
Bioactivity and Chemical Composition of Essential Oils from Three Chemotypes of Aeollanthus pubescens Benth
Abstract
Aeollanthus pubescens Benth. (Lamiaceae) is a herbaceous plant that is native to West Africa. It is commonly used in cooking and traditional healing in some parts of Togo and Benin. This study analyzed the chemical compositions of essential oils obtained from different parts of Togo (Kozah, Est-Mono, and Ogou) using gas chromatography-mass spectrometry. Additionally, the study investigated the bioactivity of essential oils and water extracts on the cowpea weevil Callosobruchus maculatus, plant parasitic nematode Meloidogyne incognita, and two plant seeds, Sesamum indicum, and Pennisetum glaucum, regarding germination inhibition. The results showed that the major components of the different essential oils were thymol (55.02%) and thymol acetate (20.03%) for Kozah, D-fenchone (65.05%) for Est-Mono, and thymol (29.48%), carvacrol (20.93%), thymol acetate (16.98%), and carvacryl acetate (13.14%) for Ogou. The Est-Mono essential oil was the most effective in controlling cowpea weevils with an EC50 value of 3.1 ± 0.6 µL/L air. Kozah and Ogou essential oils showed more potent nematicidal activity with EC50/72h values of 305.4 ± 7.3 mg/L and 275.6 ± 2.6 mg/L, respectively, while Est-Mono essential oil did not exhibit any activity. Finally, the Kozah and Ogou essential oils were found to inhibit the germination of dicotyledonous S. indicum seeds by more than 80% at 10 mg/mL, while less germicidal activity was observed on the monocotyledon P. glaucum from the different essential oils. These findings suggest that extracts of A. pubescens could be used in agriculture as a biopesticide.
Keywords:
Aeollanthus pubescens essential oils, bioactivity, cowpea weevil, Meloidogyne spp, gas chromatography-mass spectrometry, polar metabolites, column chromatographyIntroduction
The Pubescens genus belongs to the Lamiaceae family and comprises more than 40 species, all of which are native to Africa. Aeollanthus pubescens Benth. is an annual herbaceous plant with a strong aroma, but it has received relatively little attention in research. It is mainly found in savannas and rocky mountainous areas in Togo and Benin,1 and is commonly used as a spice or green vegetable in local cuisine, as well as in traditional medicine to treat diarrhea and infections.2 The chemical composition of A. pubescens essential oil strongly depends on the area of cultivation, resulting in different chemotypes, including thymol, carvacrol, thymyle acetate, and D-fenchone, as reported by Koba et al.3 However, there is no comparative study on the bioactivity of these chemotypes.
Recently, a growing number of essential oils (EOs) from plants have been tested against a wide range of pest insects, showing promising results due to their high effectiveness, different mechanisms of action, low toxicity on non-target vertebrates, and potential valorization of their byproducts as nanopesticides.4 The need to find alternatives to synthetic pesticides, which have harmful effects on human health and the environment, has encouraged research on biopesticides. A. pubescens EO, with its interesting bioactive components, could be valorized in agriculture as a potential natural pest control agent. Mawussi et al.5 demonstrated the insecticidal activity of A. pubescens against the coffee berry borer, while Bohounton et al.6 reported its effectiveness against the mosquito A. gambiae. However, there is no report on the nematicidal and germicidal activities of the plant EO, even though phytoparasitic nematodes cause severe damage and yield losses to numerous agricultural crops, and effective control measures are lacking due to the phaseout of synthetic nematicides.
In recent years, there has been an increasing interest in the development of eco-friendly and sustainable methods for controlling plant diseases and pests.7-9 Biocontrol agents such as plant essential oils have shown promising results in controlling plant pathogens and pests. The use of A. pubescens EO as a biocontrol agent against phytopathogenic fungi and bacteria could be an alternative approach to the use of synthetic chemical pesticides. In addition, the use of A. pubescens EO as a plant growth promoter has been reported in some studies, indicating its potential use in sustainable agriculture. Therefore, further studies on the nematicidal and germicidal activities of A. pubescens EO could provide important insights into its potential as a biocontrol agent against plant pathogens and pests, as well as its potential use as a plant growth promoter.
Following our research works on developing alternatives to synthetic pesticides,10-13 1) we extracted and analyze chemical composition of different chemotypes of A. pubescens; 2) tested the EOs and total extracts on Meloidogyne spp nematodes, plant seeds for germicide activity and on cowpea weevil, C. maculatus and 3) discussed results according to the different chemotypes composition.
Experimental
Plant materials – A. pubescens whole plants were collected from three localities of Togo: Ogou, Est-Mono in the South and Bebda in the North in August 2021. Plant samples were transferred to laboratory and authenticated at the Botany Department, Faculty of Science, Lomé University, where voucher specimens were deposited. Fresh plant samples were subjected to steam distillation using a modified Clevenger-type apparatus to obtain the volatile constituents. Anhydrous Na2SO4 was added to remove excess moisture in essential oils after extraction. The oil yield (% v/w) was calculated on a dry weight basis. The extracted oils were stored in tightly closed dark bottles under refrigeration at 4oC until analyzed and tested.
Hydroalcoholic extract of A. pubescens was obtained by adding 250 mL of a mixture of ethanol/water (80/20, v/v) to 25 g of the dry plant aerial part harvested at Ogou. The mixture was sonicated for 30 min by a KQ-3200E Ultrasonicator (Wuxi City Bleecker Trading Co., Jiangsu, China) and kept for 48 h. After maceration, the extract was evaporated under reduced pressure to yield the hydroalcoholic extract. The sample was stored in tightly closed dark bottles under refrigeration at 4oC until analyzed and tested.
GC-MS analysis – The method employed for analyzing the essential oils involved the use of a TRACE 1300 Series GC chromatograph equipped with a split/splitless injector and a DB5-MS capillary column (50 m × 0.25 mm ID, 0.25 µm film thickness) for achieving separations. Helium gas was utilized as the carrier gas with a flow rate of 1 mL/min. The essential oil was diluted with hexane by dissolving 1 µL of EO in 20 mL of hexane. One microliter of the sample was injected. The injector used was an Auto sampler AIS/AS 1310, which was maintained at a temperature of 250oC in splitless mode. The GC oven temperature was programmed initially at 120oC for 10 min, followed by a temperature ramp from 120 to 210oC at a rate of 3oC/min, and held constant at 210oC for 10 min. Finally, the temperature was increased to 300oC at a rate of 5oC/min and held isothermal for 2 min. The MS detection was performed by coupling an ISQ MS to the GC, which had an ionization energy of 70 eV. Data acquisition was carried out using XCalibur software, with MS spectra obtained at a range width of m/z 40–450, an interface temperature of 255oC, ion source temperature of 210oC, solvent delay of 3 min, and scan speed of 2500. Identification of compounds was done based on their MS data (Mass Spectral Library NIST08 MS), linear retention indices, and comparison with authentic standards (when available).
Column chromatography – In the current investigation, the hydroalcoholic crude extract of A. pubescens collected from Ogou was subjected to column chromatography for obtaining various fractions. The elution was carried out using hexane followed by a hexane: methyl tert-butyl ether (MTBE) mixture with increasing polarity, and the obtained fractions were tested using TLC. The column was washed using ethyl acetate. The fractions exhibiting a similar pattern in TLC were combined and concentrated. The composition of the fractions collected during the column chromatographic separation was analyzed using GC-MS to determine the main compounds. The most promising fractions identified via GC-MS were evaluated for their efficacy against Meloidogyne spp. nematodes using the following method.
Nematode Paralysis Bioassays – A population of Meloidogyne spp. was collected from the roots of tomatoes (Solanum lycopersicum Mill.) grown for 2 months in the botanic garden of University of Kara, Kara, Togo. Batches of 30 egg masses (averaging 4500 eggs/batch) were collected from infected tomato roots and placed on 2 cm sieves (215 μm diameter), positioned on a 3.5 cm Petri dish. Distilled water was used as a natural hatching agent.14 The eggs were then incubated in a growth chamber at 25 ± 2oC, in the dark. First-emerging second-stage juveniles (J2) were discarded, and only second-stage juveniles collected after 2 days were used for the experiments.
All the 3 chemotype essential oils or hydroalcoholic extract were tested on nematodes in a dose range of 500–2000 mg/L using fosthiazate as a chemical control. Stock solutions were prepared in ethanol to overcome insolubility, using aqueous Tween 20 (0.1% v/v) for further dilutions. The final concentration of ethanol in each well never exceeded 1% (v/v) because preliminary experiments showed that this concentration was not toxic to the nematodes.15 Distilled water as well as a mixture of ethanol and aqueous Tween 20 at 0.3% (v/v) served as controls. In all cases, working solutions were prepared containing twice the test concentration and mixed in 96-well cell culture plates at a ratio of 1:1 (v/v) with a suspension of 25 nematodes added to each well. Multiwell plates were covered to avoid evaporation and maintained in the dark at 20oC. Juveniles were observed with the aid of an inverted microscope (Zeiss, Gottingen, Germany) at 4× after 3 days. Nematodes were at this point moved to plain water after washing through a 20 μm sieve to remove the excess test solutions. Numbers of motile and paralyzed nematodes were assessed by pricking the juvenile body with a needle, and they were counted. Paralysis experiments were performed three times, and every treatment was replicated six times. Mortality percentage of nematode was corrected in respect to the negative control using Abbott formula.16
Insecticidal activity on Callosobruchus maculatus – Adults weevil of the reproductive form of C. maculatus aged from 0 to 24 h were used for various tests. These adults come from the strain of infested cowpea seeds (Vigna unguiculata) called “Black eye” bought in a local market. It was cultivated on the local cowpea seeds frozen for a week to eliminate any previous contamination, and dried to maintain their moisture content at 13%. Insect rearing was done according to our previous work12 in Plexiglas boxes in the laboratory at 30 ± 2oC; 72 ± 2% RH 12: 12 h LD.
Toxicity tests were conducted through fumigation in sealed 1 L glass jars at room temperature and under natural photoperiod. To facilitate the evaporation of the EOs into the jars, Whatman filter paper discs with a diameter of 5.8 cm were loaded with EO and placed in the jars. The different concentrations of all EOs used were expressed as the volume of EO per volume of air in the enclosure (µL/L) and applied in a single dose. To determine the LC50 values, 20 pairs of C. maculatus were exposed to 5 concentrations of EO (3, 5, 8, 10, and 14 µL/L). The EO concentrations were normalized against the control (0 µL/L), which consisted of only the Whatman filter paper disc without any EO. After 24 hours of treatment, the adults were removed from the jars and placed in Petri dishes for another 24 hours. Fumigant experiments were performed at least twice, and every treatment was replicated six times. The number of dead adults was counted to determine the percentage of mortality and corrected in respect to the control using Abbott formula.16
Germicide activity – Experiments were conducted on Sesamum indicum and Pennisetum glaucum seeds as described previously.17 EOs were tested at 10 mg/mL. Twenty microliter of the test solution was deposited on a 2 cm Whatman paper and 500 µL distilled water was added in every well containing 10 seeds. Germination was monitored for 7 days, and the root and aerial part lengths measured at the end of the experiment for S. indicum and P. glaucum. For that, 25 plants with enough growth were selected randomly for each experiment, digitalized, and measured with the application ImageJ (http://rsb.info.nih.gov/ij/). Phytoxicity experiments were performed at least twice, and every treatment was replicated ten times.
Statistical analysis – The percentage of nematode and cowpea weevil adults paralysis were calculated, and data were subjected to one-way ANOVA followed by Chi2 multiple mean comparisons test. A P value < 0.05 was considered statistically significant. The LC50 values were estimated using a probit analysis of the dose-response data generated from the experiments. A nonparametric analysis of variance (ANOVA) was performed on growth inhibition and root and aerial length data.
Results and Discussion
Research on the benefits of aromatic plants and their applications are various, such as in medicine, phytopharmacy, cosmetics and food industries. Currently, natural products are considered to have very few side effects, as compared to their synthetic counterparts. Furthermore, the modern phytopharmaceutical industry is developing a large diversity of plant secondary metabolites to find new interesting molecules.18-20 Aromatic plants seem to be inexhaustible source, since only few plant species have been investigated in terms of phytochemical and pharmacological properties. Consequently, more scientific interest focused on aromatic plants and their derivatives, such as essential oils, known for their interesting biological properties should continue. In this work, chemical composition, insecticidal, nematicidal and germicidal activities of essential oils from A. pubescens harvested from different geographic areas of Togo are reported.
Extraction of essential oil using steam distillation showed yields of 1.3%, 1.4% and 1.5% for EOs from Est-Mono, Ogou and Kozah, respectively. It is noted few variations of yield according to sampling locations. These yields are similar to previous values (1.2 to 1.5%) found by Alitonou et al21 when they used hydrodistillation to extract A. pubescens EOs from different sampling places in Benin. Chemical composition showed an overall of 45 compounds that are reported in Table 1. Est-Mono essential oil showed D-Fenchone (67%) and limonene (12%) as major components. Major components of Ogou essential oil were thymol (29%), carvacrol (21%), carvacryl acetate (17%) and thymyl acetate (13%). For Kozah essential oil, the main components were p-cymene (9%), γ-terpinene (17%), thymol (51%) and thymol acetate (6%). Results demonstrated that chemical composition of A. pubescens strongly depend on the area of sampling. Three chemotypes were clearly unidentified: D-fenchone, thymol and carvacrol-thymol-carvacryl acetate chemotypes.
Chemical composition of essential oils extracted from A. pubescens harvested in three different geographic zones (Est Mono, Ogou and Kozah). Chromatographic percentage area of an average of three injections is reported for every component
When Koba et al. analyzed different essential oils (EOs) of A. pubescens harvested from seven different locations in Togo, they found that D-fenchone and phenol isomers carvacrol and thymol, along with their acetate derivatives, were the main components.3 These findings are consistent with our own results. Monoterpene phenols such as thymol and carvacrol are characteristic flavor compounds found in other well-known Lamiaceae plants like thyme and oregano. These specialized metabolites are known for their antibacterial, anti-spasmolytic, insecticidal, nematicidal, and antitumor activities.9, 22 The biosynthetic pathway of these compounds begins with the formation of γ-terpinene from geranyl diphosphate. An unstable intermediate is then utilized by P450 monooxygenases in conjunction with a dehydrogenase to form the aromatic backbone of thymol and carvacrol.23
Although the chemical composition of the essential oil of A. pubescens has been studied by many authors, the polar solvent extracts have not been well-investigated. Since the water decoction of the plant is widely used in traditional medicine, it is crucial to explore relatively more polar metabolites from A. pubescens. In this study, we report for the first time the identification of some polar compounds present in the aerial part of the plant through a hydroethanolic extraction, followed by fractionation using column chromatography on silica gel with mixtures of solvents of increasing polarity. After filtration and evaporation, we obtained 2.6 g of dry extracts, corresponding to a yield of 2.6%. We separated 415 mg of extracts on a column, which resulted in the isolation of 7 fractions (Table 2).

Masses of different fractions obtained by column chromatography of hydroethanolic extraction of A. pubescens from Ogou
To determine the structures of the different compounds in the obtained fractions, we performed GC/MS analyses. We were able to identify some major compounds based on retention times, mass spectra, and the mass spectra library of NIST of different compounds (Table 3). However, GC/MS was only used for the determination of less polar compounds. Further investigations using LC-MS and other techniques are necessary to identify the structure of the most polar compounds.
Fractions A-C were found to be the less polar fractions, and we identified most of the compounds from these fractions using GC-MS. In addition to the metabolites found in the essential oils, we identified other major phenolic compounds, including 8,9-dehydrothymol, 2,6-di-tert-butylbenzoquinone, 2,4-di-tert-butylphenol, 4-methoxy-2,3,6-trimethylphenol, and p-tert-pentylphenol. The presence of these compounds may justify the use of this plant as an antibiotic in traditional medicine, as many phenolic compounds have exhibited antibiotic effects.24 This is the first report of these phenols in A. pubescens, suggesting other potential uses of polar extracts from this plant.
All concentrations of the tested essential oils influenced the life of cowpea weevils compared to the control group (Table 4). Analysis of the one-way ANOVA variable and comparison of the mean values for each treatment using the Chi2 test at P ≤ 0.05 showed that there was a significant difference between the different treatments carried out with different chemotypes. There was also an interaction between the different concentrations and the mortality rate of the cowpea weevils (P ≤ 0.003). The effects of the EOs appeared to be dose-dependent, as indicated in Table 4, where the mortality rate caused by each essential oil increased with concentration and varied among the different chemotypes. At the lowest concentration (3 µL/L air), the D-fenchone chemotype caused a mortality rate of 32.34% on C. maculatus, whereas at this concentration, the thymol/carvacrol chemotype caused only 13.51% mortality and the thymol chemotype caused 15.27% mortality. At the highest concentration (14 µL/L air), all chemotypes had significantly higher activity compared to the control group and caused 100% mortality. The LD50 values (Table 5) varied depending on the chemotypes of the EOs.

Mean percentage mortality of different essential oils chemotypes of A. pubescens on C. maculatus at different concentrations

LC50 estimation by Probit analysis of different essential oil chemotypes of Aeollanthus pubescens on C. maculatus
The insecticidal activity of the A. pubescens EO from Kozah was found to be weaker against C. maculatus compared to the EOs from Ogou and Est-Mono, which exhibited stronger activity. This is consistent with the findings of Mbata et al.25 who reported over 80% mortality induced by L-fenchone at 8.3 µL/L against C. maculatus adults, which can be attributed to the strong activity of the D-fenchone-rich Ogou EO. In addition, Wahba et al.26 demonstrated that carvacrol was more toxic to C. maculatus than thymol. Our results are consistent with these findings, as the thymol/carvacrol essential oil chemotype from Ogou showed greater efficacy than the thymol/thymol acetate chemotype. To our knowledge, this study is the first report of A. pubescens essential oil chemotypes against C. maculatus. However, various authors have reported the effectiveness of the plant EO against other pests, including mosquitoes and coffee berry borers.5,6 Further research is necessary to determine the activity of thymol and carvacrol acetates present in the extracted EOs against pests.
Meloidogyne spp., commonly known as root-knot nematodes, are among the most destructive plant-parasitic nematodes due to their polyphagous nature. They are biotrophic endoparasites and can infect almost any higher plant species, making them ubiquitous worldwide. In this work, EOs and hydroethanolic extracts of A. pubescens were evaluated for their nematicidal activity. When tested on nematodes, only the Ogou essential oil chemotype of A. pubescens showed strong nematicidal activity, with 91 ± 5% mortality on Meloidogyne spp. at 2000 mg/L and an LC50 value estimated at 629 ± 39 mg/L. However, Est-Mono and Kozah EOs showed low activity on nematodes with 08 ± 3% and 25 ± 3% mortality, respectively. Previous research has shown that carvacrol and thymol have nematicidal activity, with LC50 values of 264 mg/L and 390 mg/L, respectively.27 In this study, the carvacrol chemotype of A. pubescens showed more activity than the thymol chemotype, and was the only one that demonstrated nematicidal activity with an LC50 value of 629 ± 39 mg/L. The D-fenchone chemotype EO did not show activity on Meloidogyne spp. when tested at 2000 mg/L, consistent with the findings of Rasoul et al.28 who found a very low activity of fenchone on M. incognita, with an LC50 value of 1675 mg/L. The carvacrol chemotype of A. pubescens was more effective than reported nematicidal plants such as Origanum dictamnus (LC50 = 1720 mg/L), O. vulgare (LC50 = 1550 mg/L), Zingiber officinale (LC50 = 832 mg/L), and Cinnamomum Zeylanicum (LC50 = 428 mg/L).15,27
It is interesting to note that the hydroethanolic extract of A. pubescens harvested from Ogou exhibited significant nematicidal activity, inducing 85% mortality of nematodes at a concentration of 2000 mg/L compared to the control. The presence of carvacrol and other phenolic compounds in the polar extract of A. pubescens may be responsible for its nematicidal activity. Further investigation is needed to fully understand the mechanism of action and potential uses of the EOs and hydroethanolic extract of A. pubescens as a natural nematicide.
To investigate the ability of A. pubescens essential oil to inhibit seed germination, germicidal activity was tested for the three essential oil chemotypes at 10 mg/mL. The results showed that all chemotypes inhibited seed germination after 24, 48, and 96 hours compared to the control group of water without essential oil. Analysis of variance (ANOVA) and Chi2 test at P ≤ 0.05 indicated a significant difference between the different treatments carried out with different chemotypes, as well as an interaction between the concentration and the percentage of inhibition (P ≤ 0.005). The effects of essential oils were found to be time-dependent, as the most effective chemotype from Ogou (carvacrol) showed over 80% inhibition of the dicotyledon S. radiatum seeds after 24 hours, while the D-fenchone chemotype was the least effective with only 25% inhibition at the same concentration (Fig. 1). On the other hand, for the monocotyledon P. glaucum (Fig. 2), the thymol chemotype was the most effective with over 80% seeds inhibition after 24 hours, while the D-fenchone chemotype was the least effective with 40% seeds inhibition. Most seeds germinated from all tested samples after 96 h but only controls and Kozah (D-fenchone) chemotype grown up to give leaves. We were then able to measure the grown inhibition induced by the D-fenchone chemotype on both tested seed plants. After 7 days, the EO showed –67 ± 8 and –32 ± 6% inhibition on S. radiatum root and leaf, respectively. However, the EO did not demonstrate inhibition on P. glaucum on root and aerial parts.

Phytotoxic effects (% inhibition) of A. pubescens on Sesamum radiatum seed germination. Bars represent the average relative values ± standard error. All measured values are significantly different from the control (P < 0.05), Chi2 significant differences test (LSD).
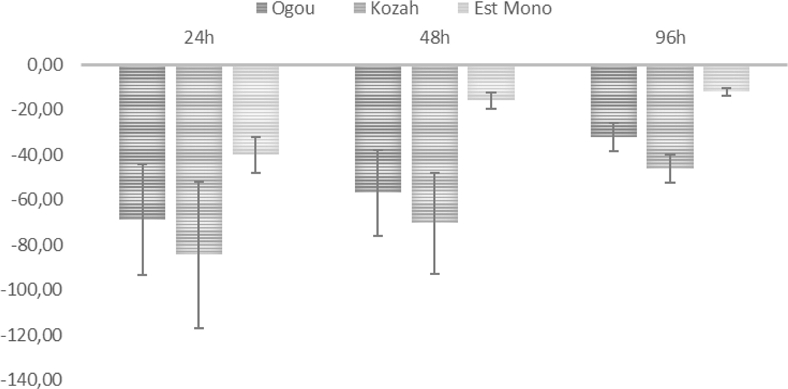
Phytotoxic effects (% inhibition) of A. pubescens on Pennisetum glaucum seed germination. Bars represent the average relative values ± standard error. All measured values are significantly different from the control (P < 0.05), Chi2 significant difference.
Carvacrol and thymol phytoxicity to seeds was reported by Pinheiro et al. on Lactuca sativa and Sorghum bicolor.29 Moreover, when the phytotoxicity of nineteen major components of essential oils was determined with rigid ryegrass (Lolium rigidum Gaudin), carvacrol and thymol showed similar germination index (89 and 86, respectively) while D-fenchone showed a germination index of 79.30 The germination indexes of the different essential chemotypes tested herein are justified by their respective chemical compositions. Of ten Peruvian Piper species studied by Ruiz-Vasquez et al., four plants showed more than 50% effective germination inhibition at 10 mg/mL31 demonstrating comparable activity with A. pubescens carvacrol and thymol chemotypes. This work demonstrates the potential herbicidal use of A. pubescens essential oils acclimatized in different geographic areas of Togo (West Africa).
In summary, the EOs extracted from A. pubescens demonstrated for the first-time insecticidal properties against C. maculatus, the cowpea weevil, across three different chemotypes (Thymol, carvacrol, and D-fenchone). Additionally, the carvacrol chemotype exhibited high nematicidal effects on root-knot nematodes. However, all three chemotypes displayed strong phytotoxicity on S. radiatum and P. glaucum seeds. The observed biological effects of the EOs were discussed in light of their chemical compositions. This study suggests the potential for developing new EO-based biopesticides using A. pubescens.
Acknowledgments
We gratefully acknowledge Prof. Komla Sanda (University of Kara, Togo) for providing us with valuable assistance in all steps of this work.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- Adjanohoun, E.; Adjakidje, V.; Ahyi, M.; Akpagana, K.; Chibon, P.; El-Hadji, A.; Eyme, J.; Garba, M.; Gassita, J.; Gbeassor, M., Contribution aux études ethnobotaniques et floristiques au Togo. Agence de coopération culturelle et technique (ACCT); From the data bank PHARMEL 2 (ref. HP 10): 1986, Paris, p 671.
-
Sanda, K.; Koba, K.; Walla, B. A.; Akpagana, K.; Garneau, F. X.; Gagnon, H.; Jean, F. I. J. Essent. Oil Res. 1999, 11, 257-258.
[https://doi.org/10.1080/10412905.1999.9701126]

-
Koba, K.; Sanda, K.; Raynaud, C.; Millet, J.; Chaumont, J. P. Comptes Rendus Chimie 2004, 7, 1107-1111.
[https://doi.org/10.1016/j.crci.2003.12.038]

-
Pavela, R.; Benelli, G. Trends Plant Sci. 2016, 21, 1000-1007.
[https://doi.org/10.1016/j.tplants.2016.10.005]

-
Mawussi, G.; Vilarem, G.; Raynaud, C.; Merlina, G.; Gbongli, A. K.; Wegbe, K.; Sanda, K. J. Essent. Oil Bearing Plants 2009, 12, 327-332.
[https://doi.org/10.1080/0972060X.2009.10643727]

-
Bohounton, R. B.; Djogbénou, L. S.; Djihinto, O. Y.; Dedome, O. S. L.; Sovegnon, P. M.; Barea, B.; Adomou, A.; Villeneuve, P.; Tchobo, F. P. Parasit. Vectors 2021, 14, 518.
[https://doi.org/10.1186/s13071-021-05012-w]

-
Ntalli, N. G.; Caboni, P. J. Agric. Food Chem. 2012, 60, 9929-9940.
[https://doi.org/10.1021/jf303107j]

-
Andres, M. F.; Coloma, A. G. In Biocontrol of Plant Disease: Recent Advances and Prospects in Plant Protection: Agro-industrial By-products and Waste as Sources of Biopesticides; Prigent-Combaret, C.; Dumas, B. Ed; John Wiley & Sons; USA, 2022, p 91.
[https://doi.org/10.1002/9781394188277.ch5]

-
Navarro-Rocha, J.; Andrés, M. F.; Díaz, C. E.; Burillo, J.; González-Coloma, A. Ind. Crops Prod. 2020, 145, 111958.
[https://doi.org/10.1016/j.indcrop.2019.111958]

-
Eloh, K.; Demurtas, M.; Deplano, A.; Ngoutane Mfopa, A.; Murgia, A.; Maxia, A.; Onnis, V.; Caboni, P. J. Agric. Food Chem. 2015, 63, 9970-9976.
[https://doi.org/10.1021/acs.jafc.5b04815]

-
Eloh, K.; Demurtas, M.; Mura, M. G.; Deplano, A.; Onnis, V.; Sasanelli, N.; Maxia, A.; Caboni, P. J. Agric. Food Chem. 2016, 64, 4876-4881.
[https://doi.org/10.1021/acs.jafc.6b02250]

- Etse, K. S.; Nébié, R.; Eloh, K.; Agbodan, A.; Dotse, K.; Koumaglo, K. J. Soc. Ouest-Afr. Chim. 2012, 34, 71-77.
-
Tocco, G.; Eloh, K.; Laus, A.; Sasanelli, N.; Caboni, P. J. Agric. Food Chem. 2020, 68, 11088-11095.
[https://doi.org/10.1021/acs.jafc.0c00835]

-
Aissani, N.; Urgeghe, P. P.; Oplos, C.; Saba, M.; Tocco, G.; Petretto, G. L.; Eloh, K.; Menkissoglu-Spiroudi, U.; Ntalli, N.; Caboni, P. J. Agric. Food Chem. 2015, 63, 6120-6125.
[https://doi.org/10.1021/acs.jafc.5b02425]

-
Eloh, K.; Kpegba, K.; Sasanelli, N.; Koumaglo, H. K.; Caboni, P. Int. J. Pest Manage. 2020, 66, 131-141.
[https://doi.org/10.1080/09670874.2019.1576950]

-
Rosenheim, J. A.; Hoy, M. A. J. Econ. Entomol. 1989, 82, 331-335.
[https://doi.org/10.1093/jee/82.2.331]

-
Santana, O.; Fe Andres, M.; Sanz, J.; Errahmani, N.; Abdeslam, L.; Gonzalez-Coloma, A. Nat. Prod. Commun. 2014, 9, 1109-1114.
[https://doi.org/10.1177/1934578X1400900812]

- Zouari, N. Med. Aromat. Plants 2013, 2, 145.
-
Huong, L. T.; Phu, H. V.; Giang, L. D.; Chau, D. T. M.; Ogunwande, I. A. J. Essent. Oil Bearing Plants 2022, 25, 1289-1300.
[https://doi.org/10.1080/0972060X.2022.2159542]

-
Zhaleh, M.; Sohrabi, N.; Zangeneh, M. M.; Zangeneh, A.; Moradi, R.; Zhaleh, H. J. Essent. Oil Bearing Plants 2018, 21, 493-501.
[https://doi.org/10.1080/0972060X.2018.1462739]

-
Alitonou, G.; Tchobo, F.; Avlessi, F.; Sohounhloue, D. K.; Menut, C. J. Essent. Oil Bearing Plants 2013, 16, 308-314.
[https://doi.org/10.1080/0972060X.2013.813203]

-
Kordali, S.; Cakir, A.; Ozer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Bioresour. Technol. 2008, 99, 8788-8795.
[https://doi.org/10.1016/j.biortech.2008.04.048]

-
Krause, S. T.; Liao, P.; Crocoll, C.; Boachon, B.; Förster, C.; Leidecker, F.; Wiese, N.; Zhao, D.; Wood, J. C.; Buell, C. R.; Gershenzon, J.; Dudareva, N.; Degenhardt, J. Proc. Natl. Acad. Sci. U S A 2021, 118, e2110092118.
[https://doi.org/10.1073/pnas.2110092118]

-
Gao, J.; Yang, Z.; Zhao, C.; Tang, X.; Jiang, Q.; Yin, Y. Sci. China Life Sci. 2022, 66, 1518-1534.
[https://doi.org/10.1007/s11427-022-2246-4]

-
Mbata, G. N.; Payton, M. E. J. Stored Prod. Res. 2013, 53, 43-47.
[https://doi.org/10.1016/j.jspr.2013.02.001]

- Wahba, T. F.; Mackled, M. I.; Selim, S.; El-Zemity, S. R. Middle East J. Appl. Sci. 2018, 8, 1061-1070.
-
Ntalli, N. G.; Ferrari, F.; Giannakou, I.; Menkissoglu-Spiroudi, U. J. Agric. Food Chem. 2010, 58, 7856-7863.
[https://doi.org/10.1021/jf100797m]

-
Abdel Rasoul, M. A. J. Plant Prot. Pathol. 2013, 4, 445-456.
[https://doi.org/10.21608/jppp.2013.87391]

-
Pinheiro, P. F.; Costa, A. V.; Alves, T. d. A.; Galter, I. N.; Pinheiro, C. A.; Pereira, A. F.; Oliveira, C. M. R.; Fontes, M. M. P. J. Agric. Food Chem. 2015, 63, 8981-8990.
[https://doi.org/10.1021/acs.jafc.5b03049]

-
Vasilakoglou, I.; Dhima, K.; Paschalidis, K.; Ritzoulis, C. J. Essent. Oil Res. 2013, 25, 1-10.
[https://doi.org/10.1080/10412905.2012.751054]

-
Ruiz-Vásquez, L.; Ruiz Mesia, L.; Caballero Ceferino, H. D.; Ruiz Mesia, W.; Andrés, M. F.; Díaz, C. E.; Gonzalez-Coloma, A. Plants 2022, 11, 1793.
[https://doi.org/10.3390/plants11141793]
