a blender to make powder. This simplicia was then macerated at room temperature using 96% ethanol in 5 cycles for 2 hours each. The extract was concentrated using a rotary evaporator and air-dried until the solvent is completely vaporized to yield a red-brownish semi-solid extract.13,14 Although batch-specific chromatographic profiling was not conducted in this proof-of-concept study, the extraction protocol followed established procedures to ensure compositional consistency with previously characterized ethanolic extracts of Secang.14,15 Other studies reported HPLC profiling of comparable ethanolic extract of Secang revealed brazilin as the major bioactive compound and its oxidized form, brazilein, which are responsible for the observed antioxidant activity.14–16
Animal preparation and treatments – To conduct a study, the care and maintenance of the animal lab followed the guidelines from the ethical committee of UAD. Soon after ethical approval was issued by the Ethics Committee of UAD (approval number: 012412360), a total of 24 male Wistar rats were obtained commercially and acclimatized for 7 days in a 55 × 40 cm cage with a temperature of ± 25°C, room humidity of 40–60%, and with 12 hours of light/dark a day. Husk replacement is carried out every 3 days, BR1 feeding and drinking ad libitum. The rats were weighed once a week using a digital scale. Before performing treatments, rats were divided into 4 groups, i.e. control group/K (only treated with distilled water), negative control/KN (exposed to 2 cigarettes smoke/day), P1 (treated with 300 mg/kg of extract then exposed to 2 cigarettes smoke/day), and P2 (treated with 600 mg/kg of extract then exposed to 2 cigarettes smoke/day). The duration of all treatments was 30 days.17
All rats in KN, P1 and P2 were exposed to cigarette smoke (Dji Sam Soe brand, containing 39 mg tar and 2.3 mg nicotine per cigarette), via whole-body inhalation chamber to simulate chronic passive exposure. The inhalation chamber (47 L high-grade plastic container, custom made) was sufficiently ventilated to avoid hypoxia and smoke from lit cigarettes was drawn into chamber via mini compressor. During 30-day treatment period, rats were placed unrestrained in the chamber (6 rats per session), exposed to 2 cigarette smoke for 15 minutes per session at 9 am daily. Control group (K) rats were placed in identical chamber with fresh air only. All procedures were carefully monitored to follow ethical guidelines for animal use.
Measurement of SGOT and SGPT levels – At the end of the treatment duration, 100 μL of rats' blood was drawn from a retro-orbital sinus vein using a sterile syringe. Blood serum was then supplemented with 1000 μL of SGOT/SGPT reagent and then incubated for 5 minutes at 37°C. After that, measurements are carried out photometrically by measuring the absorbance of the spectrophotometer at a wavelength of 340 nm. SGOT levels are calculated by the formula: SGOT (U/L) = ∆Abs/min 1746, while SGPT levels are calculated by the formula: SGPT (U/L) = ∆Abs/min 1768.18
Histopathology preparations – After the last treatments on day 30, all rats were rested for 24 hours to minimize physiological stress that could affect the parameters measured. The anesthetized rats were then euthanized by cervical dislocation and dissected to isolate the trachea. The organ was cleaned using physiological salts (NaCl 0.9%) and weighed with digital scales. Organ fixation was conducted using 10% Formalin Neutral Buffer solution for 24 hours. Histopathological preparation was performed using the paraffin method with hematoxylin-eosin (HE) staining. The observation parameters of the respiratory organ include tracheal lumen diameter, number of Goblet cells, and percentage of ciliated epithelial cells.19
Data analysis – Statistical analysis of the data was performed in GraphPad Prism Version 10.4.1 (GraphPad Software, Inc., 2024). The Shapiro-Wilk and Kolmogorov-Smirnov tests were utilized to check normal data distribution. One-way ANOVA and Duncan's test were conducted, with significance levels shown as: ns, p > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Results and Discussion
The weight of the rats was measured weekly from acclimatization to the completion of treatment. Fig. 1 illustrates the average weight of the rats that continued to increase from day 0 to the end of the treatment (week 5). This finding depicted that, in general, the treatment does not cause acute toxicity effects that inhibit the physical growth of rats. Exposure to toxic substances from cigarette smoke is also still within the limits of physiological tolerance, so that the weight of rats still increases over time. In concordance, earlier studies have shown that Secang wood did not significantly affect weight gain and relative organ weight in rats, highlighting its low toxicity and good safety profile.17,20
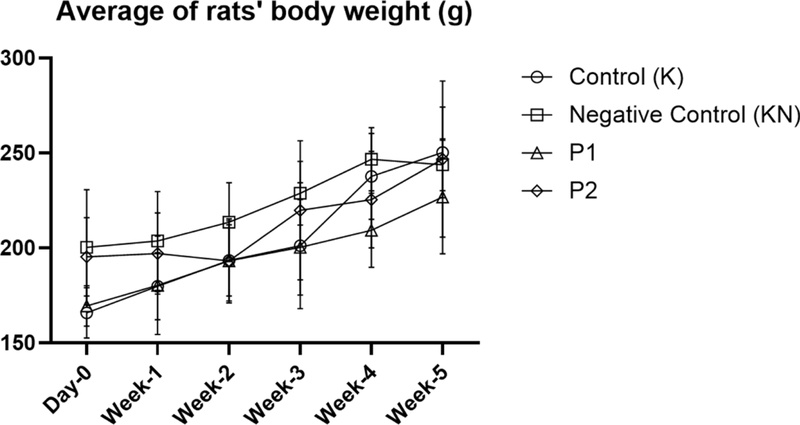
Average of rats' body weight (g) for each group. Day-0 refers to the initial average body weight on acclimatization, week-1 refers to the average rats’ body weight after acclimatization, and week-5 refers to the last day of treatment.
Blood biochemical levels in the form of serum glutamic pyruvic transaminase (SGPT) and serum glutamic oxaloacetic transaminase (SGOT) enzymes are important indicators of microscopic liver damage induced by exposure to cigarette smoke during treatment. Elevated levels of SGOT and SGPT provide crucial information regarding liver function and the severity of liver damage due to viral infection, toxic substances, or cigarette smoke. Both transaminase enzymes are produced in hepatocyte cells, and when there is cell damage due to exposure to free radicals, including those from cigarette smoke exposure, they are released from the cytoplasm into the bloodstream.21
Fig. 2 depicts that SGPT and SGOT levels have similar patterns for all groups. In the negative control group (KN), SGPT and SGOT levels increased significantly (p < 0.0001) compared to controls (K), which indicated significant liver damage due to exposure to cigarette smoke every day for 30 days. In concordance, an earlier study exhibited that cigarette smoke exposure significantly elevated SGPT and SGOT levels, emphasizing smoke-induced hepatotoxicity.22 In P1 and P2 groups, treatment with Secang wood extract was significantly effective in reducing SGPT and SGOT levels, indicating the hepatoprotective effect of Secang wood bioactive compounds at doses of 300 and 600 mg/kg. Earlier studies reported that Secang wood lowers SGPT and SGOT levels, as well as malondialdehyde (MDA), an oxidative stress biomarker, in alloxan-induced diabetic rats.23
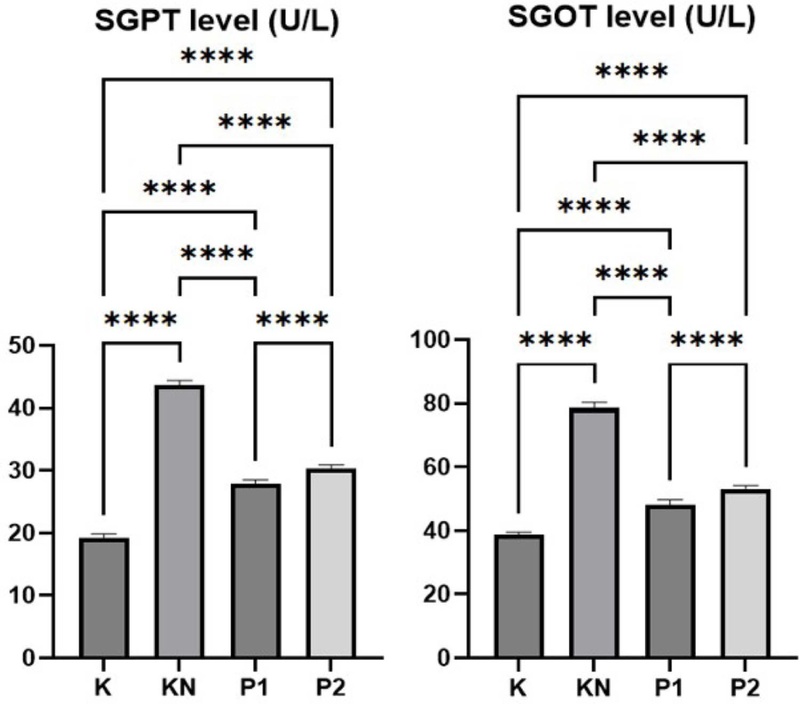
SGPT and SGOT level (U/L) after 30 days of treatment. K (control group), KN (negative control group, exposed to cigarette smoke), P1 (300 mg/kg of Secang wood extract and exposed to cigarette smoke), and P2 (600 mg/kg of Secang wood extract and exposed to cigarette smoke). Data were obtained from 6 animals in each group, presented as mean ± SD. One-way ANOVA was performed for statistical analysis with Duncan’s post hoc test (****p < 0.0001).
As seen in Fig. 2, levels of SGOT and SGPT in P1 and P2 groups were still significantly higher than in the control group, which may suggest extensive hepatocellular damage during the period of treatment with cigarette smoke that is not reversible even in the presence of antioxidants. Harmful chemicals in cigarette smoke, which contribute to liver fibrosis and cirrhosis, include nicotine, tar, nitrosamines, and free radicals. Mechanisms by which smoking induces liver injury are via three different ways: direct and indirect toxicity, immunologic, and oncogenic.24 Cytotoxic constituents cause oxidative stress, leading to fibrosis, while at the same time increasing proinflammatory cytokines (including interleukin-8/IL-8 and tumor necrosis factor alpha/TNF-α), inducing liver cell damage. Nicotine and other toxicants in cigarettes also decrease the immune system through the inhibition of lymphocyte proliferation and differentiation.24,25
One of the prominent parameters observed to determine whether treatments produce any effects on the respiratory system is the tracheal histopathological structure. As seen in Fig. 3, toxicants and free radicals from the exposure of cigarette smoke for 30 days affected the cilia structure of the tracheal cells, shown with green arrows in the KN, P1, and P2 groups. Damaged structure and decreased ciliated epithelial cells can be observed in the KN and P1 groups. In contrast, ciliated cell improvement can be seen in P2 group, depicting the protective features of Secang wood extract in overcoming the adverse effects of cigarette smoke exposure.
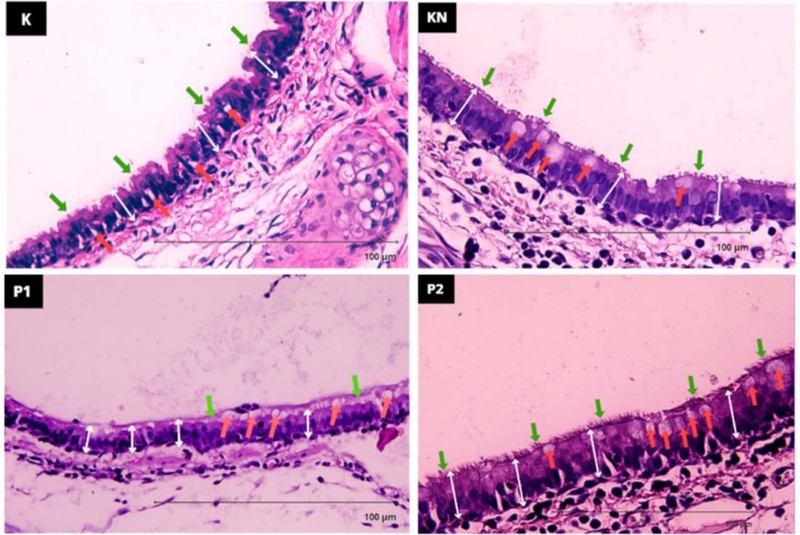
Rats’ tracheal histopathological structure after 30 days of treatment. Hematoxylin-eosin staining. K (control group), KN (negative control group, exposed to cigarette smoke), P1 (300 mg/kg of Secang wood extract and exposed to cigarette smoke), and P2 (600 mg/kg of Secang wood extract and exposed to cigarette smoke). Green arrows: ciliated cells, red arrows: goblet cells/hyperplasia, and white arrows: epithelial integrity. Magnification 40X objective, scale bars = 100 µm.
As pointed out by the red arrows, goblet cell hyperplasia was observed in the KN, P1, and P2 groups, suggesting chronic inflammatory conditions and epithelial damage due to cigarette smoke exposure. Goblet cells produce mucus, which is regulated by the secretory mucin gene, Mucin 5 subtype AC (MUC5AC).26 Prolonged chronic inflammation results in mucus overproduction, leading to the obstruction of the rats’ respiratory tract via ciliated epithelial cells, which increases epithelial growth factor receptor (EGFR) phosphorylation.27
The proposed mechanism by which hyperplasia of goblet cells occurs is as follows: EGFR activation by ligands such as EGF or transforming growth factor (TGF)-α triggers intracellular signaling cascades, including the activation of ERK pathway. Constitutive activation of EGFR due to cigarette smoking leads to increased expression of MUC5AC, which is a major mucin protein responsible for mucus production in the respiratory tract.28,29 Persistent activation of EGFR also contributes to goblet cell hyperplasia, exacerbating the respiratory condition.27
Earlier studies have shown that bioactive compounds in Secang wood, such as brazilin and brazilein, demonstrated potent antioxidant activity that mitigates oxidative stress and inflammation, reversing structural damage to the respiratory cells, including the trachea.30 Therefore, tracheal structural improvement in the ciliated epithelium and goblet cells can be observed in the P2 group.
As seen in Fig. 4A, exposure to free radicals and toxic compounds in cigarette smoke causes damage to tracheal cells, one of which is characterized by a smaller size in tracheal diameter as compared to the control. The lumen diameter (µm) of the trachea significantly (p < 0.05) decreased in KN (1604.0 ± 459.8) than in K (1956.0 ± 482.0), depicting cellular damage resulting from the cigarette smoke exposure. The P2 group showed a significant (p < 0.05) increase (1947.0 ± 570.2) in tracheal lumen diameter compared to KN, which indicated the protective effects of the antioxidant-rich compounds in Secang wood to counteract the adverse effects of cigarette smoke exposure.
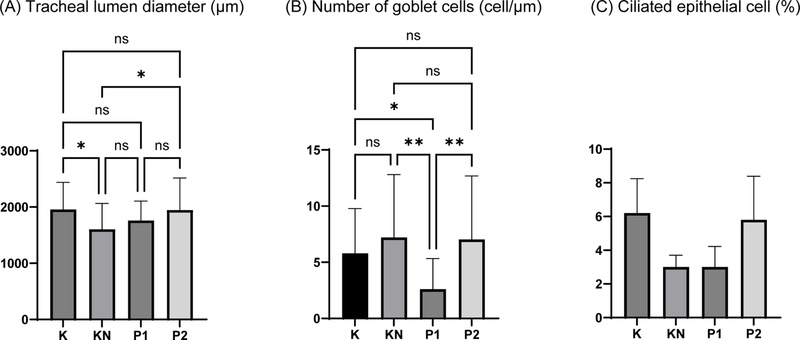
(A) Tracheal lumen diameter (µm). (B) Number of goblet cells (cell/µm). (C) Ciliated epithelium cell (%). K (control group), KN (negative control group, exposed to cigarette smoke), P1 (300 mg/kg of Secang wood extract and exposed to cigarette smoke), and P2 (600 mg/kg of Secang wood extract and exposed to cigarette smoke). Data were obtained from 6 animals in a group, with 5 technical replicates each, presented as mean ± SD. One-way ANOVA was performed for statistical analysis with Duncan’s post hoc test (*p < 0.05; **p < 0.01).
The number of goblet cells (Fig. 4B) tends to increase (ns, p > 0.05) in KN as compared to K, suggesting that toxic compounds and free radicals from cigarette smoke irritate respiratory cells in the trachea, leading to goblet cell hyperplasia. The number of goblet cells increased as a response to inflammatory conditions. Secang wood treatment in the P1 group significantly decreased the number of goblet cells as compared to K (p < 0.05) or KN (p < 0.01), which indicates effective mitigation of smoke-induced hyperplasia, likely through antioxidant-mediated reduction of oxidative stress and inflammatory signaling in tracheal epithelium.
In contrast, the average goblet cell number in the P2 group (600 mg/kg) was comparable to that of the KN. This pattern reflects a biphasic response where low-to-moderate doses optimally activate protective pathways, while higher doses reach saturation point. Brazilin has been reported to protect against oxidative damage through the activation of Nrf2/HO-1 signaling pathway in a dose-dependent manner at lower concentration, but no further protective effects observed at higher concentration.31,32
Some antioxidants display specific mechanisms to protect certain target cells. For instance, mitochondria-targeted antioxidants exhibited a higher protective effect than non-targeted cellular antioxidants for the treatment of chronic obstructive pulmonary disease.33 Antioxidants like N-acetylcysteine have shown specific protective effects on ciliated epithelium, thereby improving mucociliary clearance.34
Interestingly, Fig. 4C shows that while cigarette smoke caused ciliated epithelial damage in KN and P1 groups, the higher dose in P2 effectively preserved these structures (Fig. 3, green arrows). This suggests that the higher concentration of brazilin in P2 is required to provide sufficient Nrf2-mediated cytoprotection on the fragile ciliary cells.35 In contrast, the prominent reduction of goblet cells in P1 compared to P2 (Fig. 4B) depicted a counter-intuitive but biologically plausible result. While the P1 was insufficient to prevent initial ciliary shedding-resulting in thinner epithelial layers (Fig. 3, white arrows), it may have optimally triggered the anti-inflammatory pathway (NF-κB and ROS inhibition) that prevents basal cells from differentiating into mucus-producing goblet cells (Fig. 3, red arrows). This suggests a biphasic response, where a moderate dose of Secang (P1) is most effective at suppressing goblet cell hyperplasia, whereas the higher dose (P2) is primarily focused on preserving the existing physical barrier of the ciliated epithelium.31 These findings align with earlier reports which demonstrate that brazilin effectively inhibits MUC5AC expression by downregulating the oxidative and inflammatory signals that drive this specific cellular differentiation.32
Anatomically, the trachea is part of the upper respiratory tract after the nasal organs; hence, exposure to inhaled toxic substances can directly induce cell damage to the organ. A shrinking lumen diameter can result in increased airway resistance due to airway obstruction. Thus, the work of the respiratory muscles will increase as compensation to maintain adequate ventilation. Furthermore, the narrowing of the diameter of the tracheal lumen can lead to asphyxiation conditions due to reduced oxygen levels entering the respiratory tract and lungs.36
Earlier reports have suggested that the mechanism by which cigarette smoke induces airway remodelling, leading to a significantly reduced lung function, was through an upregulation of matrix metalloproteinase 9 (MMP-9).37,38 This enzyme plays an important role in various physiological processes, including tissue damage, inflammation, and asthma. Interestingly, bioactive compound in Secang wood, including brazilin and brazilein, inhibits the production of MMPs, including MMP-9.39
The observed histological improvements in the tracheal tissue of Secang-treated groups, including increased lumen diameter, reduced goblet cell numbers, and preserved ciliated epithelium, are consistent with mitigation of cigarette smoke-induced irritation and oxidative damage. Earlier studies underlined the potent antioxidant activity of Secang through various mechanisms including scavenging free radicals, reducing lipid peroxidation and upregulating endogenous antioxidant enzymes, e.g. quinone oxidoreductase (NQO1) and superoxide dismutase (SOD), which is also biomarkers of oxidative stress.35,40 These mechanisms likely contribute inferentially to the protective effects seen in this study, as similar histological amelioration has been linked to antioxidant interventions.40 However, the absence of inflammatory biomarkers assessment in this study limits mechanistic confirmation. Future studies incorporating such assays would strengthen the evidence for this antioxidant-mediated protection.
Acknowledgments
This study was supported by a research grant from LPPM Universitas Ahmad Dahlan, number: PD-031/SP3/LPPM-UAD/XI/2024.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
-
Wihatno, E.; Asri, Y.; Kushayati, N.; Parahita, D. P. Care J. Ilm. Ilmu Kesehat. 2025, 13, 30–41.
[https://doi.org/10.33366/jc.v13i1.6704]

-
Effendi, D. E.; Ardani, I.; Handayani, S.; Agustiya, R. I.; Nugroho, A. P.; Oktriyanto; Pramita, A.; Febriyanty, D.; Novita, R.; Yulianto, A. Clin. Epidemiol. Glob. Health 2024, 28, 101672.
[https://doi.org/10.1016/j.cegh.2024.101672]

- World Health Organization. WHO Report on the Global Tobacco Epidemic: Protect People from Tobacco Smoke; World Health Organization: Geneva, Switzerland, 2023.
-
Soleimani, F.; Dobaradaran, S.; De-la-Torre, G. E.; Schmidt, T. C.; Saeedi, R. Sci. Total Environ. 2022, 813, 152667.
[https://doi.org/10.1016/j.scitotenv.2021.152667]

-
Raharja, K. T.; Susilawati, E. F.; Romadhoni, I. F. J. SAGO Gizi Kesehat. 2024, 5, 702–709.
[https://doi.org/10.30867/gikes.v5i3A.1712]

-
Hadi, P.; Markad, P. Int. J. Health Sci. 2023, 6, 7023–7031.
[https://doi.org/10.53730/ijhs.v6nS8.14124]

-
Wirawati, K. T.; Ompusunggu, G. B.; Wardani, L.; Yanti, K. R. D. Pharm. Rep. 2023, 3, 86–86.
[https://doi.org/10.51511/pr.86]

-
Artati; Pratama, R.; Nurisyah, N.; Asyikin, A.; Abdullah, T.; Daswi, D. R.; Dewi, R. J. Penelit. Pendidik. IPA 2025, 11, 918–929.
[https://doi.org/10.29303/jppipa.v11i2.10563]

-
Vij, T.; Anil, P. P.; Shams, R.; Dash, K. K.; Kalsi, R.; Pandey, V. K.; Harsanyi, E.; Kovacs, B.; Shaikh, A. M. Molecules 2023, 28, 1–22.
[https://doi.org/10.3390/molecules28176247]

-
Hadi, K.; Setiami, C.; Azizah, W.; Hidayah, W.; Fatisa, Y. Photon: J. Sain Kesehat. 2023, 13, 48–59.
[https://doi.org/10.37859/jp.v13i2.4552]

-
Wira, A.; Winaya, I. B. O.; Adi, A. A. A. M. Indones. Medicus Vet. 2018, 7, 422–432.
[https://doi.org/10.19087/imv.2018.7.4.422]

- Mulka, K. R.; Gruenwald, R. C.; Yang, T. S.; Caswell, J. L. J. Vet. Diagn. Invest. 2026, 10406387251413159.
-
Nirmal, N. P.; Rajput, M. S.; Prasad, R. G. S. V.; Ahmad, M. Asian Pac. J. Trop. Med. 2015, 8, 421–430.
[https://doi.org/10.1016/j.apjtm.2015.05.014]

-
Septiyani, R.; Wikandari, R.; Santoso, U.; Raharjo, S. Braz. J. Food Technol. 2025, 28, 1–15.
[https://doi.org/10.1590/1981-6723.13024]

-
Septiyani, R.; Wikandari, R.; Santoso, U.; Raharjo, S. Trends Sci. 2024, 21, 1–14.
[https://doi.org/10.48048/tis.2024.8535]

-
Insuan, W.; Sillawatthumrong, N.; Chahomchuen, T.; Khamchun, S.; Chueahongthong, F.; Insuan, O. Disc. Appl. Sci. 2024, 6, 509.
[https://doi.org/10.1007/s42452-024-06222-4]

- Wawryk-Gawda, E.; Chylińska-Wrzos P.; Zarobkiewicz, M. K.; Chłapek K.; Jodłowska-Jędrych, B. Exp. Ther. Med. 2020, 19, 2826–2832.
-
Reitman, S.; Frankel, S. Am. J. Clin. Pathol. 1957, 28, 56–63.
[https://doi.org/10.1093/ajcp/28.1.56]

-
Nurrohiim, A.; Setiawan, H.; Wardani, D. K.; Nurazizah, I. F.; Azali, A. S. Berit. Biol. 2024, 23, 73–82.
[https://doi.org/10.55981/beritabiologi.2024.2063]

-
Salma, N. A.; Tukiran; Sutoyo, S.; Sabila, F. I. Folia Med. Indones. 2023, 59, 205–213.
[https://doi.org/10.20473/fmi.v59i3.44455]

-
Rosyidah, N.; Widyastuti, E.; Rahman, A. A.; Handayani, N. S.; Belgis. J. Vocat. Health Stud. 2024, 8, 1–6.
[https://doi.org/10.20473/jvhs.V8.I1.2024.1-6]

-
Indrajaya, P.; Mudawamah, M.; Kalsum, U.; Trisakti, A. R.; Rahmatullah, A. A. J. Basic Med. Vet. 2025, 14, 79–88.
[https://doi.org/10.20473/jbmv.v14i1.72636]

-
Holidah, D.; Dewi, I. P.; Siregar, I. P. A.; Aftianingsih, D. Galenika J. Pharm. 2022, 8, 1–9.
[https://doi.org/10.22487/j24428744.2022.v8.i1.15601]

- Rutledge, S. M.; Asgharpour, A. Gastroenterol. Hepatol. (N. Y.) 2020, 16, 617–625.
-
Strzelak, A.; Ratajczak, A.; Adamiec, A.; Feleszko, W. Int. J. Environ. Res. Public Health 2018, 15, 1033.
[https://doi.org/10.3390/ijerph15051033]

-
Li, J.; Ye, Z. Molecules 2020, 25, 4437.
[https://doi.org/10.3390/molecules25194437]

-
Shao, M. X. G.; Nakanaga, T.; Nadel, J. A. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 287, L420–L427.
[https://doi.org/10.1152/ajplung.00019.2004]

-
Filosto, S.; Becker, C. R.; Goldkorn, T. Mol. Cancer Ther. 2012, 11, 795–804.
[https://doi.org/10.1158/1535-7163.MCT-11-0698]

-
Kang, J. H.; Lee, E. H.; Park, S. W.; Chung, I. Y. J. Immunol. 2011, 187, 222–229.
[https://doi.org/10.4049/jimmunol.1003606]

-
Syamsunarno, M. R. A.; Safitri, R.; Kamisah, Y. Front. Pharmacol. 2021, 12, 725745.
[https://doi.org/10.3389/fphar.2021.725745]

-
Xu, W.; Lu, H.; Yuan, Y.; Deng, Z.; Zheng, L.; Li, H. Foods 2022, 11, 2439.
[https://doi.org/10.3390/foods11162439]

-
Pyun, B.-J.; Jo, K.; Lee, J. Y.; Lee, A.; Jung, M.-A; Hwang, Y.-H.; Jung, D. H.; Ji, K.-Y.; Choi, S.; Kim, Y. H.; Kim, T. Antioxidants 2022, 11, 1–16.
[https://doi.org/10.3390/antiox11112256]

-
Fairley, L. H.; Das, S.; Dharwal, V.; Amorim, N.; Hegarty, K. J.; Wadhwa, R.; Mounika, G.; Hansbro, P. M. Antioxidants 2023, 12, 973.
[https://doi.org/10.3390/antiox12040973]

-
Roe, T.; Talbot, T.; Terrington, I.; Johal, J.; Kemp, I.; Saeed, K.; Webb, E.; Cusack, R.; Grocott, M. P. W.; Dushianthan, A. Crit. Care 2025, 29, 68.
[https://doi.org/10.1186/s13054-025-05286-x]

-
Qi, B.; Zhang, X.; Yu, H.; Bao, Y.; Wu, N.; Jia, D. Ann. Transl. Med. 2021, 9, 312.
[https://doi.org/10.21037/atm-20-4414]

- Diaz, A. A.; Rahaghi, F. N.; Ross, J. C.; Harmouche, R.; Tschirren, J.; Estepar, R. S. J.; Washko, G. R. Respir. Res. 2015, 16, 23.
-
Louhelainen, N.; Stark, H.; Mazur, W.; Rytilä, P.; Djukanovic, R.; Kinnula, V. L. BMC Pulm. Med. 2010, 10, 13.
[https://doi.org/10.1186/1471-2466-10-13]

-
Zhu, W.; Han, L.; Wu, Y.; Tong, L.; He, L.; Wang, Q.; Yan, Y.; Pan, T.; Shen, J.; Song, Y.; Shen, Y.; Zhu, Q.; Zhou, J. Respir. Res. 2023, 24, 297.
[https://doi.org/10.1186/s12931-023-02598-w]

-
Laksmiani, N. P. L.; Leliqia, N. P. E.; Paramita, N. L. P. V.; Arijana, I. G. K. N.; Wijayanti, N. P. A. D.; Adiwibawa, P. I.; Putra, I. M. H.; Pratama, I. P. A. A. C. J. Pharm. Pharmacogn. Res. 2022, 10, 922–937.
[https://doi.org/10.56499/jppres22.1456_10.5.922]

-
Badami, S.; Moorkoth, S.; Rai, S.; Kannan, E.; Bhojraj, S. Biol. Pharm. Bull. 2003, 26, 1534–1537.
[https://doi.org/10.1248/bpb.26.1534]
